Chapter 1 Structure and Bonding
... 1) Ligand Field Theory shortcomings a) Energy of interactions are ambiguous b) Very complicated for multiple ligand types or non-standard geometries ...
... 1) Ligand Field Theory shortcomings a) Energy of interactions are ambiguous b) Very complicated for multiple ligand types or non-standard geometries ...
Ch 24 Part 2 PowerPoint
... We apply Hund’s rule to the 2 sets of 5 d-orbitals. The first three e-’s go into different d orbitals with their spins parallel. We have a choice for the placement of the fourth electron: if it goes into a higher energy orbital, then there is an energy cost associated with promotion (D); i ...
... We apply Hund’s rule to the 2 sets of 5 d-orbitals. The first three e-’s go into different d orbitals with their spins parallel. We have a choice for the placement of the fourth electron: if it goes into a higher energy orbital, then there is an energy cost associated with promotion (D); i ...
Ligand field theory
... increases with oxidation state and increases down a group I- < Br- < SCN- ~Cl- < F- < OH- ~ ...
... increases with oxidation state and increases down a group I- < Br- < SCN- ~Cl- < F- < OH- ~ ...
Lecture 2
... Most convenient to use a local coordinate system on each ligand with y pointing in towards the metal. py to be used for s bonding. z being perpendicular to the molecular plane. pz to be used for p bonding perpendicular to the plane, p^. x lying in the molecular plane. px to be used for p bonding in ...
... Most convenient to use a local coordinate system on each ligand with y pointing in towards the metal. py to be used for s bonding. z being perpendicular to the molecular plane. pz to be used for p bonding perpendicular to the plane, p^. x lying in the molecular plane. px to be used for p bonding in ...
Classification of Ligands
... Overview of Transition Metal Complexes 1.The coordinate covalent or dative bond applies in L:M 2.Lewis bases are called LIGANDS—all serve as σ-donors some are π-donors as well, and some are π-acceptors 3. Specific coordination number and geometries depend on metal and number of d-electrons 4. HSAB ...
... Overview of Transition Metal Complexes 1.The coordinate covalent or dative bond applies in L:M 2.Lewis bases are called LIGANDS—all serve as σ-donors some are π-donors as well, and some are π-acceptors 3. Specific coordination number and geometries depend on metal and number of d-electrons 4. HSAB ...
The chemistry of the transition metals
... • 1st & 2nd rows different, but 2nd & 3rd same – Due to small change in atomic size going down group w/large increase in nuclear charge ...
... • 1st & 2nd rows different, but 2nd & 3rd same – Due to small change in atomic size going down group w/large increase in nuclear charge ...
Lecture Notes 14 - La Salle University
... The effectiveness of many transition metal compounds as catalysts for reactions comes from the facility of these metals to complex reversibly with a variety of functional groups. ...
... The effectiveness of many transition metal compounds as catalysts for reactions comes from the facility of these metals to complex reversibly with a variety of functional groups. ...
Transition Metals
... in space than are the 3d orbitals. They have a higher quantum number n and the have fewer nodes (0 or 1) versus 2 for the d orbitals. ...
... in space than are the 3d orbitals. They have a higher quantum number n and the have fewer nodes (0 or 1) versus 2 for the d orbitals. ...
Coordination
... M with empty d- orbitals Ligand with e- pair Combine to form LA-LB (Compound) In general, the complex is more stable than the separated compound. ...
... M with empty d- orbitals Ligand with e- pair Combine to form LA-LB (Compound) In general, the complex is more stable than the separated compound. ...
Transition Metals and Coordination Chem
... M with empty d- orbitals Ligand with e- pair Combine to form LA-LB (Compound) In general, the complex is more stable than the separated compound. ...
... M with empty d- orbitals Ligand with e- pair Combine to form LA-LB (Compound) In general, the complex is more stable than the separated compound. ...
(Phosphines) - Bite angles for bidentate ligands
... Phosphines :PR3 - very important ligands - -donors - p-acceptors For years, it was assumed that p-backdonation occurred from the metal into empty d-orbitals on phosphorus. Actually: p-backdonation occurs into MOs formed by combination of two d-orbitals on phosphorus and the * orbitals involved ...
... Phosphines :PR3 - very important ligands - -donors - p-acceptors For years, it was assumed that p-backdonation occurred from the metal into empty d-orbitals on phosphorus. Actually: p-backdonation occurs into MOs formed by combination of two d-orbitals on phosphorus and the * orbitals involved ...
Phosphines - U of L Class Index
... Phosphines/Arsines in early TM and lanthanide chemistry • PR3 excellent ligands for soft late transition metals • PR3 typically NOT v. suitable to form complexes with hard early TMs (PR3 ...
... Phosphines/Arsines in early TM and lanthanide chemistry • PR3 excellent ligands for soft late transition metals • PR3 typically NOT v. suitable to form complexes with hard early TMs (PR3 ...
Chem 174_Lecture 10a..
... • L as p-complex only (C2H4, alkenes) • Ligands like ethylene form strong p-complexes with low-valent metals • The HOMO is the C=C p-bond, which is used to form the M-L s-bond • Often times, there is also a back-bonding into the p*-orbital of the C=C bond (free C2H4: 134 pm, complex: 137 pm) • Examp ...
... • L as p-complex only (C2H4, alkenes) • Ligands like ethylene form strong p-complexes with low-valent metals • The HOMO is the C=C p-bond, which is used to form the M-L s-bond • Often times, there is also a back-bonding into the p*-orbital of the C=C bond (free C2H4: 134 pm, complex: 137 pm) • Examp ...
Irradiations of the transition metal-to
... excited states. The lifetimes of most of the MMCT excited states increase more than 10-fold when their am(m)ine ligands are perdueterated. These unique emissions have weak, low frequency vibronic sidebands that correspond to the small excited-state distortions in metal−ligand bonds that are characte ...
... excited states. The lifetimes of most of the MMCT excited states increase more than 10-fold when their am(m)ine ligands are perdueterated. These unique emissions have weak, low frequency vibronic sidebands that correspond to the small excited-state distortions in metal−ligand bonds that are characte ...
axial - TAMU Chemistry
... SO32 sulfito (b) neutral ligands same as molecule name with a few exceptions: NH3 → ammonia ...
... SO32 sulfito (b) neutral ligands same as molecule name with a few exceptions: NH3 → ammonia ...
Lecture3
... The only restriction of using BuLi is that it can sometimes act not only as a base but also as a nucleophile. In these cases, it is necessary to prepare a bulky base by reaction of n-butyllithium with a bulky amine such as ...
... The only restriction of using BuLi is that it can sometimes act not only as a base but also as a nucleophile. In these cases, it is necessary to prepare a bulky base by reaction of n-butyllithium with a bulky amine such as ...
Transition Metals Complexes
... Complex compounds consist of a central metal atom surrounded by ligands. A ligand is an electron pair donor. A ligand may be an atom, an ion or a molecule. N.B. Ligands, nucleophiles and Lewis bases are all defined as electron pair donors. Since the ligand donates both of the electrons it shares wit ...
... Complex compounds consist of a central metal atom surrounded by ligands. A ligand is an electron pair donor. A ligand may be an atom, an ion or a molecule. N.B. Ligands, nucleophiles and Lewis bases are all defined as electron pair donors. Since the ligand donates both of the electrons it shares wit ...
23/24 Transition, Nuclear
... A radioactive element has a half-life of 24 days. What is the rate constant? How long would it take for 1.5 x 1021 atoms of the substance to decay down to 2.7 x 1019 atoms? How many atoms would be left after a year (365 days)? ...
... A radioactive element has a half-life of 24 days. What is the rate constant? How long would it take for 1.5 x 1021 atoms of the substance to decay down to 2.7 x 1019 atoms? How many atoms would be left after a year (365 days)? ...
Activity 2 - Why Do the D-orbitals Give us Colour
... might need to look up the names of the arrangements. In the octahedral arrangement it is found that the different d-orbitals (remember Figure 3) interact differently with the electrons in the ligands (remember the electrons in the ligands will repel the electrons in the metal centre, hence raising t ...
... might need to look up the names of the arrangements. In the octahedral arrangement it is found that the different d-orbitals (remember Figure 3) interact differently with the electrons in the ligands (remember the electrons in the ligands will repel the electrons in the metal centre, hence raising t ...
Chemistry of Coordination Compounds
... while the purple compound only has 5 ammonia molecules in the coordinated compound. As shown in the ball-and-stick model, the chlorides serve as counter ions to the cobalt/ammonia coordiation complex in the orange compound, while one of the ammonia molecules is replaced by Cl in the purple compound. ...
... while the purple compound only has 5 ammonia molecules in the coordinated compound. As shown in the ball-and-stick model, the chlorides serve as counter ions to the cobalt/ammonia coordiation complex in the orange compound, while one of the ammonia molecules is replaced by Cl in the purple compound. ...
coinage metal complexes containing new scorpionate
... F. Tisato4 1. C.I.R.C.M.S.B. - Unità di Ricerca di Camerino 2. Dipartimento di Scienze Chimiche, Università di Camerino 3. Dipartimento di Scienze Farmaceutiche, Università di Padova 4. ICIS-C.N.R., Corso Stati Uniti, 4, Padova e-mail: [email protected] ...
... F. Tisato4 1. C.I.R.C.M.S.B. - Unità di Ricerca di Camerino 2. Dipartimento di Scienze Chimiche, Università di Camerino 3. Dipartimento di Scienze Farmaceutiche, Università di Padova 4. ICIS-C.N.R., Corso Stati Uniti, 4, Padova e-mail: [email protected] ...
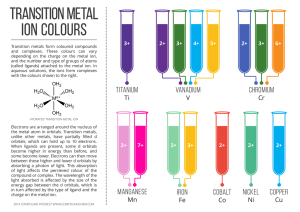
transition metal - Compound Interest
... the metal atom in orbitals. Transition metals, unlike other metals, have partially filled d orbitals, which can hold up to 10 electrons. When ligands are present, some d orbitals become higher in energy than before, and some become lower. Electrons can then move between these higher and lower d orbi ...
... the metal atom in orbitals. Transition metals, unlike other metals, have partially filled d orbitals, which can hold up to 10 electrons. When ligands are present, some d orbitals become higher in energy than before, and some become lower. Electrons can then move between these higher and lower d orbi ...
Transition metals and complex ions
... Explain the term ligand in terms of coordinate bonding. Describe and use the terms: complex ion and coordination number. State and give examples of complexes with six-fold coordination with an octahedral shape. ...
... Explain the term ligand in terms of coordinate bonding. Describe and use the terms: complex ion and coordination number. State and give examples of complexes with six-fold coordination with an octahedral shape. ...
Coordination Chemistry
... from one chemical species is donated to an empty orbital on another chemical species to form the new bond. This is different from a covalent bond because both electrons come from one atom or molecule but are shared as in a typical covalent bond. Unlike an ionic bond, a coordinate covalent bond does ...
... from one chemical species is donated to an empty orbital on another chemical species to form the new bond. This is different from a covalent bond because both electrons come from one atom or molecule but are shared as in a typical covalent bond. Unlike an ionic bond, a coordinate covalent bond does ...
Summary of Crystal Field Theory - uni
... For octahedral complexes with the electronic configurations d4, d5, d6 and d7 exist two possibilities to fill the orbitals with electrons, depending on the value of Δo in comparison to the value of the spin pairing energy. In complexes with strong ligands, which cause a large Δo, first the lower ene ...
... For octahedral complexes with the electronic configurations d4, d5, d6 and d7 exist two possibilities to fill the orbitals with electrons, depending on the value of Δo in comparison to the value of the spin pairing energy. In complexes with strong ligands, which cause a large Δo, first the lower ene ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.