Theoretical problems
... The acids which are stronger than pure sulfuric acid are called superacids. Superacids are very strong proton donors being capable of protonating even weak Lewis acids such as Xe, H2, Cl2, Br2, and CO2. Cations, which never exist in other media, have been observed in superacid solutions. George Olah ...
... The acids which are stronger than pure sulfuric acid are called superacids. Superacids are very strong proton donors being capable of protonating even weak Lewis acids such as Xe, H2, Cl2, Br2, and CO2. Cations, which never exist in other media, have been observed in superacid solutions. George Olah ...
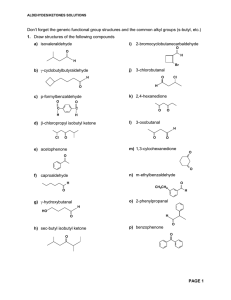
Aldehydes/Ketones Solutions
... 6. Write equations showing how the following transformations can be carried out. No mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
... 6. Write equations showing how the following transformations can be carried out. No mechanisms required but show all reagents and intermediate products formed. More than one step may be necessary a) O O C H ...
Aluminum and Copper
... Possible signs of a chemical reaction include color changes, temperature changes, formation of a precipitate, and gas evolution. The reaction of aluminum and copper(II) chloride is very vigorous—the reaction mixture gets very hot as heat is released, the blue color due to the Cu(II) ions fades, t ...
... Possible signs of a chemical reaction include color changes, temperature changes, formation of a precipitate, and gas evolution. The reaction of aluminum and copper(II) chloride is very vigorous—the reaction mixture gets very hot as heat is released, the blue color due to the Cu(II) ions fades, t ...
The Major Classes of Chemical Reactions
... do not conduct an electric current, these substances are called nonelectrolytes. Many other covalent substances, such as benzene (C6H6) and octane (C8H18), do not contain polar bonds, and these substances do not dissolve appreciably in water. A small, but very important, group of H-containing covale ...
... do not conduct an electric current, these substances are called nonelectrolytes. Many other covalent substances, such as benzene (C6H6) and octane (C8H18), do not contain polar bonds, and these substances do not dissolve appreciably in water. A small, but very important, group of H-containing covale ...
ordinary level chemistry syllabus
... to use independent approaches to solve problems. Students come to know and explore the properties of substances, as well as the processes in which those substances take part, and of materials obtained through modern industry. 1.2.3. Competences Competence is defined as the ability to use an appropri ...
... to use independent approaches to solve problems. Students come to know and explore the properties of substances, as well as the processes in which those substances take part, and of materials obtained through modern industry. 1.2.3. Competences Competence is defined as the ability to use an appropri ...
Lab # 18
... To balance equations, we follow these four rules: 1. Equations must be balanced so that the number of atoms of each element is equal on the left side (reactants) and on the right side (products) of the reaction. 2. We MUST NOT change the subscripts of any of the reactants or products; if we did that ...
... To balance equations, we follow these four rules: 1. Equations must be balanced so that the number of atoms of each element is equal on the left side (reactants) and on the right side (products) of the reaction. 2. We MUST NOT change the subscripts of any of the reactants or products; if we did that ...
Organic Chemistry/Fourth Edition: e-Text
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
... Ethyl isopropyl ketone may be alternatively named 2-methyl-3-pentanone. Its longest continuous chain has five carbons. The carbonyl carbon is C-3 irrespective of the direction in which the chain is numbered, and so we choose the direction that gives the lower number to the position that bears the me ...
Organic Chemistry - University of California, Riverside
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
... With the exception of the protonated amines and alcohols just mentioned, all of the organic molecules that we have considered have no ionic charge so they are electrically neutral. A molecule is electrically neutral because the total number of its electrons (-1 charge) is equal to the number of prot ...
Full answers
... As the reaction is endothermic, the energy of the products is higher than that of the reactants. Would you expect the forward or the reverse reaction to be faster? Why? The backward reaction would be faster as it has a lower activation energy. This is a consequence of the reaction being endothermic. ...
... As the reaction is endothermic, the energy of the products is higher than that of the reactants. Would you expect the forward or the reverse reaction to be faster? Why? The backward reaction would be faster as it has a lower activation energy. This is a consequence of the reaction being endothermic. ...
File
... c) NH4Cl (s) NH3 (g) + HCl(g) 5. At 25 °C, Kc =0.0146 for the following reaction: PCl5 PCl3 + Cl2 If, at equilibrium, the molar concentrations for PCl5 and PCl3 are 0.500 M and 0.200 M respectfully, calculate the concentration of chlorine gas. (0.0365M) 6. Consider the reaction: CO + 2H2 CH ...
... c) NH4Cl (s) NH3 (g) + HCl(g) 5. At 25 °C, Kc =0.0146 for the following reaction: PCl5 PCl3 + Cl2 If, at equilibrium, the molar concentrations for PCl5 and PCl3 are 0.500 M and 0.200 M respectfully, calculate the concentration of chlorine gas. (0.0365M) 6. Consider the reaction: CO + 2H2 CH ...
Sample Chapter - Chapter 4
... In aqueous solution, these substances are all acids, as you’ll see shortly. The molecules contain polar bonds to hydrogen, in which the atom bonded to H pulls more strongly on the shared electron pair. A good example is hydrogen chloride gas. The Cl end of the HCl molecule is partially negative, and ...
... In aqueous solution, these substances are all acids, as you’ll see shortly. The molecules contain polar bonds to hydrogen, in which the atom bonded to H pulls more strongly on the shared electron pair. A good example is hydrogen chloride gas. The Cl end of the HCl molecule is partially negative, and ...
Equilibrium
... b. If raising the temperature of the reaction results in an equilibrium with a higher concentration of C than A, how will the value of Keq change? 12. The following reaction occurs when steam is passed over hot carbon. The mixture of gases it generates is called water gas and is useful as an indust ...
... b. If raising the temperature of the reaction results in an equilibrium with a higher concentration of C than A, how will the value of Keq change? 12. The following reaction occurs when steam is passed over hot carbon. The mixture of gases it generates is called water gas and is useful as an indust ...
1.24 calculations and chemical reactions
... A solution of this acid was prepared by dissolving 2.02 g of H2A in water and making the volume up to 250 cm3 in a volumetric flask. A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodi ...
... A solution of this acid was prepared by dissolving 2.02 g of H2A in water and making the volume up to 250 cm3 in a volumetric flask. A 25.0 cm3 sample of this solution required 22.80 cm3 of 0.150 mol dm–3 aqueous NaOH for complete reaction. Calculate the relative molecular mass, Mr, of H2A 4.2) Sodi ...
- Kendriya Vidyalaya Jhunjhunu
... Name the compounds X and Y. Also write their formulae. THREE MARKS QUESTIONS 1. How many structural isomers are possible for pentane? Draw the structural formulae of all the possible isomers of pentane 2. A compound X has molecular formula C4H10.It undergoes substitution reaction more readily than a ...
... Name the compounds X and Y. Also write their formulae. THREE MARKS QUESTIONS 1. How many structural isomers are possible for pentane? Draw the structural formulae of all the possible isomers of pentane 2. A compound X has molecular formula C4H10.It undergoes substitution reaction more readily than a ...
Synthetic Polymers - McQuarrie General Chemistry
... phenolics (brake linings and structural components). The essential feature in the synthesis of condensation polymers is the presence of reactive groups at both ends of the monomers. Table S.2 lists some industrially important polymers and their annual production by region. One of the most significan ...
... phenolics (brake linings and structural components). The essential feature in the synthesis of condensation polymers is the presence of reactive groups at both ends of the monomers. Table S.2 lists some industrially important polymers and their annual production by region. One of the most significan ...
Answers - University of Waterloo
... What is the final temperature when 100.0 mL of water at 90.0 °C and 200.0 mL of water at 10.0 °C are mixed? Assume no heat is lost to the surroundings. Choose the closest value. *A 40 oC ...
... What is the final temperature when 100.0 mL of water at 90.0 °C and 200.0 mL of water at 10.0 °C are mixed? Assume no heat is lost to the surroundings. Choose the closest value. *A 40 oC ...
Chapter 4 Chemical Reactions and Solution Stoichiometry 4.1
... the combination of two different compounds to form a new compound. For example, oxygen reacts with sulfur dioxide to make sulfur trioxide. 2 SO2(g) + O2(g) 2 SO3(g) Sulfur trioxide and water undergo a combination reaction to form sulfuric acid. SO3(g) + H2O() H2SO4(aq) Notice that in all combin ...
... the combination of two different compounds to form a new compound. For example, oxygen reacts with sulfur dioxide to make sulfur trioxide. 2 SO2(g) + O2(g) 2 SO3(g) Sulfur trioxide and water undergo a combination reaction to form sulfuric acid. SO3(g) + H2O() H2SO4(aq) Notice that in all combin ...
Advanced Practical Organic Chemistry
... atoms can occur within organic molecules, these groups of atoms are called functional groups. One good example is the hydroxyl functional group. The hydroxyl group consists of a single oxygen atom bound to a single hydrogen atom (-OH). The group of hydrocarbons that contain a hydroxyl functional gro ...
... atoms can occur within organic molecules, these groups of atoms are called functional groups. One good example is the hydroxyl functional group. The hydroxyl group consists of a single oxygen atom bound to a single hydrogen atom (-OH). The group of hydrocarbons that contain a hydroxyl functional gro ...
BC Science 10 Workbook Answers
... 3. Terms and arrows could be similar to Fig 2.17 on page 70. Students may also add other facts or effects that they have thought of. 4. Changes in the carbon, nitrogen, and phosphorus cycles can affect the health and variety of organisms that live in an ecosystem. 5. Answers will vary but they shoul ...
... 3. Terms and arrows could be similar to Fig 2.17 on page 70. Students may also add other facts or effects that they have thought of. 4. Changes in the carbon, nitrogen, and phosphorus cycles can affect the health and variety of organisms that live in an ecosystem. 5. Answers will vary but they shoul ...
AP Chemistry Lab Manual
... AP Chemistry class. As you record information in your notebook, keep in mind that someone who is unfamiliar with your work may be using this notebook to evaluate your lab experience in chemistry. When you explain your work, list your data, calculate values and answer questions, be sure that the mean ...
... AP Chemistry class. As you record information in your notebook, keep in mind that someone who is unfamiliar with your work may be using this notebook to evaluate your lab experience in chemistry. When you explain your work, list your data, calculate values and answer questions, be sure that the mean ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.