16.7 Addition of Alcohols: Hemiacetals and Acetals
... • The carbonyl group is a C=O. The group is polar, with a partial (-) charge on O and a partial (+) charge on C. The O and the two substituents on the carbonyl-group C atom form a planar triangle. • The simplest aldehydes and ketones are known by common names. Aldehydes are named systematically by r ...
... • The carbonyl group is a C=O. The group is polar, with a partial (-) charge on O and a partial (+) charge on C. The O and the two substituents on the carbonyl-group C atom form a planar triangle. • The simplest aldehydes and ketones are known by common names. Aldehydes are named systematically by r ...
Document
... a. HCO3- reacts with water to form H2PO4, which has a pKa close to the pH of blood. b. H2CO3 in the blood dissociates to form HCO3-. c. H2CO3 in the blood dissociates to form HCO3- and dissolved CO2 in the blood forms H2CO3. d. Dissolved CO2 in the blood forms H2CO3. e. H2CO3 in the blood dissociate ...
... a. HCO3- reacts with water to form H2PO4, which has a pKa close to the pH of blood. b. H2CO3 in the blood dissociates to form HCO3-. c. H2CO3 in the blood dissociates to form HCO3- and dissolved CO2 in the blood forms H2CO3. d. Dissolved CO2 in the blood forms H2CO3. e. H2CO3 in the blood dissociate ...
Biodiesel Session 2
... Reactions of esters • Other side reactions may be: Reaction with bases in water or water to form free fatty acids and acylates ...
... Reactions of esters • Other side reactions may be: Reaction with bases in water or water to form free fatty acids and acylates ...
Chapter 8. CARBONYL COMPOUNDS
... The aldol can be easily dehydrated on heating because it retains the acidic α-hydrogen. α,βUnsaturated aldehydes and ketones are formed when the reaction is performed at elevated temperature. Crotonaldehyde is obtained from the simplest aldol in this way, therefore reactions of this type are called ...
... The aldol can be easily dehydrated on heating because it retains the acidic α-hydrogen. α,βUnsaturated aldehydes and ketones are formed when the reaction is performed at elevated temperature. Crotonaldehyde is obtained from the simplest aldol in this way, therefore reactions of this type are called ...
Functional Derivatives of Carboxylic Acids
... • Hydrolysis in aqueous acid is the reverse of Fischer esterification – the role of the acid catalyst is to protonate the carbonyl oxygen and increase its electrophilic character toward attack by water (a weak nucleophile) to form a tetrahedral carbonyl addition intermediate – collapse of this inter ...
... • Hydrolysis in aqueous acid is the reverse of Fischer esterification – the role of the acid catalyst is to protonate the carbonyl oxygen and increase its electrophilic character toward attack by water (a weak nucleophile) to form a tetrahedral carbonyl addition intermediate – collapse of this inter ...
Chemistry of Life
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
... • Oxygen and Hydrogen are bonded together by ________ electrons, but the Oxygen atom _________the shared electrons closer to it, creating negative and positive sides of the water molecules. Water has a partial negative charge due to the extra unshared e- that Oxygen and a partial + charge near the h ...
Unit 12 Worksheet Answers
... 3. What is the difference between a nonelectrolyte, a strong electrolyte and a weak electrolyte? Nonelectrolyte does not conduct electricity (no ions) Weak electrolyte conducts electricity a little (little ions) Strong electrolyte conducts electricity well (a lot of ions) 4. What is the difference b ...
... 3. What is the difference between a nonelectrolyte, a strong electrolyte and a weak electrolyte? Nonelectrolyte does not conduct electricity (no ions) Weak electrolyte conducts electricity a little (little ions) Strong electrolyte conducts electricity well (a lot of ions) 4. What is the difference b ...
Topic 8.4 Acids and Bases The pH Scale
... developed by the head of Carlsberg Brewery Laboratory’s Chemical Department in 1909. Dr Søren Sørensen (1868-1939) developed the pH scale during his pioneering research into proteins, amino acids and enzymes the basis of today’s protein chemistry. Basically meaning ‘the power of hydrogen’, the sca ...
... developed by the head of Carlsberg Brewery Laboratory’s Chemical Department in 1909. Dr Søren Sørensen (1868-1939) developed the pH scale during his pioneering research into proteins, amino acids and enzymes the basis of today’s protein chemistry. Basically meaning ‘the power of hydrogen’, the sca ...
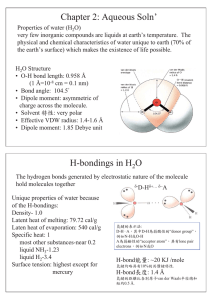
Chapter 2: Aqueous Soln` H
... Representation of the transport of CO2 from the tissues to the blood with delivery of O2 to the tissues. The opposite process occurs when O2 is taken up from the alveoli of the lungs and the CO2 is expelled. All of the processes of the transport of CO2 and O2 are not shown such as the formation and ...
... Representation of the transport of CO2 from the tissues to the blood with delivery of O2 to the tissues. The opposite process occurs when O2 is taken up from the alveoli of the lungs and the CO2 is expelled. All of the processes of the transport of CO2 and O2 are not shown such as the formation and ...
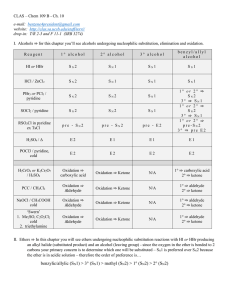
Chapter 10 - UCSB CLAS
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
... as epoxides in basic/neutral conditions or rearrangement reactions to produce phenol (primary focus) – since there’s a carbocation formed in the rearrangement reaction this will determine which carbon gets the OH – any additional substituents that can stabilize a carbocation (electron donating) will ...
Document
... • Methanol (CH3OH) • Often Called Wood Alcohol (Distilled From Wood) • Prepared Now via Catalytic Hydrogenation Reactions • Ethanol (CH3CH2OH) • Made Through Fermentation of Sugars, in Alcoholic Drinks • Common Solvent in Organic Labs (Absolute Ethanol) • Ethylene Glycol (HOCH2CH2OH) ...
... • Methanol (CH3OH) • Often Called Wood Alcohol (Distilled From Wood) • Prepared Now via Catalytic Hydrogenation Reactions • Ethanol (CH3CH2OH) • Made Through Fermentation of Sugars, in Alcoholic Drinks • Common Solvent in Organic Labs (Absolute Ethanol) • Ethylene Glycol (HOCH2CH2OH) ...
Alcohols I Reading: Wade chapter 10, sections 10-1- 10
... Organometallic compounds contain a highly polarized covalent bond between carbon and a metal atom (C–M, M=Li, Na, K, Mg). The C–M bond is polarized so that most of the electron density resides on carbon, since it is the more electronegative atom of the pair. This makes carbon a good nucleophile (and ...
... Organometallic compounds contain a highly polarized covalent bond between carbon and a metal atom (C–M, M=Li, Na, K, Mg). The C–M bond is polarized so that most of the electron density resides on carbon, since it is the more electronegative atom of the pair. This makes carbon a good nucleophile (and ...
CHAPTER II. A Facile Synthesis of Arylacetic Acid Derivatives via
... “Design and Synthesis of Some Biologically Active Aryl Alkanoic Acid Derivatives and Development of Novel Synthetic Methodologies” The thesis has been presented as five chapters. Chapter-I deals with general introduction on the synthesis of arylacetic acids, -hydroxy arylacetic acid (mandelic acid) ...
... “Design and Synthesis of Some Biologically Active Aryl Alkanoic Acid Derivatives and Development of Novel Synthetic Methodologies” The thesis has been presented as five chapters. Chapter-I deals with general introduction on the synthesis of arylacetic acids, -hydroxy arylacetic acid (mandelic acid) ...
Chapter 4 - Aqueous Reactions
... A metal can be oxidized by any ion below it Metals above H, react with acids to give H2 The further up the series, the more readily the metal is oxidized See your textbook (p 136) for more elements ...
... A metal can be oxidized by any ion below it Metals above H, react with acids to give H2 The further up the series, the more readily the metal is oxidized See your textbook (p 136) for more elements ...
haloalkanes 2013
... (i) C2H5Br + NH3 (aq / alc) ——> C2H5NH2 + HBr (ii) HBr + NH3 (aq / alc) ——> NH4Br ...
... (i) C2H5Br + NH3 (aq / alc) ——> C2H5NH2 + HBr (ii) HBr + NH3 (aq / alc) ——> NH4Br ...
CHM230 OXIDATION OF CYCLOHEXANOL TO CYCLOHEXANONE
... graduated cylinder. The distillate should be a mixture of cyclohexanone and water that contains excess acetic acid. RECORD the boiling point of the cyclohexanone. Transfer the distillate to a separatory funnel or beaker. 6. Add 3.5 grams of sodium carbonate to neutralize any excess acetic acid, and ...
... graduated cylinder. The distillate should be a mixture of cyclohexanone and water that contains excess acetic acid. RECORD the boiling point of the cyclohexanone. Transfer the distillate to a separatory funnel or beaker. 6. Add 3.5 grams of sodium carbonate to neutralize any excess acetic acid, and ...
Chapter one
... * 15-4 Explore - Colder then ice water - in this activity you have discovered what happens to the freezing point of water when a substance is dissolved ...
... * 15-4 Explore - Colder then ice water - in this activity you have discovered what happens to the freezing point of water when a substance is dissolved ...
GRADE 12A: Chemistry 6
... • Ask students to draw the structure of the named compound and the name of the structure onto a small whiteboard and hold it up for you to see. ...
... • Ask students to draw the structure of the named compound and the name of the structure onto a small whiteboard and hold it up for you to see. ...
Williamson Ether Synthesis
... Ethers are prepared by SN2 reactions. Ethers can be prepared by the reaction of an alkoxide with a primary haloalkane or sulfonate ester under SN2 conditions. The parent alcohol of the alkoxide can be used as the solvent, however other polar solvents are often better, such as DMSO (dimethyl sulfoxid ...
... Ethers are prepared by SN2 reactions. Ethers can be prepared by the reaction of an alkoxide with a primary haloalkane or sulfonate ester under SN2 conditions. The parent alcohol of the alkoxide can be used as the solvent, however other polar solvents are often better, such as DMSO (dimethyl sulfoxid ...
2009-10 Chemistry 1st Semester Final Exam Topics and Review
... 35. Write and balance the chemical equations for the following reactions. Include the physical states (aq, s, l, g) of the reactants and products. a. When zinc metal and sulfur powder are heated, they form solid zinc sulfide. b. When sodium metal is placed in a beaker of water, hydrogen gas and sodi ...
... 35. Write and balance the chemical equations for the following reactions. Include the physical states (aq, s, l, g) of the reactants and products. a. When zinc metal and sulfur powder are heated, they form solid zinc sulfide. b. When sodium metal is placed in a beaker of water, hydrogen gas and sodi ...
Exam 2 Fall 2005 Chemsitry 1211
... This exam is twenty five questions long. Each question is worth 4 points. Please read through all of the questions first and ask about anything you do not understand. You will have one hour and 15 minutes to complete this exam. Exams will be picked up at the end of the class period. No late exams wi ...
... This exam is twenty five questions long. Each question is worth 4 points. Please read through all of the questions first and ask about anything you do not understand. You will have one hour and 15 minutes to complete this exam. Exams will be picked up at the end of the class period. No late exams wi ...
unit 17 organic compounds containing oxygen and nitrogen atoms
... Aliphatic aldehydes form a homologous series with the general formula RCHO and ketons with general formula RCOR'. Aldehydes and ketones have the same general formula CnH,"O. Since carbonyl group is present in both aldehydes and ketones, many of their properties are common. But in aldehyde, there is ...
... Aliphatic aldehydes form a homologous series with the general formula RCHO and ketons with general formula RCOR'. Aldehydes and ketones have the same general formula CnH,"O. Since carbonyl group is present in both aldehydes and ketones, many of their properties are common. But in aldehyde, there is ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.