Preparatory Problems of the 40th IChO - IChO-2016
... The first inorganic compound containing a nitrogen-only species different from the above was synthesized by Christe and co-workers in 1999. The starting material of the synthesis is an unstable liquid A that is a weak, monoprotic acid. It was liberated from its sodium salt (that contains 35.36 % sod ...
... The first inorganic compound containing a nitrogen-only species different from the above was synthesized by Christe and co-workers in 1999. The starting material of the synthesis is an unstable liquid A that is a weak, monoprotic acid. It was liberated from its sodium salt (that contains 35.36 % sod ...
HSC Chemistry Syllabus Notes 2007
... memorise the content. Secondly, you need to understand the concepts. Questions do not always ask you to recall a memorised slab of information, they sometimes require you to think critically and solve problems. To be able to do this you need to have a clear and deep understanding of the chemistry. T ...
... memorise the content. Secondly, you need to understand the concepts. Questions do not always ask you to recall a memorised slab of information, they sometimes require you to think critically and solve problems. To be able to do this you need to have a clear and deep understanding of the chemistry. T ...
Problem 1-2
... of air a ternary, white, crystalline compound B and a gas C form. In the presence of air gas C burns with a light blue flame. The elementary analysis of B shows 24.5 % (w/w) of carbon and 28.6 % (w/w) of nitrogen. When annealed with carbon another ionic compound D also results in compound B, too, bu ...
... of air a ternary, white, crystalline compound B and a gas C form. In the presence of air gas C burns with a light blue flame. The elementary analysis of B shows 24.5 % (w/w) of carbon and 28.6 % (w/w) of nitrogen. When annealed with carbon another ionic compound D also results in compound B, too, bu ...
Introduction to Inorganic Chemistry
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
... Chemistry comprises two related but distinct activities: (i) the quest for an understanding of matter and material change, (ii) the utilization of material change for human ends. Ideally, the first activity provides the necessary know-how for the pursuit of the second, but in practice, the help it c ...
KCET – CHEMISTRY – 2016 - Medicine.careers360.com
... The correct statement regarding entropy is, 1) At absolute zero temperature, entropy of a perfectly crystalline solid is zero 2) At absolute zero temperature, the entropy of a perfectly crystalline substance is +Ve 3) At absolute zero temperature, the entropy of all crystalline substance is zero 4) ...
... The correct statement regarding entropy is, 1) At absolute zero temperature, entropy of a perfectly crystalline solid is zero 2) At absolute zero temperature, the entropy of a perfectly crystalline substance is +Ve 3) At absolute zero temperature, the entropy of all crystalline substance is zero 4) ...
Organic Chemistry with a Biological Emphasis Volume I
... the brain - 'it's hot, stay away!' This strategy works quite well on all mammalian species except one: we humans (some of us, at least) appear to be alone in our tendency to actually seek out the burn of the hot pepper in our food. Interestingly, birds also have a heat receptor protein which is very ...
... the brain - 'it's hot, stay away!' This strategy works quite well on all mammalian species except one: we humans (some of us, at least) appear to be alone in our tendency to actually seek out the burn of the hot pepper in our food. Interestingly, birds also have a heat receptor protein which is very ...
4 Types of Chemical Reactions and Solution Stoichiometry
... Because of this unequal charge distribution, water is said to be a polar molecule. It is this polarity that gives water its great ability to dissolve compounds. A schematic of an ionic solid dissolving in water is shown in Fig. 4.2. Note that the “positive ends” of the water molecules are attracted ...
... Because of this unequal charge distribution, water is said to be a polar molecule. It is this polarity that gives water its great ability to dissolve compounds. A schematic of an ionic solid dissolving in water is shown in Fig. 4.2. Note that the “positive ends” of the water molecules are attracted ...
PREPARATION, STRUCTURAL STUDIES AND CHEMICAL
... There are many useful oxidizing agents for the first step: chlorine gas and xenon(II) fluoride XeF2 lead to hypervalent iodine(III) dichloride and difluoride, respectively. Other examples of oxidizing agents include peroxides (hydrogen peroxide, peracetic acid, MCPBA, Oxone®, etc.). To obtain hetero ...
... There are many useful oxidizing agents for the first step: chlorine gas and xenon(II) fluoride XeF2 lead to hypervalent iodine(III) dichloride and difluoride, respectively. Other examples of oxidizing agents include peroxides (hydrogen peroxide, peracetic acid, MCPBA, Oxone®, etc.). To obtain hetero ...
Question Bank for Pre Board Exam(XII Chemistry)
... 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Why does conductivity of silicon increase with the rise in temperature? 41.Name the crystal defect which lowers the density of an ionic crystal. 42 What makes the crystal of KCl somet ...
... 39.Which point defect is observed in a crystal when a vacancy is created by an atom missing from a lattice site. 40. Why does conductivity of silicon increase with the rise in temperature? 41.Name the crystal defect which lowers the density of an ionic crystal. 42 What makes the crystal of KCl somet ...
chem textbook 2015 - Manitowoc Public School District
... The intent of the following information is to give answers and suggestions to questions that students often ask, it is meant to work in conjunction with Suggestions for Boosting Grades. “It makes sense in class but not when I get home.” This generally means that your notes are incomplete, meaning th ...
... The intent of the following information is to give answers and suggestions to questions that students often ask, it is meant to work in conjunction with Suggestions for Boosting Grades. “It makes sense in class but not when I get home.” This generally means that your notes are incomplete, meaning th ...
chemistry - The Aga Khan University
... Group 7-Elements:Halogens 13.5.1 Atomic and Physical Properties 13.5.1.1 Trends in Atomic Radius 13.5.1.2 Trends in Electronegativity 13.5.1.3 Trends in Electron Affinity 13.5.1.4 Trends in Melting and Boiling Points 13.5.1.5 Bond Enthalpies 13.5.1.5.1 Bond Enthalpies in Halogens 13.5.1.5.2 Bond Ent ...
... Group 7-Elements:Halogens 13.5.1 Atomic and Physical Properties 13.5.1.1 Trends in Atomic Radius 13.5.1.2 Trends in Electronegativity 13.5.1.3 Trends in Electron Affinity 13.5.1.4 Trends in Melting and Boiling Points 13.5.1.5 Bond Enthalpies 13.5.1.5.1 Bond Enthalpies in Halogens 13.5.1.5.2 Bond Ent ...
Peter Ertl - American Chemical Society
... An analysis of the substituents described hitherto produced a large database of about 850 000 substituents. To use this database in practical applications, the substituents need to be classified in some way. For applications in drug design, a classification based on substituent physicochemical prope ...
... An analysis of the substituents described hitherto produced a large database of about 850 000 substituents. To use this database in practical applications, the substituents need to be classified in some way. For applications in drug design, a classification based on substituent physicochemical prope ...
Unit 6 Chemical Energy
... Everything humans do requires energy. It is a major factor in social change on our planet. Technologies, which inevitably consume energy, are developed for a social purpose. These technologies, however, often have drawbacks related to their use of energy. The control and use of our present sources o ...
... Everything humans do requires energy. It is a major factor in social change on our planet. Technologies, which inevitably consume energy, are developed for a social purpose. These technologies, however, often have drawbacks related to their use of energy. The control and use of our present sources o ...
OCR A Level Chemistry A H432 Specification
... Our new A Level in Chemistry A builds on our existing popular course. We’ve based the redevelopment of our A level sciences on an understanding of what works well in centres large and small and have updated areas of content and assessment where stakeholders have identified that improvements could be ...
... Our new A Level in Chemistry A builds on our existing popular course. We’ve based the redevelopment of our A level sciences on an understanding of what works well in centres large and small and have updated areas of content and assessment where stakeholders have identified that improvements could be ...
Study materials of Chemistry for class XII
... Number of atoms in 200g of the element =3.47×1024 . Q12. Pure silicon is an insulator. Silicon doped with phosphorus is a semiconductor. Silicon doped with gallium is also a semiconductor what is the difference between the two semiconductors? 2M Ans. In pure silicon all electrons are involved in bon ...
... Number of atoms in 200g of the element =3.47×1024 . Q12. Pure silicon is an insulator. Silicon doped with phosphorus is a semiconductor. Silicon doped with gallium is also a semiconductor what is the difference between the two semiconductors? 2M Ans. In pure silicon all electrons are involved in bon ...
Chemistry - Department of Education and Skills
... have been selected around the content and structure of the syllabus to provide easy access to resource material in a way which supports the implementation of the course. However, it is important to realise that these modules do not define the syllabus. They do not determine the scope of the syllabus ...
... have been selected around the content and structure of the syllabus to provide easy access to resource material in a way which supports the implementation of the course. However, it is important to realise that these modules do not define the syllabus. They do not determine the scope of the syllabus ...
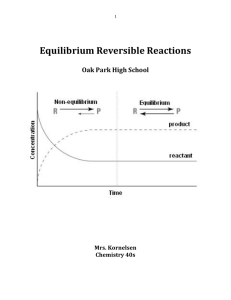
Equilibrium Reversible Reactions
... You start at the top (reactants) and end up at the bottom (products). But when you are partway down you start walking up the escalator as it continues going down. If you match your rate of w ...
... You start at the top (reactants) and end up at the bottom (products). But when you are partway down you start walking up the escalator as it continues going down. If you match your rate of w ...
Chem Soc Rev
... region. Various noble and non-noble catalysts have been reported as candidates for PROX catalysts. These have resulted in an interesting direction in ‘‘selective oxidation’’ (preferential oxidation) catalysis. Here, the control of selectivity means how we can accelerate the oxidation of CO while avo ...
... region. Various noble and non-noble catalysts have been reported as candidates for PROX catalysts. These have resulted in an interesting direction in ‘‘selective oxidation’’ (preferential oxidation) catalysis. Here, the control of selectivity means how we can accelerate the oxidation of CO while avo ...
Thermal Decomposition of Polymers - Marcelo Hirschler
... a fluid state. Due to their structure, it is not possible for the original material to change state at temperatures below that at which thermal decomposition occurs. Hence, there are no notable physical transformations in the material before decomposition. In cellulosic materials, there is an import ...
... a fluid state. Due to their structure, it is not possible for the original material to change state at temperatures below that at which thermal decomposition occurs. Hence, there are no notable physical transformations in the material before decomposition. In cellulosic materials, there is an import ...
Problem 1-2
... reacts in a vigorous and light-emitting reaction to form a grey-yellow powder (experiment A). If water is added to this grey-yellow powder (experiment B) moistened pH-paper turns blue if held over the mixture. The mixture is heated to boiling for some minutes and pH-paper is held into the solution. ...
... reacts in a vigorous and light-emitting reaction to form a grey-yellow powder (experiment A). If water is added to this grey-yellow powder (experiment B) moistened pH-paper turns blue if held over the mixture. The mixture is heated to boiling for some minutes and pH-paper is held into the solution. ...
Organic Chemistry/Fourth Edition: e-Text
... The mechanisms of all the reactions cited in Table 20.2 are similar to the mechanism of hydrolysis of an acyl chloride outlined in Figure 20.3. They differ with respect to the nucleophile that attacks the carbonyl group. In the first stage of the mechanism, water undergoes nucleophilic addition to t ...
... The mechanisms of all the reactions cited in Table 20.2 are similar to the mechanism of hydrolysis of an acyl chloride outlined in Figure 20.3. They differ with respect to the nucleophile that attacks the carbonyl group. In the first stage of the mechanism, water undergoes nucleophilic addition to t ...
From Kinetics to Equilibrium
... undergo a change in electrical conductivity. In the reaction above, hydrochloric acid is a mix of equal molar amounts of two ions: hydronium, H3O+ , and chloride, Cl− . The MgCl2 that is produced exists as three separate ions in solution: one Mg2+ ion and two Cl− ions. Since there is an increase in ...
... undergo a change in electrical conductivity. In the reaction above, hydrochloric acid is a mix of equal molar amounts of two ions: hydronium, H3O+ , and chloride, Cl− . The MgCl2 that is produced exists as three separate ions in solution: one Mg2+ ion and two Cl− ions. Since there is an increase in ...
102MSJc14 - Louisiana Tech University
... Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. Forward reaction is when reactants combine to form products whereas products are converted back to reactants in the backward reaction. In most chemical reactions, the rate of backward rea ...
... Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. Forward reaction is when reactants combine to form products whereas products are converted back to reactants in the backward reaction. In most chemical reactions, the rate of backward rea ...
chem 102 class notes - Louisiana Tech University
... Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. Forward reaction is when reactants combine to form products whereas products are converted back to reactants in the backward reaction. In most chemical reactions, the rate of backward rea ...
... Irreversible or complete reactions: Chemical reactions can be considered to have forward and backward reactions. Forward reaction is when reactants combine to form products whereas products are converted back to reactants in the backward reaction. In most chemical reactions, the rate of backward rea ...