An efficient acetylation of dextran using in situ activated acetic
... has been developed for the acetylation of dextran. Dextran acetates were successfully synthesized using different molar ratios of acetic anhydride in the presence of iodine as a catalyst without the use of any solvent. The reactions were realized at 50 °C for 3 h under stirring and nitrogen. This ef ...
... has been developed for the acetylation of dextran. Dextran acetates were successfully synthesized using different molar ratios of acetic anhydride in the presence of iodine as a catalyst without the use of any solvent. The reactions were realized at 50 °C for 3 h under stirring and nitrogen. This ef ...
Covalently Bonded Platinum(II) Complexes of [alpha]
... are direct consequences of the steric and electronic environment around the observed nuclei, and different values are therefore usually obtained, depending on the R group attached to the organometallic site. Thus, peptide functionalization with these complexes provides a biomarker not only for bioch ...
... are direct consequences of the steric and electronic environment around the observed nuclei, and different values are therefore usually obtained, depending on the R group attached to the organometallic site. Thus, peptide functionalization with these complexes provides a biomarker not only for bioch ...
Final Exam - Dawson College
... Hydrofluoric acid, (HF) is a weak acid that can be used in the fluoridation of water. An aqueous solution of 0.100 M HF has an osmotic pressure of 2.64 atm at 25°C. a. Calculate the van’t Hoff factor for HF at this concentration ...
... Hydrofluoric acid, (HF) is a weak acid that can be used in the fluoridation of water. An aqueous solution of 0.100 M HF has an osmotic pressure of 2.64 atm at 25°C. a. Calculate the van’t Hoff factor for HF at this concentration ...
Use the following answers for questions 1
... (B) Carbon dioxide (C) Aluminum hydroxide (D) Ammonia (E) Hydrogen peroxide 4. Is a good oxidizing agent 5. Is used to etch glass chemically 6. Is used extensively for the production of fertilizers 7. Has amphoteric properties 63. Which of the following characteristics is common to elemental sulfur, ...
... (B) Carbon dioxide (C) Aluminum hydroxide (D) Ammonia (E) Hydrogen peroxide 4. Is a good oxidizing agent 5. Is used to etch glass chemically 6. Is used extensively for the production of fertilizers 7. Has amphoteric properties 63. Which of the following characteristics is common to elemental sulfur, ...
Review Answers - cloudfront.net
... Diamonds graphite is a spontaneous process, but the kinetics of the reaction cause it to happen very very slowly. ii. ...
... Diamonds graphite is a spontaneous process, but the kinetics of the reaction cause it to happen very very slowly. ii. ...
NO - Blue Devil Chem
... with concentrated sulfuric acid. Soon an exothermic reaction takes places during which a column of carbon rises from the beaker and a cloud of steam is produced. Concentrated sulfuric acid acts as a catalyst to dehydrate sucrose to produce carbon and water. The heat of the reaction vaporizes the wat ...
... with concentrated sulfuric acid. Soon an exothermic reaction takes places during which a column of carbon rises from the beaker and a cloud of steam is produced. Concentrated sulfuric acid acts as a catalyst to dehydrate sucrose to produce carbon and water. The heat of the reaction vaporizes the wat ...
Mechanistic Details of the Oscillatory Belousov
... reactive abstracting radical, e.g., Br*), the structure of the transition state is closer to that of the products than the reactants. Thus the stability of the product radical determines the rate. In contrast, the rates of exothermic H atom abstractions are rapid and not very sensitive to either the ...
... reactive abstracting radical, e.g., Br*), the structure of the transition state is closer to that of the products than the reactants. Thus the stability of the product radical determines the rate. In contrast, the rates of exothermic H atom abstractions are rapid and not very sensitive to either the ...
Introduction to Qualitative Analysis
... It is important to recognize the distinction between these groups and the groups of the Periodic Table (alkali metals, transition metals, etc.); the groups A-D do not necessarily correlate with groups in the Periodic Table. Periodic Table groups are based upon similarities in electron configurations ...
... It is important to recognize the distinction between these groups and the groups of the Periodic Table (alkali metals, transition metals, etc.); the groups A-D do not necessarily correlate with groups in the Periodic Table. Periodic Table groups are based upon similarities in electron configurations ...
Answers - Pearson-Global
... equal volumes of liquids, and equal volumes of water. All this stresses the importance of a “fair test”. Likely suggestions would involve having two tubes (gas jars, measuring cylinders, burettes, for example) of water with the coloured liquids introduced into the bottom of them. A simple observatio ...
... equal volumes of liquids, and equal volumes of water. All this stresses the importance of a “fair test”. Likely suggestions would involve having two tubes (gas jars, measuring cylinders, burettes, for example) of water with the coloured liquids introduced into the bottom of them. A simple observatio ...
Redox Reactions C12-1-10
... hence, an oxidizing agent and a reducing agent. This makes sense since as one reactant is losing electrons (being oxidized), the other is gaining electrons (being reduced) Oxidation numbers can be helpful in determining whether a reaction is redox or non-redox. When a change in oxidation number occu ...
... hence, an oxidizing agent and a reducing agent. This makes sense since as one reactant is losing electrons (being oxidized), the other is gaining electrons (being reduced) Oxidation numbers can be helpful in determining whether a reaction is redox or non-redox. When a change in oxidation number occu ...
Test-tube Reactions - University of Manitoba
... hence, an oxidizing agent and a reducing agent. This makes sense since as one reactant is losing electrons (being oxidized), the other is gaining electrons (being reduced) Oxidation numbers can be helpful in determining whether a reaction is redox or non-redox. When a change in oxidation number occu ...
... hence, an oxidizing agent and a reducing agent. This makes sense since as one reactant is losing electrons (being oxidized), the other is gaining electrons (being reduced) Oxidation numbers can be helpful in determining whether a reaction is redox or non-redox. When a change in oxidation number occu ...
Year 11 C2 Mock Exam Revision Questions
... Draw a ring around the correct answer in each box to explain why propane evaporates easily. ...
... Draw a ring around the correct answer in each box to explain why propane evaporates easily. ...
Activity C14: Rate of a Chemical Reaction 1
... In this activity you will determine the effect of changes in concentration of the reactants on the rate of the chemical reaction. The reaction for this activity is the acidic reduction of the thiosulfate ion to sulfur and sulfur dioxide. The equation for the reaction is: S2O32-(aq) + 2 H+(aq) ====== ...
... In this activity you will determine the effect of changes in concentration of the reactants on the rate of the chemical reaction. The reaction for this activity is the acidic reduction of the thiosulfate ion to sulfur and sulfur dioxide. The equation for the reaction is: S2O32-(aq) + 2 H+(aq) ====== ...
Exam Edge Digital
... The Chemical Bonding: Chemical Formulas chapter is important in that it teaches you several fundamental principles that apply to many aspects of chemistry. Questions on this chapter appear frequently in questions 4, 5, 10 and 11 of the exam paper. You should be able to draw diagrams to show the elec ...
... The Chemical Bonding: Chemical Formulas chapter is important in that it teaches you several fundamental principles that apply to many aspects of chemistry. Questions on this chapter appear frequently in questions 4, 5, 10 and 11 of the exam paper. You should be able to draw diagrams to show the elec ...
A flask contains 0
... If you have time after you did the starred () questions, go to the circle questions and look at them again…maybe a second time through will jog your memory. REMEMBER every time you turn the page…make sure that you have bubbled the correct number on the answer sheet. This way if you get off trac ...
... If you have time after you did the starred () questions, go to the circle questions and look at them again…maybe a second time through will jog your memory. REMEMBER every time you turn the page…make sure that you have bubbled the correct number on the answer sheet. This way if you get off trac ...
Chemical Equations and Reactions
... the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be recovered unchanged. ...
... the pressure at which the reaction occurs or to specify that the pressure must be above normal. Many reactions are speeded up and can take place at lower temperatures in the presence of a catalyst. A catalyst is a substance that changes the rate of a chemical reaction but can be recovered unchanged. ...
STUDY MATERIAL 2016-17 CHEMISTRY CLASS XII
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
Question Bank - Edudel.nic.in
... The well known mineral fluorite is chemically calcium fluoride. It is known that in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell ...
... The well known mineral fluorite is chemically calcium fluoride. It is known that in one unit cell of this mineral there are 4 Ca2+ ions and 8F– ions and that Ca2+ ions are arranged in a fcc lattice. The F– ions fill all the tetrahedral holes in the fcc lattice of Ca2+ ions. The edge of the unit cell ...
The Wizard Test Maker
... strong acid and a strong base? (A) When enough base is added to the solution, phenolphthalein acts as a catalyst for the color change. (B) Phenolphthalein changes the rate of reaction, causing a color change. (C) As the solution being titrated approaches its end point, a large change in pH takes pla ...
... strong acid and a strong base? (A) When enough base is added to the solution, phenolphthalein acts as a catalyst for the color change. (B) Phenolphthalein changes the rate of reaction, causing a color change. (C) As the solution being titrated approaches its end point, a large change in pH takes pla ...
P. Mignon, J. Steyaert, R. Loris, P. Geerlings, and S. Loverix, J. Biol
... Because RNase-catalyzed phosphodiester cleavage is not a dissociative process (16), the nucleophilic attack contributes largely to the rate-limiting step of the reaction. The nucleophilic attack on hard centers such as phosphates involves the donation of electrons from a nucleophile to a phosphorus, ...
... Because RNase-catalyzed phosphodiester cleavage is not a dissociative process (16), the nucleophilic attack contributes largely to the rate-limiting step of the reaction. The nucleophilic attack on hard centers such as phosphates involves the donation of electrons from a nucleophile to a phosphorus, ...
AQA GCSE Chemistry My Revision Notes
... Crude oil is used to produce many useful fuels, but oil reserves are being used up. One way to conserve crude oil reserves would be to increase the production of biofuels. Ethanol can be produced for use as a bio-fuel. Cars can be powered by ethanol or ethanol–petrol mixtures. Sugar cane can be ferm ...
... Crude oil is used to produce many useful fuels, but oil reserves are being used up. One way to conserve crude oil reserves would be to increase the production of biofuels. Ethanol can be produced for use as a bio-fuel. Cars can be powered by ethanol or ethanol–petrol mixtures. Sugar cane can be ferm ...
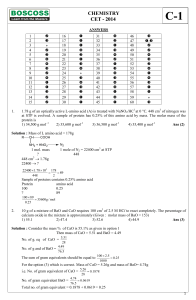
CHEMISTRY CET
... 'Z' is slowly passed into an aqueous solution of Y, colloidal sulphur is obtained. X and Z could be, respectively 1) Na2SO4, H2S 2) Na2SO4, SO2 3) Na2S, SO3 4) Na2SO3, H2S Ans (4) ...
... 'Z' is slowly passed into an aqueous solution of Y, colloidal sulphur is obtained. X and Z could be, respectively 1) Na2SO4, H2S 2) Na2SO4, SO2 3) Na2S, SO3 4) Na2SO3, H2S Ans (4) ...
5. Coenzyme HAD+ is derived
... Nomenclature. Isomerism. Mutual influence of atoms and how to transfer in biologically important molecules. Classification of organic reactions by type and mechanism. Reactivity of hydrocarbons. Radical substitution reaction. Of electrophilic substitution and addition. Acidity and basicity of organi ...
... Nomenclature. Isomerism. Mutual influence of atoms and how to transfer in biologically important molecules. Classification of organic reactions by type and mechanism. Reactivity of hydrocarbons. Radical substitution reaction. Of electrophilic substitution and addition. Acidity and basicity of organi ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.
![Covalently Bonded Platinum(II) Complexes of [alpha]](http://s1.studyres.com/store/data/022412983_1-66c66ee18551a43164a79702fd995f95-300x300.png)