chapter 10: coordination chemistry ii: bonding
... If we assume that each low spin configuration will have the maximum number of paired electrons, then the configurations with asymmetrically occupied degenerate orbitals are those for d1, d3, d5, and d7 metals. These are expected to give rise to Jahn-Teller distortion. ...
... If we assume that each low spin configuration will have the maximum number of paired electrons, then the configurations with asymmetrically occupied degenerate orbitals are those for d1, d3, d5, and d7 metals. These are expected to give rise to Jahn-Teller distortion. ...
Transition Metal Complexes: Definitions and Terminology.
... Neither has an aqueous solution that is basic to pH paper, therefore all the ammonias are bonded to cobalt (III). The obvious possibility is that the ambidentate nitrite group is differently bonded in these two complexes: [Co(NH3)5NO2]Cl2 and [Co(NH3)5ONO]Cl2. Today, we would assign the structures o ...
... Neither has an aqueous solution that is basic to pH paper, therefore all the ammonias are bonded to cobalt (III). The obvious possibility is that the ambidentate nitrite group is differently bonded in these two complexes: [Co(NH3)5NO2]Cl2 and [Co(NH3)5ONO]Cl2. Today, we would assign the structures o ...
FULL PAPER
... the title compounds are governed by hydrogen bonding. Two peculiarities that are typical for polyolato ± metal structures are apparent: 1) an almost balanced donor:acceptor ratio, and 2) the predominance of cooperative hydrogen bond sequences. The structure of 4 may be analyzed as an example. Examin ...
... the title compounds are governed by hydrogen bonding. Two peculiarities that are typical for polyolato ± metal structures are apparent: 1) an almost balanced donor:acceptor ratio, and 2) the predominance of cooperative hydrogen bond sequences. The structure of 4 may be analyzed as an example. Examin ...
unit 5 revision checklist - A
... Recall that most metal ions exist in aqueous solution as the hexaaqua complex, write equations to show why these complexes are acidic and explain why +3 complexes are more acidic than +2 complexes ...
... Recall that most metal ions exist in aqueous solution as the hexaaqua complex, write equations to show why these complexes are acidic and explain why +3 complexes are more acidic than +2 complexes ...
the PDF file
... (vi) Heteroleptic complexes: Heteroleptic complexes are those complexes where the central metal ion is bound to more than one type of a donor group. ...
... (vi) Heteroleptic complexes: Heteroleptic complexes are those complexes where the central metal ion is bound to more than one type of a donor group. ...
Open Access
... and the high valence clusters. High valence metal clusters contains metals in a relatively high oxidation states, usually +2 or +3. These compounds are usually made up of early transition metals and have good π donors as ligands such as chloride, bromide and iodine. The low-valence clusters on the o ...
... and the high valence clusters. High valence metal clusters contains metals in a relatively high oxidation states, usually +2 or +3. These compounds are usually made up of early transition metals and have good π donors as ligands such as chloride, bromide and iodine. The low-valence clusters on the o ...
Learning Objectives
... MA2B4 and MA3B3. 4) To give examples of optical isomerism for octahedral complexes. 5) Define ionization isomerism and linkage isomerism 6) To identify what types of isomerism can occur for a given octahedral or square planar complex. 7) Be able to perform electron counts for low oxidation state met ...
... MA2B4 and MA3B3. 4) To give examples of optical isomerism for octahedral complexes. 5) Define ionization isomerism and linkage isomerism 6) To identify what types of isomerism can occur for a given octahedral or square planar complex. 7) Be able to perform electron counts for low oxidation state met ...
course outline - Midlands State University
... The course is a foundation for Chemistry in general upon which the understanding of Chemistry in general is based . It is especially important to have sufficient appreciation of the theories of bonding and how some atomic and molecular properties such as magnetism and electronic properties can be ex ...
... The course is a foundation for Chemistry in general upon which the understanding of Chemistry in general is based . It is especially important to have sufficient appreciation of the theories of bonding and how some atomic and molecular properties such as magnetism and electronic properties can be ex ...
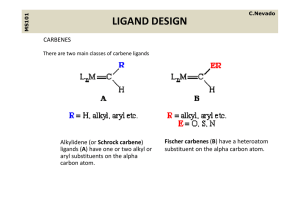
ligand design - UZH - Department of Chemistry
... containing pi acceptor ligands These are electrophilic at the alpha carbon count as neutral two containing pi‐acceptor ligands. These are electrophilic at the alpha‐carbon, count as neutral two‐ electron donor ligands, their ligation being similar to CO. We can draw a resonance structure as follows ...
... containing pi acceptor ligands These are electrophilic at the alpha carbon count as neutral two containing pi‐acceptor ligands. These are electrophilic at the alpha‐carbon, count as neutral two‐ electron donor ligands, their ligation being similar to CO. We can draw a resonance structure as follows ...
I find searching for polymeric structures difficult and I get results I
... differently defined asymmetric units in related structures. Therefore, if you are searching for any structure where, for example, the metal coordination sphere is completed by polymeric ligand atoms, or you have drawn an extensive substructural unit of an organic polymer (which may include atoms not ...
... differently defined asymmetric units in related structures. Therefore, if you are searching for any structure where, for example, the metal coordination sphere is completed by polymeric ligand atoms, or you have drawn an extensive substructural unit of an organic polymer (which may include atoms not ...
S R (II) N-
... configuration is [Kr] 4d75s. One relevant feature for this element together with the osmium, is the diversity of the oxidation states that can display its complexes (from -2 in [Ru(CO)4]2-] to +8 in [RuO4]). In addition, it can present several coordination geometries (trigonal bipyramid, octahedral. ...
... configuration is [Kr] 4d75s. One relevant feature for this element together with the osmium, is the diversity of the oxidation states that can display its complexes (from -2 in [Ru(CO)4]2-] to +8 in [RuO4]). In addition, it can present several coordination geometries (trigonal bipyramid, octahedral. ...
Answers to Topic 15 Exercises - A
... Prepare samples of the transition metal ion over a range of known concentrations. Add a small quantity of a suitable ligand to each in order to identify the colour. Choose the filter which gives the largest absorbance, and measure the absorbance of each sample using this filter. Plot a graph of abso ...
... Prepare samples of the transition metal ion over a range of known concentrations. Add a small quantity of a suitable ligand to each in order to identify the colour. Choose the filter which gives the largest absorbance, and measure the absorbance of each sample using this filter. Plot a graph of abso ...
View PDF
... occurs widely in systems where metal ions and two or more different ligands are present7. Way back in 1971 it was reported by Hallman et.al8 that at the physiological pH of 7.4 about 98% of copper (II) in the simulated plasma solution is present as a mixed complex of copper-histidine-cysteine, while ...
... occurs widely in systems where metal ions and two or more different ligands are present7. Way back in 1971 it was reported by Hallman et.al8 that at the physiological pH of 7.4 about 98% of copper (II) in the simulated plasma solution is present as a mixed complex of copper-histidine-cysteine, while ...
Experiment 1. Formation of silver thiosulphate complex
... Werner proposed that the forth ion consists of a central cobalt ion surrounded by six ammonia molecules. These ammonia molecules are ligands that are bonded to the central metal ion by coordinate covalent bonds. The formula for this complex ion is Co(NH3)63+. The central metal ion with its attached ...
... Werner proposed that the forth ion consists of a central cobalt ion surrounded by six ammonia molecules. These ammonia molecules are ligands that are bonded to the central metal ion by coordinate covalent bonds. The formula for this complex ion is Co(NH3)63+. The central metal ion with its attached ...
5 - ChemWeb (UCC)
... Most transition metal compounds contain covalent bonds between ligands and central metal atom. Some are simple polyatomic ions, e.g. permanganate [MnO4]-, a polar covalent compound. Electrons - shared between central Mn and O ligands. However most are "complex compounds" or “complexes”. These contai ...
... Most transition metal compounds contain covalent bonds between ligands and central metal atom. Some are simple polyatomic ions, e.g. permanganate [MnO4]-, a polar covalent compound. Electrons - shared between central Mn and O ligands. However most are "complex compounds" or “complexes”. These contai ...
transition metal complexes
... For Sc, Ti, V, Cr, Mn: highest oxidation states common Highest oxidation states = number of valence (4s + 3d) electrons. Sc [Ar]3d14s2 Mn [Ar]3d54s2 ...
... For Sc, Ti, V, Cr, Mn: highest oxidation states common Highest oxidation states = number of valence (4s + 3d) electrons. Sc [Ar]3d14s2 Mn [Ar]3d54s2 ...
Document
... bonded to number of ligands. Complex contains central metal ion bonded to one or more molecules or anions Lewis acid = metal = center of coordination ...
... bonded to number of ligands. Complex contains central metal ion bonded to one or more molecules or anions Lewis acid = metal = center of coordination ...
Complex Ion Formation
... REVIEW: A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei. In the formation of a simple covalent bond, each atom supplies one electron to the bond. But that doesn't have to be the case! A co-or ...
... REVIEW: A covalent bond is formed by two atoms sharing a pair of electrons. The atoms are held together because the electron pair is attracted by both of the nuclei. In the formation of a simple covalent bond, each atom supplies one electron to the bond. But that doesn't have to be the case! A co-or ...
Document
... 21.1 Rates of ligand substitution 21.2 The classification of mechanisms Ligand substitution in square planar complexes 21.3 The shape of the transition state Ligand substitution in octahedral complexes 21.5 Rate law and their interpretation 21.6 The activation of octahedral complexes ...
... 21.1 Rates of ligand substitution 21.2 The classification of mechanisms Ligand substitution in square planar complexes 21.3 The shape of the transition state Ligand substitution in octahedral complexes 21.5 Rate law and their interpretation 21.6 The activation of octahedral complexes ...
IOSR Journal of Applied Chemistry (IOSR-JAC)
... The 1H NMR spectrum of the ligand GAAP, L shows three singlets[7] at δ ( 2.7 -2.98) ppm, δ (3.053.39) ppm and δ (3.83 - 3.91) ppm corresponds to methyl protons of >C-CH3, >N-CH3 and –OCH3 respectively. The signal due to five aromatic protons of the antipyrine phenyl ring appear as multiplet between ...
... The 1H NMR spectrum of the ligand GAAP, L shows three singlets[7] at δ ( 2.7 -2.98) ppm, δ (3.053.39) ppm and δ (3.83 - 3.91) ppm corresponds to methyl protons of >C-CH3, >N-CH3 and –OCH3 respectively. The signal due to five aromatic protons of the antipyrine phenyl ring appear as multiplet between ...
Potintiometric and Thermodynamic Studies of Some Divalent Metal
... (Table 2), in 75 % (v/v) different mixed solvents (ethanol- water, acetone–water and isopropanol–water) is as follows; acetone-water > isopropanol-water > ethanol-water in contrast with the order of the dielectric constants of the organic solvents which is, ethanol > acetone > isopropanol. Gregely26 ...
... (Table 2), in 75 % (v/v) different mixed solvents (ethanol- water, acetone–water and isopropanol–water) is as follows; acetone-water > isopropanol-water > ethanol-water in contrast with the order of the dielectric constants of the organic solvents which is, ethanol > acetone > isopropanol. Gregely26 ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.