
What is ligand?
... chemical compound with the formula CuSO4. This salt exists as a series of compounds that differ in their degree of hydration. The anhydrous form is a pale green or gray-white powder, while the pentahydrate, the most commonly encountered salt, is bright blue. This hydrated copper sulfate occurs in na ...
... chemical compound with the formula CuSO4. This salt exists as a series of compounds that differ in their degree of hydration. The anhydrous form is a pale green or gray-white powder, while the pentahydrate, the most commonly encountered salt, is bright blue. This hydrated copper sulfate occurs in na ...
Early transition metal clusters with π
... liquid crystalline properties and allow a large range of potential 'advanced materials' with interesting and unusual properties to be constructed. Liquid crystalline materials containing a metal atom in the organic molecular skeleton have properties derived from both the organic ligand and the metal ...
... liquid crystalline properties and allow a large range of potential 'advanced materials' with interesting and unusual properties to be constructed. Liquid crystalline materials containing a metal atom in the organic molecular skeleton have properties derived from both the organic ligand and the metal ...
H H H H H N HO O NC[ ]- - Teacher`s Tools® Chemistry
... Chemical Reactions: Complexation Reactions: Student Review Notes ...
... Chemical Reactions: Complexation Reactions: Student Review Notes ...
Electron Counting, Formal Oxidation States and the 18 Electron
... By this method, the metal and its bonded ligands are all counted as neutral fragments. Thus a methyl group is considered to be a 1 electron ligand because `CH3 is the corresponding neutral fragment. Similarly, ligands which are already neutral in their free state (eg. CO, pyridine, PMe3) are viewed ...
... By this method, the metal and its bonded ligands are all counted as neutral fragments. Thus a methyl group is considered to be a 1 electron ligand because `CH3 is the corresponding neutral fragment. Similarly, ligands which are already neutral in their free state (eg. CO, pyridine, PMe3) are viewed ...
NAME: Chem 1b, 2005, 3rd
... A periodic table and useful data are provided on the insert. You may use the insert for scratch work but enter all work to be graded in the space with each question. 1) (58 points) For each of the following species, give its IUPAC chemical name and the hybridization of the transition-metal atom. Dra ...
... A periodic table and useful data are provided on the insert. You may use the insert for scratch work but enter all work to be graded in the space with each question. 1) (58 points) For each of the following species, give its IUPAC chemical name and the hybridization of the transition-metal atom. Dra ...
ELECTRON COUNTING IN TRANSITION METAL COMPLEXES
... occurring. Because reactions involve the transfer of electrons from one atom to another during the making and breaking of chemical bonds, we need to keep track of where the electrons are going. Counting electrons on a transition metal in a coordination compound can be a little tricky. Instead of an ...
... occurring. Because reactions involve the transfer of electrons from one atom to another during the making and breaking of chemical bonds, we need to keep track of where the electrons are going. Counting electrons on a transition metal in a coordination compound can be a little tricky. Instead of an ...
Assignment #9 KEY 1. Most ligands are written as abbreviations
... En: bidentate, both nitrogen atoms are donors DMSO: monodentate, donor is either the sulfur or the oxygen EDTA: hexadentate, donor atoms are the nitrogen atoms (neutral) and the oxygen atoms (-1 charge for each) Dien: tridentate, donor atoms are the three nitrogen atoms Bipy: bidentate, donor atoms ...
... En: bidentate, both nitrogen atoms are donors DMSO: monodentate, donor is either the sulfur or the oxygen EDTA: hexadentate, donor atoms are the nitrogen atoms (neutral) and the oxygen atoms (-1 charge for each) Dien: tridentate, donor atoms are the three nitrogen atoms Bipy: bidentate, donor atoms ...
Crystal Field Theory
... There are only 4 ligands in the tetrahedral complex, and hence the ligand field is roughly 2/3 of the octahedral field. The direction of ligand approach in tetrahedral complex does not coincide with the d-orbitals. This reduces the field by a factor of 2/3. Therefore ∆t is roughly 2/3 x 2/3 = 4/9 of ...
... There are only 4 ligands in the tetrahedral complex, and hence the ligand field is roughly 2/3 of the octahedral field. The direction of ligand approach in tetrahedral complex does not coincide with the d-orbitals. This reduces the field by a factor of 2/3. Therefore ∆t is roughly 2/3 x 2/3 = 4/9 of ...
Chelate chemistry for molecular imaging Ligands versus Chelates
... On size matching of metal ion and ligand cavity Thermodynamics (how stable does the chelate need to be?) Kinetics of ML complex formation Charge on resulting chelate; perhaps it needs to be charged or neutral ...
... On size matching of metal ion and ligand cavity Thermodynamics (how stable does the chelate need to be?) Kinetics of ML complex formation Charge on resulting chelate; perhaps it needs to be charged or neutral ...
Chapter 1 Structure and Bonding
... b) 18-electron complex: Fe2+ = d6 and 2 Cp x 6 ec) Bonding Group Orbitals of 2 eclipsed Cp rings ...
... b) 18-electron complex: Fe2+ = d6 and 2 Cp x 6 ec) Bonding Group Orbitals of 2 eclipsed Cp rings ...
03 Complexation equilibrium
... Those that bond through electron pairs on more than one donor atom are termed polydentate ligands (“many-toothed” ligands). For example, ethylenediamine (NH2CH2CH2NH2 abbreviated en) is a bidentate ligand because it bonds to a metal using an electron pair on each of its two nitrogen atoms. ...
... Those that bond through electron pairs on more than one donor atom are termed polydentate ligands (“many-toothed” ligands). For example, ethylenediamine (NH2CH2CH2NH2 abbreviated en) is a bidentate ligand because it bonds to a metal using an electron pair on each of its two nitrogen atoms. ...
Chapter 21 Transition Metals and Coordination Chemistry
... A coordination compound consists of a complex ion along with one or more counterions which are anions or cations required to produce a compound with no net charge Square brackets are placed around the complex ion to indicate the composition of the complex ion Counterions are shown outside the bracke ...
... A coordination compound consists of a complex ion along with one or more counterions which are anions or cations required to produce a compound with no net charge Square brackets are placed around the complex ion to indicate the composition of the complex ion Counterions are shown outside the bracke ...
H,mR,cn,nii,f^{aq) + H,O(1)
... sites that are ideally suited to binding hard Fe^-^. The ligand has a flexible organic backbone and can wrap around the metal ion forming a complex with three 5membered chelate rings involving three ONCO-unite. This can be represented below and explains the origin of the term chelation therapy. ...
... sites that are ideally suited to binding hard Fe^-^. The ligand has a flexible organic backbone and can wrap around the metal ion forming a complex with three 5membered chelate rings involving three ONCO-unite. This can be represented below and explains the origin of the term chelation therapy. ...
(Lecture(25) - MSU Chemistry
... spacing'between'energy'levels'(ΔE).'Coordination'compounds'absorb'visible'and' ultraviolet'light'when'the'photon'energy'matches'the'spacing'between'd'orbitals'split' by'the'ligand'field.' • The'perceived'color'of'an'object'comes'from'the'visible'light'that'is'reflected'and'not' absorbed.'(FigureLMcM ...
... spacing'between'energy'levels'(ΔE).'Coordination'compounds'absorb'visible'and' ultraviolet'light'when'the'photon'energy'matches'the'spacing'between'd'orbitals'split' by'the'ligand'field.' • The'perceived'color'of'an'object'comes'from'the'visible'light'that'is'reflected'and'not' absorbed.'(FigureLMcM ...
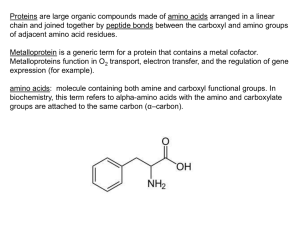
amino acids
... sensitive to both the ligand donor atoms and the stereochemistry at the metal centre. Example: Cu(I) adopts tetrahedral or trigonal geometries and prefers soft donor atoms. Cu(II) adopts square planar (or square pyramidal or octahedral) geometry and prefers harder donor atoms. By changing the ligand ...
... sensitive to both the ligand donor atoms and the stereochemistry at the metal centre. Example: Cu(I) adopts tetrahedral or trigonal geometries and prefers soft donor atoms. Cu(II) adopts square planar (or square pyramidal or octahedral) geometry and prefers harder donor atoms. By changing the ligand ...
Electronic spectrum of a 0.1 M aqueous solution of [Ti(H2O)6]3+
... The spectrochemical series is therefore at odds with the assumption of purely electrostatic interactions between ligands and metal ions (CFT). The observations mentioned above have led to the development of the ligand field theory, which allows for (and describes) covalent interactions between ligan ...
... The spectrochemical series is therefore at odds with the assumption of purely electrostatic interactions between ligands and metal ions (CFT). The observations mentioned above have led to the development of the ligand field theory, which allows for (and describes) covalent interactions between ligan ...
A Summary of Organometallic Chemistry
... Exceptions to the 18-electrons rule - Early transition metal complexes and/or complexes with bulky ligands are stable with less than 18 v.e. mainly for steric reasons. - Complexes with many π-donor ligands (see below) are stable even with less than 18 v.e. because the πdonation adds electron densit ...
... Exceptions to the 18-electrons rule - Early transition metal complexes and/or complexes with bulky ligands are stable with less than 18 v.e. mainly for steric reasons. - Complexes with many π-donor ligands (see below) are stable even with less than 18 v.e. because the πdonation adds electron densit ...
Chapter 23 – Transition Metals and Coordination Chemistry
... Complex of porphine and metal is known as porphyrin. Variations possess diff. metals, diff. groups attached to porphine. This type of complex is a component of myoglobin (stores oxygen), hemoglobin (transports oxygen in blood) and chlorophyl (needed for photosynthesis in plants). The iron in hemoglo ...
... Complex of porphine and metal is known as porphyrin. Variations possess diff. metals, diff. groups attached to porphine. This type of complex is a component of myoglobin (stores oxygen), hemoglobin (transports oxygen in blood) and chlorophyl (needed for photosynthesis in plants). The iron in hemoglo ...
Chapter 23 - Transition Metals and Coordination Chemistry
... CoCl3 • 3NH3 + 3 Ag+ → 0 AgCl(s) i.e. no rxn not all Cl– in the compounds are equal ...
... CoCl3 • 3NH3 + 3 Ag+ → 0 AgCl(s) i.e. no rxn not all Cl– in the compounds are equal ...
TRANSITION METALS - Pennsylvania State University
... Ligands bonded to metal ions metal complexes or coordination compounds. Coordination number: number of electron donor atoms attached to the metal. Chelates are ligands possessing two or more donor atoms. ...
... Ligands bonded to metal ions metal complexes or coordination compounds. Coordination number: number of electron donor atoms attached to the metal. Chelates are ligands possessing two or more donor atoms. ...
role of the ancillary ligands on the coordination modes
... Nuovi farmaci inorganici in oncologia agreement with the formation of a single reaction product. In the 1H NMR spectrum, the presence of one of the NH resonances at δ 10.6 suggests that platination of the neutral nucleobase occurs at the N(4) atom, as result of a tautomeric shift of one of the exocy ...
... Nuovi farmaci inorganici in oncologia agreement with the formation of a single reaction product. In the 1H NMR spectrum, the presence of one of the NH resonances at δ 10.6 suggests that platination of the neutral nucleobase occurs at the N(4) atom, as result of a tautomeric shift of one of the exocy ...
Electron Counting 2000
... more instructional "classification" of organometallic compounds has been suggested by Green. We will describe this in some detail later. ...
... more instructional "classification" of organometallic compounds has been suggested by Green. We will describe this in some detail later. ...
metal ion
... If the ligand is a negative ion: ‘ide’ is dropped and replaced with ‘o’ e.g. chloride becomes chloro , cyanide becomes cyano; ‘ate’ and ‘ite’ change to ‘ato’ or ‘ito’ e.g. oxalate becomes oxalato, nitrite becomes nitrito. ...
... If the ligand is a negative ion: ‘ide’ is dropped and replaced with ‘o’ e.g. chloride becomes chloro , cyanide becomes cyano; ‘ate’ and ‘ite’ change to ‘ato’ or ‘ito’ e.g. oxalate becomes oxalato, nitrite becomes nitrito. ...
FACTORS AFFECT
... VALENCE BOND THEORY This theory was developed by Linus Pauling. According to this theory the bonding in metal complexes arises when a filled ligand orbital containing a pair of electrons overlaps with a vacant hybrid orbital on the metal atom. The VBT assumes the bonding between the metal atom an ...
... VALENCE BOND THEORY This theory was developed by Linus Pauling. According to this theory the bonding in metal complexes arises when a filled ligand orbital containing a pair of electrons overlaps with a vacant hybrid orbital on the metal atom. The VBT assumes the bonding between the metal atom an ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.

![H H H H H N HO O NC[ ]- - Teacher`s Tools® Chemistry](http://s1.studyres.com/store/data/017018154_1-68467b392d8cadb27de319df72045839-300x300.png)












![Electronic spectrum of a 0.1 M aqueous solution of [Ti(H2O)6]3+](http://s1.studyres.com/store/data/005719667_1-a6d66a78471c6778162e27e0ef555131-300x300.png)







