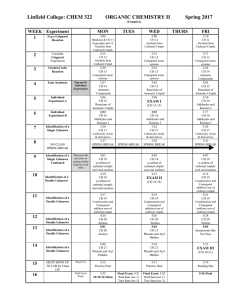
Lab 6
... 2- Fenton's Reagent: (Oxidation of tartaric acid in the presence of iron) To a solution of tartaric acid or a tartarate add 1 drop of freshly prepared ferrous sulphate solution, 1 drop of hydrogen peroxide solution and excess of NaOH solution. An intense violet colour is produced due to the ferrous ...
... 2- Fenton's Reagent: (Oxidation of tartaric acid in the presence of iron) To a solution of tartaric acid or a tartarate add 1 drop of freshly prepared ferrous sulphate solution, 1 drop of hydrogen peroxide solution and excess of NaOH solution. An intense violet colour is produced due to the ferrous ...
... Class Environment: Talking is completely prohibited except you ask any questions regarding the class materials or you are asked any questions by the Instructor. Private talking in the class will hurt your understanding and also will hurt others. No sleeping in the class room allowed Objectives of th ...
Functional Group Chemistry
... Tertiary alcohols (in which three carbon atoms are attached to same carbon as the –OH group) react very quickly in the Lucas test, producing alkyl halides that are insoluble in the aqueous Lucas reagent. Secondary alcohols (in which two carbon atoms are attached to same carbon as the –OH group) reac ...
... Tertiary alcohols (in which three carbon atoms are attached to same carbon as the –OH group) react very quickly in the Lucas test, producing alkyl halides that are insoluble in the aqueous Lucas reagent. Secondary alcohols (in which two carbon atoms are attached to same carbon as the –OH group) reac ...
Part 1
... Functional groups: special groups of atoms attached to a hydrocarbon skeleton; the most common sites of chemical reactivity Organic halides: a hydrogen is replaced by a halogen (fluoro-, chloro-, bromo-, iodo-) ...
... Functional groups: special groups of atoms attached to a hydrocarbon skeleton; the most common sites of chemical reactivity Organic halides: a hydrogen is replaced by a halogen (fluoro-, chloro-, bromo-, iodo-) ...
SN1 vs. SN2 Reactions - Master Organic Chemistry
... Look for halogens (Cl, Br, I) or tosylates/mesylates (OTs, OMs) Alternatively, look for alcohols (OH) if acid is present Once you've identified the leaving group, instpect the carbon it is attached to. How many carbons is that carbon connected to? That will tell you if the carbon is primary, seconda ...
... Look for halogens (Cl, Br, I) or tosylates/mesylates (OTs, OMs) Alternatively, look for alcohols (OH) if acid is present Once you've identified the leaving group, instpect the carbon it is attached to. How many carbons is that carbon connected to? That will tell you if the carbon is primary, seconda ...
Chapter 19. Aldehydes and Ketones
... Electron-donating resonance effect of aromatic ring makes C=O less reactive electrophile than the carbonyl group of an aliphatic aldehyde ...
... Electron-donating resonance effect of aromatic ring makes C=O less reactive electrophile than the carbonyl group of an aliphatic aldehyde ...
S3 Chemistry - eduBuzz.org
... The pH scale; which colours and numbers represent an acid, an alkali and neutral substances. The names and formula of common lab acids. Examples of acids and alkalis you would find in everyday ...
... The pH scale; which colours and numbers represent an acid, an alkali and neutral substances. The names and formula of common lab acids. Examples of acids and alkalis you would find in everyday ...
Chapter 2 – Chemical Composition of the Body
... • Molecules that are formed by polar covalent bonds have a tendency to break apart when the electron from the hydrogen is transferred to the more electronegative atom. This is called dissociation or ionization. • Water ionizes to form equal amounts of hydroxyl (OH-) and hydrogen (hydronium, H+) io ...
... • Molecules that are formed by polar covalent bonds have a tendency to break apart when the electron from the hydrogen is transferred to the more electronegative atom. This is called dissociation or ionization. • Water ionizes to form equal amounts of hydroxyl (OH-) and hydrogen (hydronium, H+) io ...
Honors Chemistry
... 5. What is an isotope? 6. How can the number of protons be determined? Neutrons? Electrons? 7. Determine the # of p+, no and e- in the following: oxygen-14, 356210XY34- and Astatine. 8. Draw a wave. Label the wavelength and amplitude on your drawing. 9. What are the colors that make up white light? ...
... 5. What is an isotope? 6. How can the number of protons be determined? Neutrons? Electrons? 7. Determine the # of p+, no and e- in the following: oxygen-14, 356210XY34- and Astatine. 8. Draw a wave. Label the wavelength and amplitude on your drawing. 9. What are the colors that make up white light? ...
Alcohols - Structure - University of Nebraska Omaha
... • When isomeric alkenes are obtained, the more stable alkene (the one with the greater number of substituents on the double bond) generally ...
... • When isomeric alkenes are obtained, the more stable alkene (the one with the greater number of substituents on the double bond) generally ...
Chemistry Worksheet on Mass Mol and Mol Mass
... Chemistry Worksheet on MassMol and MolMass Calculations Assume all reactions go to completion. Write the formula equation, balance the equations, and solve the problems. Show all work. Draw a rectangle around the answer and don’t forget the units. One in a series of reactions that inflate air bags ...
... Chemistry Worksheet on MassMol and MolMass Calculations Assume all reactions go to completion. Write the formula equation, balance the equations, and solve the problems. Show all work. Draw a rectangle around the answer and don’t forget the units. One in a series of reactions that inflate air bags ...
Chapter 12
... A Chemical reaction involves only the separation, combination, or rearrangement of atoms. It does not result in their creation or destruction. Law of Definite Proportions: Different samples of the same compounds always contain the same elements in the same proportions by mass Example: In water (H2 ...
... A Chemical reaction involves only the separation, combination, or rearrangement of atoms. It does not result in their creation or destruction. Law of Definite Proportions: Different samples of the same compounds always contain the same elements in the same proportions by mass Example: In water (H2 ...
This is an author version of the contribution published on: Questa è
... water and/or use of excess amounts of the reactants if satisfactory conversion rates are to be obtained.1 The major drawbacks of these common methods are the final neutralization of the homogeneous acid catalyst and/or the separation of a metal catalyst on a solid support. Moreover, the removal of a ...
... water and/or use of excess amounts of the reactants if satisfactory conversion rates are to be obtained.1 The major drawbacks of these common methods are the final neutralization of the homogeneous acid catalyst and/or the separation of a metal catalyst on a solid support. Moreover, the removal of a ...
2.10 Alcohols notes - A
... polymers to be produced without using crude oil (assuming that the original ethanol was produced by fermentation). The dehydration of alcohols is favoured by acidic conditions, as the -OH group becomes protonated by H+ ions which produces a water molecule which then leaves. The acid acts as a cataly ...
... polymers to be produced without using crude oil (assuming that the original ethanol was produced by fermentation). The dehydration of alcohols is favoured by acidic conditions, as the -OH group becomes protonated by H+ ions which produces a water molecule which then leaves. The acid acts as a cataly ...
Mill Hill County High School
... polymers to be produced without using crude oil (assuming that the original ethanol was produced by fermentation). The dehydration of alcohols is favoured by acidic conditions, as the -OH group becomes protonated by H+ ions which produces a water molecule which then leaves. The acid acts as a cataly ...
... polymers to be produced without using crude oil (assuming that the original ethanol was produced by fermentation). The dehydration of alcohols is favoured by acidic conditions, as the -OH group becomes protonated by H+ ions which produces a water molecule which then leaves. The acid acts as a cataly ...
Carboxylic Acid Structure and Chemistry
... Principles of Drug Action 1, Spring 2005, Carboxylic Acids Part 1 It is important to note that the water solubility of carboxylic acids and other organic compounds with dipolar functionality may be a function of more than dipolar interactions alone. For example, carboxylic acids and other polar org ...
... Principles of Drug Action 1, Spring 2005, Carboxylic Acids Part 1 It is important to note that the water solubility of carboxylic acids and other organic compounds with dipolar functionality may be a function of more than dipolar interactions alone. For example, carboxylic acids and other polar org ...
No Slide Title - McMaster Chemistry
... Weak ACIDS/ weak BASES only react partially with water - an EQUILIBRIUM is formed : the conjugate ACID and the conjugate BASE are both present at the same time WEAK ACID: (acetic acid a.k.a. vinegar) CH3CO2H + H2O CH3CO2- (aq) + H3O+ (aq) WEAK BASE: NH3 (g) + H2O NH4+ (aq) + OH- (aq) 1A03/1E03 T ...
... Weak ACIDS/ weak BASES only react partially with water - an EQUILIBRIUM is formed : the conjugate ACID and the conjugate BASE are both present at the same time WEAK ACID: (acetic acid a.k.a. vinegar) CH3CO2H + H2O CH3CO2- (aq) + H3O+ (aq) WEAK BASE: NH3 (g) + H2O NH4+ (aq) + OH- (aq) 1A03/1E03 T ...
Project Overview
... Since Grignard reagents are powerful nucleophiles, we cannot prepare a Grignard reagent from any organic halide that contains a carbonyl, epoxy, nitro, or cyano (–CN) group Ch. 12 - 62 ...
... Since Grignard reagents are powerful nucleophiles, we cannot prepare a Grignard reagent from any organic halide that contains a carbonyl, epoxy, nitro, or cyano (–CN) group Ch. 12 - 62 ...
Lecture syllabus - Linfield College
... organic compounds are studied. During second semester (CHEM 322), aromatics, organometallics, alcohols, ethers, aldehydes, ketones, carboxylic acids, acid derivatives, and amines are the classes stressed. The course is designed to provide a fundamental knowledge of organic chemistry - the study of c ...
... organic compounds are studied. During second semester (CHEM 322), aromatics, organometallics, alcohols, ethers, aldehydes, ketones, carboxylic acids, acid derivatives, and amines are the classes stressed. The course is designed to provide a fundamental knowledge of organic chemistry - the study of c ...
Alkene - Synthesis
... Acetone can dissolve both the iodide and the alkyl halide (if small). If Zn is used, reaction is heterogeneous and takes place on the surface of the Zn. Reduction because “Br2” is removed. ...
... Acetone can dissolve both the iodide and the alkyl halide (if small). If Zn is used, reaction is heterogeneous and takes place on the surface of the Zn. Reduction because “Br2” is removed. ...
Sodium hydroxide
... through solutions containing copper compounds, such as copper(II) sulphate. Pure copper forms on the negative electrode. The animation shows how this works: ...
... through solutions containing copper compounds, such as copper(II) sulphate. Pure copper forms on the negative electrode. The animation shows how this works: ...
A Few More Notes on Acidity
... Ka is the equilibrium constant for the dissociation of an acid into H+ and its conjugate base. Where possible, Ka values are measured using water as the solvent. ...
... Ka is the equilibrium constant for the dissociation of an acid into H+ and its conjugate base. Where possible, Ka values are measured using water as the solvent. ...
Nucleophilic acyl substitution

Nucleophilic acyl substitution describe a class of substitution reactions involving nucleophiles and acyl compounds. In this type of reaction, a nucleophile – such as an alcohol, amine, or enolate – displaces the leaving group of an acyl derivative – such as an acid halide, anhydride, or ester. The resulting product is a carbonyl-containing compound in which the nucleophile has taken the place of the leaving group present in the original acyl derivative. Because acyl derivatives react with a wide variety of nucleophiles, and because the product can depend on the particular type of acyl derivative and nucleophile involved, nucleophilic acyl substitution reactions can be used to synthesize a variety of different products.