Exp`t 88 - Chemistry Courses
... 1. The same experiment could be performed using acetic acid, isopropyl alcohol, and sulfuric acid; in this particular experiment, what advantage could acetic anhydride have over acetic acid? (Hint: consider what by-product is formed if acetic acid were used, and how this could affect the equilibrium ...
... 1. The same experiment could be performed using acetic acid, isopropyl alcohol, and sulfuric acid; in this particular experiment, what advantage could acetic anhydride have over acetic acid? (Hint: consider what by-product is formed if acetic acid were used, and how this could affect the equilibrium ...
AP Chemistry Test Review
... 34) Calculate K by doing ICE box problems 35) acid/base definitions…Bronsted-Lowry = acids donate protons; Lewis= acids accept e- pair 36) calculate pH, pOH, [H+], [OH−] 37) acid equilibrium problems…ICE box…remember pH can be used to find [H+]. 38) salt pH…example: Na2CO3 = slightly basic; Al(NO3)3 ...
... 34) Calculate K by doing ICE box problems 35) acid/base definitions…Bronsted-Lowry = acids donate protons; Lewis= acids accept e- pair 36) calculate pH, pOH, [H+], [OH−] 37) acid equilibrium problems…ICE box…remember pH can be used to find [H+]. 38) salt pH…example: Na2CO3 = slightly basic; Al(NO3)3 ...
Organic Chem Functional Groups
... catalyst such as Pt or Pd common application is the manufacture of margarine from unsaturated vegetable oils (making them solid from a liquid) ...
... catalyst such as Pt or Pd common application is the manufacture of margarine from unsaturated vegetable oils (making them solid from a liquid) ...
Acids, Bases, and Salts Section 1 Acids and Bases
... A strong base dissociates (or forms OH-) completely in solution Equations for strong acids and bases use a single arrow indicating ions are formed Strong acids and bases conduct more electricity than weak ones ...
... A strong base dissociates (or forms OH-) completely in solution Equations for strong acids and bases use a single arrow indicating ions are formed Strong acids and bases conduct more electricity than weak ones ...
CfE Higher Chemistry Homework Unit 2: Natures Chemistry
... butanone (a) Name the two products formed by the dehydration of butan-2-ol (b) Name a reagent which could be used to oxidise butan-2-ol to butanone. ...
... butanone (a) Name the two products formed by the dehydration of butan-2-ol (b) Name a reagent which could be used to oxidise butan-2-ol to butanone. ...
acids and bases - Althea`s Academy
... dissolved in water will furnish H1+ base – subs which contain OH group and when dissolved in water, will furnish OH12. Bronsted-Lowry Concept (protonic concept of acidity) ...
... dissolved in water will furnish H1+ base – subs which contain OH group and when dissolved in water, will furnish OH12. Bronsted-Lowry Concept (protonic concept of acidity) ...
Organic Chemistry I (CHEM 2010 and 2012)
... expected to read and study the material to be discussed prior to the lecture. This includes working in-chapter and end-of-chapter problems and exercises in the text. Students should review the material discussed until comprehension is acquired and seek assistance when necessary. It is also highly re ...
... expected to read and study the material to be discussed prior to the lecture. This includes working in-chapter and end-of-chapter problems and exercises in the text. Students should review the material discussed until comprehension is acquired and seek assistance when necessary. It is also highly re ...
Practice exam 1 - Little Dumb doctor, homework solutions
... Answer all of the questions in the spaces provided. Point values are in parentheses. Be sure to read the questions carefully. 1.(15) Propose the best possible Lewis-dot structures for the following species. Use lines for covalent bonds, show any formal charges where appropriate, and indicate the hyb ...
... Answer all of the questions in the spaces provided. Point values are in parentheses. Be sure to read the questions carefully. 1.(15) Propose the best possible Lewis-dot structures for the following species. Use lines for covalent bonds, show any formal charges where appropriate, and indicate the hyb ...
Practice with Chemical Equilibrium (Chapter 14) (Due 2/17)
... Here are four additional practice exercises concerning chemical equilibrium (Chapter 14). See your instructor if you have questions. Note that for these questions, the symbol "=" is used to indicate a reversible reaction. Your textbook uses a double-headed arrow. 1. Suppose that equal molar amounts ...
... Here are four additional practice exercises concerning chemical equilibrium (Chapter 14). See your instructor if you have questions. Note that for these questions, the symbol "=" is used to indicate a reversible reaction. Your textbook uses a double-headed arrow. 1. Suppose that equal molar amounts ...
T. V. RajanBabu Chemistry, 730 Autumn 1997
... Mechanism of acid and base catalyzed enolization, kinetic vs thermodynamic control Detailed mechanism of -substitution of a carbonyl compound (e. g., bromination) Carbanions as nucleophiles Enolate structure - X-ray structures of enolates, effect of aggregation, how to control reactivity C vs O-alk ...
... Mechanism of acid and base catalyzed enolization, kinetic vs thermodynamic control Detailed mechanism of -substitution of a carbonyl compound (e. g., bromination) Carbanions as nucleophiles Enolate structure - X-ray structures of enolates, effect of aggregation, how to control reactivity C vs O-alk ...
730-2005 topics
... Mechanism of acid and base catalyzed enolization, kinetic vs thermodynamic control Detailed mechanism of -substitution of a carbonyl compound (e. g., bromination) Carbanions as nucleophiles Enolate structure - X-ray structures of enolates, effect of aggregation, how to control reactivity C vs O-alk ...
... Mechanism of acid and base catalyzed enolization, kinetic vs thermodynamic control Detailed mechanism of -substitution of a carbonyl compound (e. g., bromination) Carbanions as nucleophiles Enolate structure - X-ray structures of enolates, effect of aggregation, how to control reactivity C vs O-alk ...
CH 3 - IBChem.com
... Acids are named according to standard IUPAC rules • select the longest chain of C atoms containing the COOH group; • remove the e and add oic acid after the basic name • number the chain starting from the end nearer the COOH group • as in alkanes, prefix with alkyl substituents • side chain position ...
... Acids are named according to standard IUPAC rules • select the longest chain of C atoms containing the COOH group; • remove the e and add oic acid after the basic name • number the chain starting from the end nearer the COOH group • as in alkanes, prefix with alkyl substituents • side chain position ...
CH 3 OH(l) + CH 3 COCl(l) ——> CH 3 COOCH 3
... Acids are named according to standard IUPAC rules • select the longest chain of C atoms containing the COOH group; • remove the e and add oic acid after the basic name • number the chain starting from the end nearer the COOH group • as in alkanes, prefix with alkyl substituents • side chain position ...
... Acids are named according to standard IUPAC rules • select the longest chain of C atoms containing the COOH group; • remove the e and add oic acid after the basic name • number the chain starting from the end nearer the COOH group • as in alkanes, prefix with alkyl substituents • side chain position ...
Acids and Bases and Aqueous Equilibria
... "When we discuss aqueous solutions of substances which do not contain hydroxyl [ion], it is simplest to define a base as a substance which adds hydrogen ion. . . . Since hydrogen is a constituent of most of our electrolytic solvents, the definition of an acid or base as a substance which gives up or ...
... "When we discuss aqueous solutions of substances which do not contain hydroxyl [ion], it is simplest to define a base as a substance which adds hydrogen ion. . . . Since hydrogen is a constituent of most of our electrolytic solvents, the definition of an acid or base as a substance which gives up or ...
Slide 1
... as steroids, heparin, chondroitin, catecholamine, and thyroxine. Involves the activation of inorganic sulfate to its co-enzyme. ...
... as steroids, heparin, chondroitin, catecholamine, and thyroxine. Involves the activation of inorganic sulfate to its co-enzyme. ...
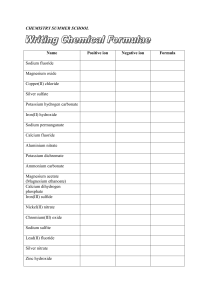
chemistry summer school
... Silver nitrate and copper(II) chloride solutions combine to form solid silver chloride and a solution of copper(II) nitrate. Sulfuric acid reacts with aluminium oxide powder. Glucose (C6H12O6) is fermented to carbon dioxide and ethanol (C2H5OH) Lead(II) nitrate and potassium iodide solutions combine ...
... Silver nitrate and copper(II) chloride solutions combine to form solid silver chloride and a solution of copper(II) nitrate. Sulfuric acid reacts with aluminium oxide powder. Glucose (C6H12O6) is fermented to carbon dioxide and ethanol (C2H5OH) Lead(II) nitrate and potassium iodide solutions combine ...
Carboxylic Acids
... 5. Construct a model of an acetylene lamp that could have been used as a lantern for early automobiles. Explain how your model works to make the light and direct the light down the road. 6. Write the formula equations for each of the following: A) hydrogenation of ethyne, B) hydrolysis of ethyne , C ...
... 5. Construct a model of an acetylene lamp that could have been used as a lantern for early automobiles. Explain how your model works to make the light and direct the light down the road. 6. Write the formula equations for each of the following: A) hydrogenation of ethyne, B) hydrolysis of ethyne , C ...
IE EA
... f) SF6 Neither; the coordination number of six is rarely exceeded so that this molecule does not act as a Lewis acid and the high electronegativity of fluorine does not allow for it to act as a base. g) PCl5 Acidic; this compound reacts with a wide variety of Lewis bases to form adducts. h) (CH3)3N ...
... f) SF6 Neither; the coordination number of six is rarely exceeded so that this molecule does not act as a Lewis acid and the high electronegativity of fluorine does not allow for it to act as a base. g) PCl5 Acidic; this compound reacts with a wide variety of Lewis bases to form adducts. h) (CH3)3N ...
Alcohols
... Give a reagent that can be used in a test to distinguish between a ketone and an aldehyde. State what you would observe in the test. Reagent ............................................................................................................. Observation with ketone ......................... ...
... Give a reagent that can be used in a test to distinguish between a ketone and an aldehyde. State what you would observe in the test. Reagent ............................................................................................................. Observation with ketone ......................... ...
Microsoft Word - Final Exam Study Guide
... B. Would you expect the elimination of cis-1-t-butyl-4-chlorocyclohexane using a strong base to be faster or slower than the elimination of chlorocyclohexane with a strong base? ...
... B. Would you expect the elimination of cis-1-t-butyl-4-chlorocyclohexane using a strong base to be faster or slower than the elimination of chlorocyclohexane with a strong base? ...
Working with Hazardous Chemicals
... 9. Wang, S.-S.; Yang, C. C.; Kulesha, I. D.; Sonenberg, M.; Merrifield, R. B. Int. J. Pept. Protein Res. 1974, 6, 103; Wang, S.-S.; Kulesha, I. D. J. Org. Chem. 1975, 40, 1227; Chang, C.-D.; Meienhofer, J. Int. J. Pept. Protein Res. 1978, 11, 246. 10. Atherton, E.; Benoiton, N. L.; Brown, E.; Sheppa ...
... 9. Wang, S.-S.; Yang, C. C.; Kulesha, I. D.; Sonenberg, M.; Merrifield, R. B. Int. J. Pept. Protein Res. 1974, 6, 103; Wang, S.-S.; Kulesha, I. D. J. Org. Chem. 1975, 40, 1227; Chang, C.-D.; Meienhofer, J. Int. J. Pept. Protein Res. 1978, 11, 246. 10. Atherton, E.; Benoiton, N. L.; Brown, E.; Sheppa ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.