VSEPR theory From Wikipedia, the free encyclopedia Valence shell
... The ammonia molecule (NH3) has three pairs of electrons involved in bonding, but there is a lone pair of electrons on the nitrogen atom. It is not bonded with another atom; however, it influences the overall shape through repulsions. As in methane above, there are four regions of electron density. ...
... The ammonia molecule (NH3) has three pairs of electrons involved in bonding, but there is a lone pair of electrons on the nitrogen atom. It is not bonded with another atom; however, it influences the overall shape through repulsions. As in methane above, there are four regions of electron density. ...
IOSR Journal of Applied Chemistry (IOSRJAC)
... A large number of transition metal complexes with various aliphatic, aromatic and heterocyclic dithiocarbamate ligands have been reported1. Dithiocarbamates are an important class of ligands, being capable of stabilising transition metals in a wide range of oxidation states. In by far the vast major ...
... A large number of transition metal complexes with various aliphatic, aromatic and heterocyclic dithiocarbamate ligands have been reported1. Dithiocarbamates are an important class of ligands, being capable of stabilising transition metals in a wide range of oxidation states. In by far the vast major ...
CHEM 481. Transition Metal Complexes. Assignment 7. Answers
... A transition element is an element from the d block of the periodic table; more precisely, the characteristic behavior of such elements belongs to those species with d n configurations from d 1 to d 9 in at least one common oxidation state. Thus Sc3+ and Zn2+ do not behave like typical TM, but Ti3+, ...
... A transition element is an element from the d block of the periodic table; more precisely, the characteristic behavior of such elements belongs to those species with d n configurations from d 1 to d 9 in at least one common oxidation state. Thus Sc3+ and Zn2+ do not behave like typical TM, but Ti3+, ...
Electronic Structures of Oxo
... Abstract The dianionic oxo ligand occupies a very special place in coordination chemistry, owing to its ability to donate p electrons to stabilize high oxidation states of metals. The ligand field theory of multiple bonding in oxo-metal ions, which was formulated in Copenhagen 50 years ago, predicts ...
... Abstract The dianionic oxo ligand occupies a very special place in coordination chemistry, owing to its ability to donate p electrons to stabilize high oxidation states of metals. The ligand field theory of multiple bonding in oxo-metal ions, which was formulated in Copenhagen 50 years ago, predicts ...
Chapter 1 - DORAS
... lone pair of electrons on the nitrogen forms a coordination bond with the central ion. This results in a lowering of the energy of the n electron pair which can no longer be excited by low energy light, as seen on the right hand side of figure 1.4. On the other ...
... lone pair of electrons on the nitrogen forms a coordination bond with the central ion. This results in a lowering of the energy of the n electron pair which can no longer be excited by low energy light, as seen on the right hand side of figure 1.4. On the other ...
Synthesis and thermal decarbonylation of W(CO)5 complexes
... CO2Prn) groups (see Scheme 1). The reaction of the polymeric ligands 1, 2 and 3 with the corresponding stoichiometric or substoichiometric amounts of [W(MeOH)(CO)5] in a mixture of dichloromethane–methanol gave the complexes (Scheme 2) {[NP(O2C12H8)]0.5[NP(O–C6H4–CO2Prn)(O–C6H4–CN)(W(CO)5)0.4]0.5}n ...
... CO2Prn) groups (see Scheme 1). The reaction of the polymeric ligands 1, 2 and 3 with the corresponding stoichiometric or substoichiometric amounts of [W(MeOH)(CO)5] in a mixture of dichloromethane–methanol gave the complexes (Scheme 2) {[NP(O2C12H8)]0.5[NP(O–C6H4–CO2Prn)(O–C6H4–CN)(W(CO)5)0.4]0.5}n ...
Slide 1
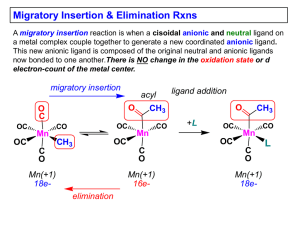
... 1) No change in formal oxidation state (exception: alkylidenes) 2) The two groups that react must be cisoidal to one another 3) A vacant coordination site is generated by the migratory insertion. Therefore, a vacant site is required for the back elimination reaction (e.g., b-hydride elimination). A ...
... 1) No change in formal oxidation state (exception: alkylidenes) 2) The two groups that react must be cisoidal to one another 3) A vacant coordination site is generated by the migratory insertion. Therefore, a vacant site is required for the back elimination reaction (e.g., b-hydride elimination). A ...
Document
... " They require empirical corrections in order to give satisfactory agreement with experimental results (e.g., electronic spectra). " Empirically corrected CFT is known as modified crystal field theory, or more commonly ligand field theory (LFT). ! The need for corrections to CFT arises from metal-li ...
... " They require empirical corrections in order to give satisfactory agreement with experimental results (e.g., electronic spectra). " Empirically corrected CFT is known as modified crystal field theory, or more commonly ligand field theory (LFT). ! The need for corrections to CFT arises from metal-li ...
Solutions - UCI Chemistry
... The reference provides a much more detailed analysis, including a discussion of the paramagnetism of this complex and other factors related to its electronic spectrum. ...
... The reference provides a much more detailed analysis, including a discussion of the paramagnetism of this complex and other factors related to its electronic spectrum. ...
answers
... series of platinum complexes that contained ammonia and chloride ions. One of these had the empirical formula PtCl4.4NH3 and when reacted with silver nitrate released two chloride ions per formula unit. Write the structural formula of this compound and write the name of this compound. Ag+ will react ...
... series of platinum complexes that contained ammonia and chloride ions. One of these had the empirical formula PtCl4.4NH3 and when reacted with silver nitrate released two chloride ions per formula unit. Write the structural formula of this compound and write the name of this compound. Ag+ will react ...
Chapter 27, Nickel, Palladium and Platinum
... Trans Effect •“Trans effect” – Kinetic - Strong π-acceptor character drains electron density from the metal and from the trans-ligand’s π orbitals thus encouraging nucleophilic attack. •“Trans influence” – Thermodynamic – Strong σ-donor ligands produce an axial polarization of the metal and ligand l ...
... Trans Effect •“Trans effect” – Kinetic - Strong π-acceptor character drains electron density from the metal and from the trans-ligand’s π orbitals thus encouraging nucleophilic attack. •“Trans influence” – Thermodynamic – Strong σ-donor ligands produce an axial polarization of the metal and ligand l ...
Here
... 1. Molecule – a neutral group of atoms held together by covalent bonds 2. Chemical Formula – indicates the relative numbers of each kind of atom in a chemical compound by using atomic symbols and numerical subscripts 3. Diatomic Molecule – a molecule containing only two atoms 4. Bond Length a. the a ...
... 1. Molecule – a neutral group of atoms held together by covalent bonds 2. Chemical Formula – indicates the relative numbers of each kind of atom in a chemical compound by using atomic symbols and numerical subscripts 3. Diatomic Molecule – a molecule containing only two atoms 4. Bond Length a. the a ...
Inorganic concepts relevant to metal binding, activity
... ligands, L - , approach the positively-charged metal ion, M 2 + , in the gas phase (Fig. 1), the five-fold degenerate d-orbitals become differentiated in the presence of the electrostatic field of the ligands. Orbitals oriented in the direction of the incoming ligands (dz2, dx2_y2) are preferentiall ...
... ligands, L - , approach the positively-charged metal ion, M 2 + , in the gas phase (Fig. 1), the five-fold degenerate d-orbitals become differentiated in the presence of the electrostatic field of the ligands. Orbitals oriented in the direction of the incoming ligands (dz2, dx2_y2) are preferentiall ...
Chemistry of silver(II): a cornucopia of peculiarities
... vacuum. This must lead to substantial mixing of the metal and ligand valence orbitals in chemical compounds, since free anions (if stable in the gas phase!) usually exhibit small electron binding energies. The chemical potentials of cation and anion(s) of course become equal upon formation of a neut ...
... vacuum. This must lead to substantial mixing of the metal and ligand valence orbitals in chemical compounds, since free anions (if stable in the gas phase!) usually exhibit small electron binding energies. The chemical potentials of cation and anion(s) of course become equal upon formation of a neut ...
chemistry of transition elements
... configurations of the M+ ions are disrupted, with considerable loss of exchange energy. The value for Zn is correspondingly low as the ionisation leads to the production of the stable d10 configuration. The trend in the third ionisation enthalpies is not complicated by the 4s orbital factor and show ...
... configurations of the M+ ions are disrupted, with considerable loss of exchange energy. The value for Zn is correspondingly low as the ionisation leads to the production of the stable d10 configuration. The trend in the third ionisation enthalpies is not complicated by the 4s orbital factor and show ...
Practice final
... They observed diffraction patterns from photons confirming that light had wave properties They observed diffraction patterns of electrons confirming that matter had wave properties ...
... They observed diffraction patterns from photons confirming that light had wave properties They observed diffraction patterns of electrons confirming that matter had wave properties ...
211 - Chimica
... ever-increasing attention over recent years due to the interest in the potential properties of these species as new materials. Applications can be envisaged in fields such as molecular recognition, host–guest chemistry, ion exchange, catalysis, electrical conductivity, magnetism and optics.1d,3 Ligan ...
... ever-increasing attention over recent years due to the interest in the potential properties of these species as new materials. Applications can be envisaged in fields such as molecular recognition, host–guest chemistry, ion exchange, catalysis, electrical conductivity, magnetism and optics.1d,3 Ligan ...
Final Exam S06
... geometric structures, i.e. molecular geometry, of each of these species. In recording your answer, be sure to also provide the name of each molecule and include a well-articulated explanation of the origin of the observed magnetism by showing a d orbital splitting diagram for each of these species. ...
... geometric structures, i.e. molecular geometry, of each of these species. In recording your answer, be sure to also provide the name of each molecule and include a well-articulated explanation of the origin of the observed magnetism by showing a d orbital splitting diagram for each of these species. ...
Lecture 7 - Crystal Field Theory for Octahedral Complexes
... Boats and propellers: If you have a single engine, inboard installation, the stern will pull to port (left) when you go into reverse, if you have a right–handed propeller. A left-handed propeller will pull the stern to starboard (right) when in reverse. (Note: You can tell whether your propeller is ...
... Boats and propellers: If you have a single engine, inboard installation, the stern will pull to port (left) when you go into reverse, if you have a right–handed propeller. A left-handed propeller will pull the stern to starboard (right) when in reverse. (Note: You can tell whether your propeller is ...
MO description of σ only bonding in an O transition metal complex
... electron density from the metal centre via πbackbonding with the metal t2g orbitals. The CO HOMO orbital is a bonding orbital of σ symmetry with significant electron density on the carbon. This orbital forms a σ bond with metal p and eg orbitals. This is a filled orbital and donates electron density ...
... electron density from the metal centre via πbackbonding with the metal t2g orbitals. The CO HOMO orbital is a bonding orbital of σ symmetry with significant electron density on the carbon. This orbital forms a σ bond with metal p and eg orbitals. This is a filled orbital and donates electron density ...
21 - Department of Chemistry | Oregon State University
... A Lewis base donates a pair of electrons to make a new bond. A Lewis acid accepts a pair of electrons to make a new bond. We previously identified ammonia as a base because of its ability to accept a proton: :NH3 (aq) + H2O (l) H:NH3+ (aq) + OH- (aq) Ammonia is a Lewis base because it donates a pa ...
... A Lewis base donates a pair of electrons to make a new bond. A Lewis acid accepts a pair of electrons to make a new bond. We previously identified ammonia as a base because of its ability to accept a proton: :NH3 (aq) + H2O (l) H:NH3+ (aq) + OH- (aq) Ammonia is a Lewis base because it donates a pa ...
Valence bond theory (VBT)
... Predicted theoretical form spatial pyramid of four aspects (sp3) due to the fullness of the cover (d) with elictrons (d9), but proved to X-ray, this overlay is a four-level which makes it imperative to have a hybridization of the type (dsp2) and a solution to this problem suggested Pauling order e N ...
... Predicted theoretical form spatial pyramid of four aspects (sp3) due to the fullness of the cover (d) with elictrons (d9), but proved to X-ray, this overlay is a four-level which makes it imperative to have a hybridization of the type (dsp2) and a solution to this problem suggested Pauling order e N ...
Ligand
4-3D-balls.png?width=300)
In coordination chemistry, a ligand (/lɪɡənd/) is an ion or molecule (functional group) that binds to a central metal atom to form a coordination complex. The bonding between metal and ligand generally involves formal donation of one or more of the ligand's electron pairs. The nature of metal-ligand bonding can range from covalent to ionic. Furthermore, the metal-ligand bond order can range from one to three. Ligands are viewed as Lewis bases, although rare cases are known to involve Lewis acidic ""ligand.""Metals and metalloids are bound to ligands in virtually all circumstances, although gaseous ""naked"" metal ions can be generated in high vacuum. Ligands in a complex dictate the reactivity of the central atom, including ligand substitution rates, the reactivity of the ligands themselves, and redox. Ligand selection is a critical consideration in many practical areas, including bioinorganic and medicinal chemistry, homogeneous catalysis, and environmental chemistry.Ligands are classified in many ways like : their charge, their size (bulk), the identity of the coordinating atom(s), and the number of electrons donated to the metal (denticity or hapticity). The size of a ligand is indicated by its cone angle.