Chapter Three
... Of the ways in which we can define the terms acid and base, two are especially important in organic chemistry According to the Brønsted-Lowry definition, in a chemical reaction an acid donates a proton (H+), and a base accepts a proton ...
... Of the ways in which we can define the terms acid and base, two are especially important in organic chemistry According to the Brønsted-Lowry definition, in a chemical reaction an acid donates a proton (H+), and a base accepts a proton ...
Organic Chemistry Lecture Outline Carbonyl
... substitution reactions. Carbonyl groups bonded to large, bulky substituents (eg., tertiary carbons) react slower with nucleophiles than carbonyl groups with smaller, less bulky substituents. Electronic Effects Electronic effects influence the reactivity of the electrophilic carbonyl carbon in nucleo ...
... substitution reactions. Carbonyl groups bonded to large, bulky substituents (eg., tertiary carbons) react slower with nucleophiles than carbonyl groups with smaller, less bulky substituents. Electronic Effects Electronic effects influence the reactivity of the electrophilic carbonyl carbon in nucleo ...
INTRODUCTION TO LEWIS ACID
... with an incomplete octet, a full positive charge, or a partial positive charge. Water is an example of a Lewis base. Carbocations are examples of Lewis acids. When water reacts with a carbocation as shown below, one of the electron pairs from oxygen is used to form a new sigma bond to the central ca ...
... with an incomplete octet, a full positive charge, or a partial positive charge. Water is an example of a Lewis base. Carbocations are examples of Lewis acids. When water reacts with a carbocation as shown below, one of the electron pairs from oxygen is used to form a new sigma bond to the central ca ...
Specialty Chemicals
... Embracing biodegradability L.V. Lomas continues to offer innovative, environmentally safe and beneficial products to our customer base. We view it as an investment in our collective future. It’s also what increasingly well-informed consumers are demanding. ...
... Embracing biodegradability L.V. Lomas continues to offer innovative, environmentally safe and beneficial products to our customer base. We view it as an investment in our collective future. It’s also what increasingly well-informed consumers are demanding. ...
Chapter 20 Carboxylic Acids
... • An activated form of the carboxylic acid. • Chloride is a good leaving group, so undergoes acyl substitution easily. • To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
... • An activated form of the carboxylic acid. • Chloride is a good leaving group, so undergoes acyl substitution easily. • To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
Chapter 20 Carboxylic Acids
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
... An activated form of the carboxylic acid. Chloride is a good leaving group, so undergoes acyl substitution easily. To synthesize acid chlorides use thionyl chloride or oxalyl chloride with the acid. O ...
Chemistry Scavenger Hunt
... ____ Most common atom in universe ____ Atomic number of 9 ____ Found in buckyballs ____ Used in laundry detergents ____ Atomic number of 2 ...
... ____ Most common atom in universe ____ Atomic number of 9 ____ Found in buckyballs ____ Used in laundry detergents ____ Atomic number of 2 ...
+ :O
... Carboxylic acids are weaker acids than mineral acids like HCl, HNO3, or H2SO4, but they are more acidic than organic weak acids such as aliphatic alcohols. Carboxylic acids are converted into their carboxylate salts by aqueous solutions of hydroxide. -/H O ...
... Carboxylic acids are weaker acids than mineral acids like HCl, HNO3, or H2SO4, but they are more acidic than organic weak acids such as aliphatic alcohols. Carboxylic acids are converted into their carboxylate salts by aqueous solutions of hydroxide. -/H O ...
Synthesis of Benzene Derivatives: Electrophilic Aromatic Substitution
... Fuming sulfuric acid (8% SO3 in concentrated H2SO4) reacts with benzene to form benzenesulfonic acid. ...
... Fuming sulfuric acid (8% SO3 in concentrated H2SO4) reacts with benzene to form benzenesulfonic acid. ...
Chemistry Review for End of year final honors
... 2.) A piece of metal is heated, then submerged in cool water. What will happen in the transfer of heat? (Where does the heat come from, where does it go to?) 3.) If the heat involved in a chemical reaction (q or ΔH) has a negative value, what does this ...
... 2.) A piece of metal is heated, then submerged in cool water. What will happen in the transfer of heat? (Where does the heat come from, where does it go to?) 3.) If the heat involved in a chemical reaction (q or ΔH) has a negative value, what does this ...
ORGSEQPP.pps
... The second ammonia molecule ensures the removal of HBr which would lead to the formation of a salt. A large excess ammonia ensures that further substitution doesn’t take place - see below Problem The amine produced is also a nucleophile (lone pair on N) and can attack another molecule of halogenoalk ...
... The second ammonia molecule ensures the removal of HBr which would lead to the formation of a salt. A large excess ammonia ensures that further substitution doesn’t take place - see below Problem The amine produced is also a nucleophile (lone pair on N) and can attack another molecule of halogenoalk ...
C 2 H 5 OH(l)
... The second ammonia molecule ensures the removal of HBr which would lead to the formation of a salt. A large excess ammonia ensures that further substitution doesn’t take place - see below Problem The amine produced is also a nucleophile (lone pair on N) and can attack another molecule of halogenoalk ...
... The second ammonia molecule ensures the removal of HBr which would lead to the formation of a salt. A large excess ammonia ensures that further substitution doesn’t take place - see below Problem The amine produced is also a nucleophile (lone pair on N) and can attack another molecule of halogenoalk ...
Chapter 1 Structure and Bonding
... Nucleophile approaches, causing C to rehybridize p-bond electrons move to Oxygen, producing an alkoxide anion Protonation from solvent yields the product The new Nu—C bond has both electrons from Nu- (like in SN2) An electron pair is the “leaving group” Strongly basic nucleophiles typically follow t ...
... Nucleophile approaches, causing C to rehybridize p-bond electrons move to Oxygen, producing an alkoxide anion Protonation from solvent yields the product The new Nu—C bond has both electrons from Nu- (like in SN2) An electron pair is the “leaving group” Strongly basic nucleophiles typically follow t ...
OXYGEN CONTAINING ORGANIC COMPOUNDS
... stronger acids than water and will dissolve in 5% NaOH weaker acids than carbonic acid ...
... stronger acids than water and will dissolve in 5% NaOH weaker acids than carbonic acid ...
ALDEHYDES AND KETONES
... If the nucleophile that adds to the aldehyde or ketone is an O or an N, a nucleophilic addition–elimination reaction ...
... If the nucleophile that adds to the aldehyde or ketone is an O or an N, a nucleophilic addition–elimination reaction ...
Chapter 1-
... Acylations: The Clemmensen Reduction Primary alkyl halides often yield rearranged products in FriedelCrafts alkylation which is a major limitation of this reaction Unbranched alkylbenzenes can be obtained in good yield by acylation followed by Clemmensen reduction ...
... Acylations: The Clemmensen Reduction Primary alkyl halides often yield rearranged products in FriedelCrafts alkylation which is a major limitation of this reaction Unbranched alkylbenzenes can be obtained in good yield by acylation followed by Clemmensen reduction ...
Slide 1 - Alfred State College intranet site
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
Chapter 20 Carboxylic Acids
... Carboxylic acids are converted to their methyl esters very simply by adding an ether solution of diazomethane. The reaction usually produces quantitative yields of ester. Diazomethane is very toxic, explosive. Dissolve in ether. Chapter 20 ...
... Carboxylic acids are converted to their methyl esters very simply by adding an ether solution of diazomethane. The reaction usually produces quantitative yields of ester. Diazomethane is very toxic, explosive. Dissolve in ether. Chapter 20 ...
PowerPoint **
... Draw mechanisms for the following reaction and explain why carbonyl acid can’t undergo similar reaction ...
... Draw mechanisms for the following reaction and explain why carbonyl acid can’t undergo similar reaction ...
Notes 07 Organometallic Compounds with notes
... formaldehyde (H2C=O) followed by H3O+? A) 2 B) 3 C) 4 D) 5 ...
... formaldehyde (H2C=O) followed by H3O+? A) 2 B) 3 C) 4 D) 5 ...
CHM412 June 2013 paper
... alcohold that can be oxided to methyl carbonyls will also give a positive test result for this reaction, because the reagents used in the triiodomethane test are oxidising, i.e. I 2(aq) + NaOH. They oxidise methyl 1o alcohols and methyl 2o alcohols to methyl carbonyls, which then undergo the usual b ...
... alcohold that can be oxided to methyl carbonyls will also give a positive test result for this reaction, because the reagents used in the triiodomethane test are oxidising, i.e. I 2(aq) + NaOH. They oxidise methyl 1o alcohols and methyl 2o alcohols to methyl carbonyls, which then undergo the usual b ...
ALKENES and SULPHURIC ACID
... In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count. The ones in the CH3 group are totally irrelevant. Warning! Markovniko ...
... In this case, the hydrogen becomes attached to the CH2 group, because the CH2 group has more hydrogens than the CH group. Notice that only the hydrogens directly attached to the carbon atoms at either end of the double bond count. The ones in the CH3 group are totally irrelevant. Warning! Markovniko ...
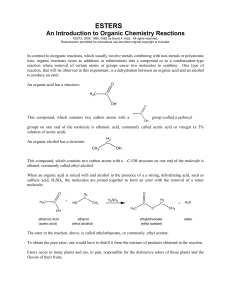
Esters - chymist.com
... In contrast to inorganic reactions, which usually involve metals combining with non-metals or polyatomic ions, organic reactions occur as additions or substitutions into a compound or as a condensation-type reaction where removal of certain atoms or groups cause two molecules to combine. One type of ...
... In contrast to inorganic reactions, which usually involve metals combining with non-metals or polyatomic ions, organic reactions occur as additions or substitutions into a compound or as a condensation-type reaction where removal of certain atoms or groups cause two molecules to combine. One type of ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.