Problem Set 5_Chem165_Spring14
... there have been a lot of small leaks. That’s not good, and it needs to be fixed. But the cleanup needs to be done carefully and thoughtfully. The materials is unbelievably hazardous and corrosive, so the process can’t be rushed and the government contractors are going to make mistakes. In my view, i ...
... there have been a lot of small leaks. That’s not good, and it needs to be fixed. But the cleanup needs to be done carefully and thoughtfully. The materials is unbelievably hazardous and corrosive, so the process can’t be rushed and the government contractors are going to make mistakes. In my view, i ...
The complex in biological systems Plan 1. Definition of complex
... functional groups - COOH, NH2 - nitrogen primary, secondary are able to coordinate ions metals. Examples include porphyry system. It etradentate macromolecule. Schematically, are as follows: ...
... functional groups - COOH, NH2 - nitrogen primary, secondary are able to coordinate ions metals. Examples include porphyry system. It etradentate macromolecule. Schematically, are as follows: ...
Absorption Spectra and Colours of Complexes
... The reason that the two species are differently colored is because the d-orbital splitting energy is different when chloride is a ligand than when water is a ligand. It is possible to arrange ligands into a series that reflects their ability to split the d-orbitals. This series is essentially the sa ...
... The reason that the two species are differently colored is because the d-orbital splitting energy is different when chloride is a ligand than when water is a ligand. It is possible to arrange ligands into a series that reflects their ability to split the d-orbitals. This series is essentially the sa ...
Periodicity (AHL)
... How does the metal attract so many ligands?? You may be wondering why a metal ion will attract more ligands than it has charges. +2 should attract -2 and +3 should attract -3, right?? Let’s look at an example: ...
... How does the metal attract so many ligands?? You may be wondering why a metal ion will attract more ligands than it has charges. +2 should attract -2 and +3 should attract -3, right?? Let’s look at an example: ...
Chapter 2 - U of L Class Index
... Recall that photons are emitted when electrons drop from a higher energy orbital to a lower energy orbital. (see Atomic Line Spectra in Chemistry 1000) Similarly, the electrons get to the higher energy orbital by absorbing photons of light. In co-ordination complexes with crystal field splitting, th ...
... Recall that photons are emitted when electrons drop from a higher energy orbital to a lower energy orbital. (see Atomic Line Spectra in Chemistry 1000) Similarly, the electrons get to the higher energy orbital by absorbing photons of light. In co-ordination complexes with crystal field splitting, th ...
- LSU Chemistry
... Electron Density: The presence and number of valence electrons around an atom. Electrons are represented by a probability distribution spread out over a region of space defined by the orbital: s, p, d, f, and/or hybrid orbitals such sp3, sp2, sp, etc. Atoms with quite a few valence electrons such as ...
... Electron Density: The presence and number of valence electrons around an atom. Electrons are represented by a probability distribution spread out over a region of space defined by the orbital: s, p, d, f, and/or hybrid orbitals such sp3, sp2, sp, etc. Atoms with quite a few valence electrons such as ...
Document
... All s interactions with the ligands are stabilizing to the ligands and destabilizing to the d orbitals. The interaction of a ligand with a d orbital depends on their orientation with respect to each other, estimated by their overlap which can be calculated. The total destabilization of a d orbital c ...
... All s interactions with the ligands are stabilizing to the ligands and destabilizing to the d orbitals. The interaction of a ligand with a d orbital depends on their orientation with respect to each other, estimated by their overlap which can be calculated. The total destabilization of a d orbital c ...
Coordination Compounds Coordination
... In coordination compounds, there are two types of linkages (valences) – primary and secondary. The primary valences are ionisable and are satisfied by negative ions. The secondary valences are non-ionisable and are satisfied by negative ions or neutral molecules. The secondary valence is equal to th ...
... In coordination compounds, there are two types of linkages (valences) – primary and secondary. The primary valences are ionisable and are satisfied by negative ions. The secondary valences are non-ionisable and are satisfied by negative ions or neutral molecules. The secondary valence is equal to th ...
Chapter 22 - U of L Class Index
... Recall that photons are emitted when electrons drop from a higher energy orbital to a lower energy orbital. (see Atomic Line Spectra in Chemistry 1000) Similarly, the electrons get to the higher energy orbital by absorbing photons of light. In co-ordination complexes with crystal field splitting, th ...
... Recall that photons are emitted when electrons drop from a higher energy orbital to a lower energy orbital. (see Atomic Line Spectra in Chemistry 1000) Similarly, the electrons get to the higher energy orbital by absorbing photons of light. In co-ordination complexes with crystal field splitting, th ...
Supramolecular photochemistry
... several research groups because of a unique combination of ground and excited states properties (ref. 13). The prototype of these complexes is the famous Ru(bpy)$' that is extensively used as photoluminescent compound and sensitizer in the interconversion of light and chemical energy. Comparison bet ...
... several research groups because of a unique combination of ground and excited states properties (ref. 13). The prototype of these complexes is the famous Ru(bpy)$' that is extensively used as photoluminescent compound and sensitizer in the interconversion of light and chemical energy. Comparison bet ...
Slide 1
... From the other hand, Dt is usually small and the electron count up to 18 electrons for ML4 is s possible but rare. ...
... From the other hand, Dt is usually small and the electron count up to 18 electrons for ML4 is s possible but rare. ...
Chelating agents are ligands for metals that bind via
... mercury, arsenic, and lead, by converting them to a chemically inert form that can be excreted without further interaction with the body. This therapy was approved by the U.S. Food and Drug Administration in 1991. Although they can be beneficial in cases of heavy metal poisoning, chelating agents al ...
... mercury, arsenic, and lead, by converting them to a chemically inert form that can be excreted without further interaction with the body. This therapy was approved by the U.S. Food and Drug Administration in 1991. Although they can be beneficial in cases of heavy metal poisoning, chelating agents al ...
Document
... • Formation of chelate ring ⇒ reaction proceeds in forward direction & the product is stable. This stability is purely kinetic in nature. This is known as chelate effect. • ΔG = ΔH - TΔS ...
... • Formation of chelate ring ⇒ reaction proceeds in forward direction & the product is stable. This stability is purely kinetic in nature. This is known as chelate effect. • ΔG = ΔH - TΔS ...
CBSE Class 12 Chemistry notes and questions for
... Didentate: Ligates through two donor atoms. Ex- C2O4 2- (ox); H2NCH2CH2NH2(en) c) Polydentate: which ligates through two or more donor atoms present in a single ligand. Ex(EDTA)4d) Chelating ligands: Di- or polydentate ligands that uses two or more donor atoms to bind to a single metal ion to form r ...
... Didentate: Ligates through two donor atoms. Ex- C2O4 2- (ox); H2NCH2CH2NH2(en) c) Polydentate: which ligates through two or more donor atoms present in a single ligand. Ex(EDTA)4d) Chelating ligands: Di- or polydentate ligands that uses two or more donor atoms to bind to a single metal ion to form r ...
Modelling for Coordination Complexes: Structure
... which already causes problems since now there are two possible ‘ideal’ angles at the metal – 90 or 180° – but the L-M-L triad is the same. A simple remedy to this problem is to look at a model like valence shell electron pair repulsion (VSEPR) theory covered in most first year undergraduate chemistr ...
... which already causes problems since now there are two possible ‘ideal’ angles at the metal – 90 or 180° – but the L-M-L triad is the same. A simple remedy to this problem is to look at a model like valence shell electron pair repulsion (VSEPR) theory covered in most first year undergraduate chemistr ...
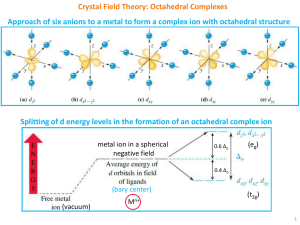
Crystal Field Theory: Octahedral Complexes
... There are only 4 ligands in the tetrahedral complex and hence the ligand field is roughly 2/3 of the octahedral field. The direction of ligand approach in tetrahedral complex does not coincide with the d-orbitals. This reduces the field by a factor of 2/3. Therefore Δt is roughly 2/3 x 2/3 = 4/9 ...
... There are only 4 ligands in the tetrahedral complex and hence the ligand field is roughly 2/3 of the octahedral field. The direction of ligand approach in tetrahedral complex does not coincide with the d-orbitals. This reduces the field by a factor of 2/3. Therefore Δt is roughly 2/3 x 2/3 = 4/9 ...
Transition Elements and Complexes
... Central metal cation surrounded by ligands (anions or molecules) Ligand is bonded to metal through an electron pair Ligands act like Lewis bases by donating an electron pair Ligands bond in first coordination sphere [does not dissociate in water] Coordination number- the number of atoms bonded direc ...
... Central metal cation surrounded by ligands (anions or molecules) Ligand is bonded to metal through an electron pair Ligands act like Lewis bases by donating an electron pair Ligands bond in first coordination sphere [does not dissociate in water] Coordination number- the number of atoms bonded direc ...
What Is A Coordination Compound? A coordination complex is the
... What Is A Coordination Compound? A coordination complex is the product of a Lewis acid-base reaction in which neutral molecules or anions (called ligands) bond to a central metal atom (or ion) by coordinate covalent bonds. ...
... What Is A Coordination Compound? A coordination complex is the product of a Lewis acid-base reaction in which neutral molecules or anions (called ligands) bond to a central metal atom (or ion) by coordinate covalent bonds. ...
Trace Metal Biogeochemistry (Marine Bioinorganic Chemistry
... Tables in Morel and Hering and Stumm and Morgan are made for teaching They have been back corrected to zero ionic strength from constants If your application really matters, go to the literature or NIST databases for each constant You can use the textbooks as guidelines of species to look for though ...
... Tables in Morel and Hering and Stumm and Morgan are made for teaching They have been back corrected to zero ionic strength from constants If your application really matters, go to the literature or NIST databases for each constant You can use the textbooks as guidelines of species to look for though ...
Chemistry of Transition Metals
... Imagine a tetrahedral molecule inside a cube with metal ions in the center of the cube. The ligands occupy the four alternate corners of the cube leaving the rest four corners empty. The two ‘e’ orbitals point to the center of the face of the cube while the three ‘t2’ orbitals point to the center of ...
... Imagine a tetrahedral molecule inside a cube with metal ions in the center of the cube. The ligands occupy the four alternate corners of the cube leaving the rest four corners empty. The two ‘e’ orbitals point to the center of the face of the cube while the three ‘t2’ orbitals point to the center of ...
Assignment 5 (key)
... This iron complex is three-coordinate, which is unusually low. It should have trigonal planar geometry: the sum of the bond angles is 360°, which is consistent with planarity at Fe, but the angles are distorted from the usual 120° we’d expect. This is because the two bulky “silyl” ligands, SiR3– (wh ...
... This iron complex is three-coordinate, which is unusually low. It should have trigonal planar geometry: the sum of the bond angles is 360°, which is consistent with planarity at Fe, but the angles are distorted from the usual 120° we’d expect. This is because the two bulky “silyl” ligands, SiR3– (wh ...
Structural Knowledge Base Development for Metal Complexes
... Bond lengths, valence angles and torsion angles Compiled from the CSD ...
... Bond lengths, valence angles and torsion angles Compiled from the CSD ...
Metal Complexes
... Metal Complexes • metal cation is attached to a group of surrounding molecules or ions (ligands) by coordinate covalent bonds – coordinate => ligand donates both electrons ...
... Metal Complexes • metal cation is attached to a group of surrounding molecules or ions (ligands) by coordinate covalent bonds – coordinate => ligand donates both electrons ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.