CHAPTER 5
... lkenes are hydrocarbons that contain a carbon–carbon double bond. A carbon–carbon double bond is both an important structural unit and an important functional group in organic chemistry. The shape of an organic molecule is influenced by the presence of this bond, and the double bond is the site of m ...
... lkenes are hydrocarbons that contain a carbon–carbon double bond. A carbon–carbon double bond is both an important structural unit and an important functional group in organic chemistry. The shape of an organic molecule is influenced by the presence of this bond, and the double bond is the site of m ...
Comprehensive Organic Reactions in Aqueous Media
... as a solvent can make many chemical processes more economical. Safety. Many organic solvents are flammable, potentially explosive, mutagenic, and/or carcinogenic. Water, on the other hand, is none of these. Synthetic efficiency. In many organic syntheses, it may be possible to eliminate the need for t ...
... as a solvent can make many chemical processes more economical. Safety. Many organic solvents are flammable, potentially explosive, mutagenic, and/or carcinogenic. Water, on the other hand, is none of these. Synthetic efficiency. In many organic syntheses, it may be possible to eliminate the need for t ...
f-Element Disiloxanediolates: Novel Si[minus]O
... the species, however, is their similarity with certain lanthanide metallocenes such as [(C5Me5)2LnN(SiMe3)2] (cf. 2) or [(C5Me5)2LnCl(thf)] (cf. 3).[29] Such organolanthanide complexes are important as catalyst precursors or highly active catalysts in a variety of organic transformations.[37] It can ...
... the species, however, is their similarity with certain lanthanide metallocenes such as [(C5Me5)2LnN(SiMe3)2] (cf. 2) or [(C5Me5)2LnCl(thf)] (cf. 3).[29] Such organolanthanide complexes are important as catalyst precursors or highly active catalysts in a variety of organic transformations.[37] It can ...
Analysis of structure–function relationships in cytochrome c oxidase
... metals with other molecules. In respect to the design of molecular catalysts and their long term stability this protection mechanism is of high importance and should be considered carefully. 4. Functional CcO inspired metal complexes The information gained by spectroscopy on the reaction mechanism o ...
... metals with other molecules. In respect to the design of molecular catalysts and their long term stability this protection mechanism is of high importance and should be considered carefully. 4. Functional CcO inspired metal complexes The information gained by spectroscopy on the reaction mechanism o ...
Investigation of the steric and electronic properties of 3
... more readily hydroaminated than alkenes and unactivated dienes, which is reflected in the number of precatalysts published for the catalysis of each of these respective transformations. At first glance, the greater electron density of the sp hybridized carbon of an alkyne may make it seem to be less ...
... more readily hydroaminated than alkenes and unactivated dienes, which is reflected in the number of precatalysts published for the catalysis of each of these respective transformations. At first glance, the greater electron density of the sp hybridized carbon of an alkyne may make it seem to be less ...
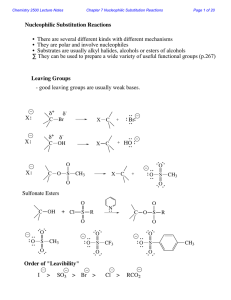
Nucleophilic Substitution Reactions
... With a charged nucleophile (i.e. Nu: = HO or NC ) and a leaving group, LG H ...
... With a charged nucleophile (i.e. Nu: = HO or NC ) and a leaving group, LG H ...
Chapter 1 Chemical Bonding and Chemical Structure
... Separations Using Amine Basicity • Ammonium salts are ionic compounds which imparts a high degree of water-solubility • This property can be useful in separation of amines from other compounds ...
... Separations Using Amine Basicity • Ammonium salts are ionic compounds which imparts a high degree of water-solubility • This property can be useful in separation of amines from other compounds ...
direct synthesis of hydrogen peroxide from oxygen and hydrogen
... Table 20 Calculation of greenness index for a chemical (GIC)................................................... 58 Table 21 Calculation of greenness index for a chemical formula GIF ........................................ 59 Table 22 The greenness index GIO for various oxidants .................... ...
... Table 20 Calculation of greenness index for a chemical (GIC)................................................... 58 Table 21 Calculation of greenness index for a chemical formula GIF ........................................ 59 Table 22 The greenness index GIO for various oxidants .................... ...
Slide 1
... the methyl group. In excess HX, both C-O are cleaved, producing water and two C-X bonds. In phenyl ethers, the benzene ring is not attacked, even in excess HX. ...
... the methyl group. In excess HX, both C-O are cleaved, producing water and two C-X bonds. In phenyl ethers, the benzene ring is not attacked, even in excess HX. ...
Iodomethylzinc_iodid.. - Groupe Charette
... generating iodomethylzinc reagents, which can be categorized into three general classes: type 1, the oxidative addition of a dihalomethane to zinc metal, as typified by the original Simmons– Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and ...
... generating iodomethylzinc reagents, which can be categorized into three general classes: type 1, the oxidative addition of a dihalomethane to zinc metal, as typified by the original Simmons– Smith procedure;7,8 type 2, the reaction of a zinc(II) salt with a diazoalkane, first reported by Wittig and ...
Electrocatalysts Prepared by Galvanic Replacement
... βMnoble , βM the Tafel slopes of the same reactions, Ee noble , EeM their equilibrium potentials and β1 = βMnoble /(βMnoble + βM ), β2 = βM /(βMnoble + βM ). The equilibrium potentials can be estimated based on their standard values and the experimental conditions (temperature, metal ion concentrati ...
... βMnoble , βM the Tafel slopes of the same reactions, Ee noble , EeM their equilibrium potentials and β1 = βMnoble /(βMnoble + βM ), β2 = βM /(βMnoble + βM ). The equilibrium potentials can be estimated based on their standard values and the experimental conditions (temperature, metal ion concentrati ...
An efficient acetylation of dextran using in situ activated acetic
... and derivative thermogravimetric (DTG) analysis were realized on a SDT Q600 (USA) thermal analyzer. Thermal decomposition temperatures (td) of the dextran esters were determined from the DTG curves. The td is reported as the onset of significant weight loss from a heated sample. Samples (10 mg) were ...
... and derivative thermogravimetric (DTG) analysis were realized on a SDT Q600 (USA) thermal analyzer. Thermal decomposition temperatures (td) of the dextran esters were determined from the DTG curves. The td is reported as the onset of significant weight loss from a heated sample. Samples (10 mg) were ...
Consecutive Stability Constants in Solution
... ! Two minor variations on the I mechanism are Id (dissociative interchange) and Ia (associative interchange) interchange. ! If breaking the M–X bond is more important, the mechanism is Id. ! If bond formation between ML5X and Y is significant, the mechanism is Ia. ! Difference between Id and Ia is s ...
... ! Two minor variations on the I mechanism are Id (dissociative interchange) and Ia (associative interchange) interchange. ! If breaking the M–X bond is more important, the mechanism is Id. ! If bond formation between ML5X and Y is significant, the mechanism is Ia. ! Difference between Id and Ia is s ...
File
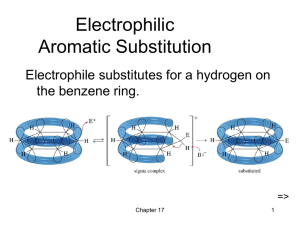
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
... 6. How can you tell the difference between a “cis” and “trans” isomer? In what types of hydrocarbons can they be found? 7. What is an aromatic compound, and what is the simplest aromatic structure? 8. Do aromatic hydrocarbons react more than saturated or unsaturated hydrocarbons? Explain why. 9. Wha ...
Coordination Chemistry and Reactivity of Monomeric Alkoxides and
... related LZnNSi2Me6 compound prepared and characterized by Coates,6 which also contains three coordinate zinc (2+) ions, the M-N distances differ by 0.044 Å to the amide nitrogen with the Zn-NiPr2 having the shorter distance. LZn(OtBu), 4. An ORTEP drawing of this threecoordinate zinc(II) complex is ...
... related LZnNSi2Me6 compound prepared and characterized by Coates,6 which also contains three coordinate zinc (2+) ions, the M-N distances differ by 0.044 Å to the amide nitrogen with the Zn-NiPr2 having the shorter distance. LZn(OtBu), 4. An ORTEP drawing of this threecoordinate zinc(II) complex is ...
Ring-closing metathesis

Ring-closing metathesis, or RCM, is a widely used variation of olefin metathesis in organic chemistry for the synthesis of various unsaturated rings via the intramolecular metathesis of two terminal alkenes, which forms the cycloalkene as the E- or Z- isomers and volatile ethylene.The most commonly synthesized ring sizes are between 5-7 atoms; however, reported syntheses include 45- up to 90- membered macroheterocycles. These reactions are metal-catalyzed and proceed through a metallacyclobutane intermediate. It was first published by Dider Villemin in 1980 describing the synthesis of an Exaltolide precursor, and later become popularized by Robert H. Grubbs and Richard R. Schrock, who shared the Nobel Prize in Chemistry, along with Yves Chauvin, in 2005 for their combined work in olefin metathesis. RCM is a favorite among organic chemists due to its synthetic utility in the formation of rings, which were previously difficult to access efficiently, and broad substrate scope. Since the only major by-product is ethylene, these reactions may also be considered atom economic, an increasingly important concern in the development of green chemistry.There are several reviews published on ring-closing metathesis.




![f-Element Disiloxanediolates: Novel Si[minus]O](http://s1.studyres.com/store/data/014304880_1-eb67bfb0c3fc05ae875f0048d30df664-300x300.png)