Protein?
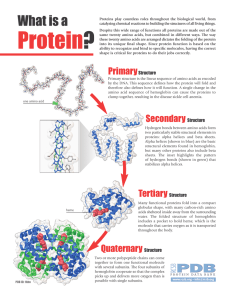
... Alpha helices (shown in blue) are the basic structural elements found in hemoglobin, but many other proteins also include beta sheets. The inset highlights the pattern of hydrogen bonds (shown in green) that stabilizes alpha helices. ...
... Alpha helices (shown in blue) are the basic structural elements found in hemoglobin, but many other proteins also include beta sheets. The inset highlights the pattern of hydrogen bonds (shown in green) that stabilizes alpha helices. ...
Making Proteins
... rRNA = RNA that makes up a ribosome tRNA = RNA that transfers specific amino acids mRNA = carries the DNA message; RNA transcribed from DNA Codon = 3 nucleotides in a row on a strand of mRNA that code for an amino acid Anticodon = 3 nucleotides in tRNA that base pair with the codon Amino Acids = mon ...
... rRNA = RNA that makes up a ribosome tRNA = RNA that transfers specific amino acids mRNA = carries the DNA message; RNA transcribed from DNA Codon = 3 nucleotides in a row on a strand of mRNA that code for an amino acid Anticodon = 3 nucleotides in tRNA that base pair with the codon Amino Acids = mon ...
Abstract
... Modern sequencing technologies provide us with a rich source of data about the evolutionary history of proteins. Inferring a joint probability distribution of amino acid sequences that are members of a protein family, signals from amino acid coevolution at different sequence positions in multiple se ...
... Modern sequencing technologies provide us with a rich source of data about the evolutionary history of proteins. Inferring a joint probability distribution of amino acid sequences that are members of a protein family, signals from amino acid coevolution at different sequence positions in multiple se ...
A dead-end street of protein folding
... force of the structural shift induced global transformation from the original to disease related amyloid fold is expected to lie in the protein backbone, common to all proteins.(Nelson, R. et al. Nature 2005, 435, 773-778., and Wright, C. F. etal. Nature 2005, 438, 878-881.) These structures show lo ...
... force of the structural shift induced global transformation from the original to disease related amyloid fold is expected to lie in the protein backbone, common to all proteins.(Nelson, R. et al. Nature 2005, 435, 773-778., and Wright, C. F. etal. Nature 2005, 438, 878-881.) These structures show lo ...
Powerpoint
... GENE is a section of a DNA molecule that contains the information to code for one complete protein PROTEINS are made up of a chain of amino acids Proteins determine many of the traits in an organism ...
... GENE is a section of a DNA molecule that contains the information to code for one complete protein PROTEINS are made up of a chain of amino acids Proteins determine many of the traits in an organism ...
3-in-1: A novel approach to study membrane protein pharmacology
... mediates crucial functions in the nervous system and has been implicated a numerous diseases. Most LGICs are molecular assemblies of more than one subunit, but conventional methods to study these proteins cannot easily address the contribution of individual subunits within such a protein complex. Re ...
... mediates crucial functions in the nervous system and has been implicated a numerous diseases. Most LGICs are molecular assemblies of more than one subunit, but conventional methods to study these proteins cannot easily address the contribution of individual subunits within such a protein complex. Re ...
CH 107 SI Summer 2015 Worksheet 13 Answers What are the two
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
... 1. What are the two major types of secondary protein structure and what bonds are present in each? α-helices and β-sheets Hydrogen bonds 2. What types of interactions can be present in tertiary protein structure? Rank the interactions from strongest to weakest. disulfide bonds >> salt bridges > hydr ...
Structure and Properties of Proteins
... proteins would bend. When the proteins bend because of the attractions, it’s going to form the B-pleated sheet (functional group) or alpha helix and it’ll depend on what the amino acids are and how they interact. When they interact or when the amino acids get closer together. There will be further i ...
... proteins would bend. When the proteins bend because of the attractions, it’s going to form the B-pleated sheet (functional group) or alpha helix and it’ll depend on what the amino acids are and how they interact. When they interact or when the amino acids get closer together. There will be further i ...
HANDOUT: CH 17 pt 1 Study
... CHAPTER 17 STUDY QUESTIONS, part 1: From Gene to Protein (p. 325-344) 1) Summarize the one gene – one polypeptide hypothesis. ...
... CHAPTER 17 STUDY QUESTIONS, part 1: From Gene to Protein (p. 325-344) 1) Summarize the one gene – one polypeptide hypothesis. ...
1.Contrast and compare the structure of a saturated fat versus an
... 1. Contrast and compare the structure of a saturated fat versus an unsaturated fat. 2. Identify and describe the four levels of protein structure. 3. Speculate (predict) on why a change in pH or Na+ concentration could cause a protein to lose its secondary or tertiary structure and denature. 4. Disc ...
... 1. Contrast and compare the structure of a saturated fat versus an unsaturated fat. 2. Identify and describe the four levels of protein structure. 3. Speculate (predict) on why a change in pH or Na+ concentration could cause a protein to lose its secondary or tertiary structure and denature. 4. Disc ...
Quiz on Proteins (2.4) - Peoria Public Schools
... State one example of a fibrous protein and one example of a globular protein. Fibrous protein: ........................................................................................................... Globular protein: ............................................................................... ...
... State one example of a fibrous protein and one example of a globular protein. Fibrous protein: ........................................................................................................... Globular protein: ............................................................................... ...
Proteins and Nucleic Acids Proteins (pp.46-48) Monomer
... Proteins (pp.46-48) Monomer-basic structure Types of proteins and their functions Number of amino acids o what makes them different from one another o what's responsible for giving them their chemical properties Polymers o Bond responsible for linking amino acids together o Levels of Protein ...
... Proteins (pp.46-48) Monomer-basic structure Types of proteins and their functions Number of amino acids o what makes them different from one another o what's responsible for giving them their chemical properties Polymers o Bond responsible for linking amino acids together o Levels of Protein ...
LSm
In molecular biology, LSm proteins are a family of RNA-binding proteins found in virtually every cellular organism. LSm is a contraction of 'like Sm', because the first identified members of the LSm protein family were the Sm proteins. LSm proteins are defined by a characteristic three-dimensional structure and their assembly into rings of six or seven individual LSm protein molecules, and play a large number of various roles in mRNA processing and regulation.The Sm proteins were first discovered as antigens targeted by so-called Anti-Sm antibodies in a patient with a form of Systemic lupus erythematosus (SLE), a debilitating autoimmune disease. They were named Sm proteins in honor of Stephanie Smith, a patient who suffered from SLE. Other proteins with very similar structures were subsequently discovered and named LSm proteins. New members of the LSm protein family continue to be identified and reported.Proteins with similar structures are grouped into a hierarchy of protein families, superfamilies, and folds. The LSm protein structure is an example of a small beta sheet folded into a short barrel. Individual LSm proteins assemble into a six or seven member doughnut ring (more properly termed a torus), which usually binds to a small RNA molecule to form a ribonucleoprotein complex. The LSm torus assists the RNA molecule to assume and maintain its proper three-dimensional structure. Depending on which LSm proteins and RNA molecule are involved, this ribonucleoprotein complex facilitates a wide variety of RNA processing including degradation, editing, splicing, and regulation.Alternate terms for LSm family are LSm fold and Sm-like fold, and alternate capitalization styles such as lsm, LSM, and Lsm are common and equally acceptable.













