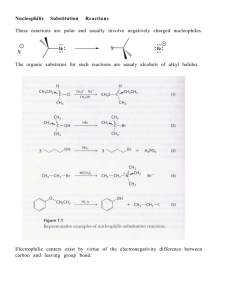
Chapter Seven - U of L Class Index
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
... H Carbocation rearrangments are often promoted by the presence of Lewis Acids. In this case, the intermediates are said to be “carbocation-like” if not carbocations. ...
Organic Chemistry - UCR Chemistry
... transfers a proton (H+) to a solvent water molecule. While we show HBr as a product in the overall transformation (reaction (6)), HBr actually exists in water as H3 O+ and Br- that we see are products of reactions (7) and (9). Solvent Stabilizes the Intermediate Ions. The carbocation formed by ioniz ...
... transfers a proton (H+) to a solvent water molecule. While we show HBr as a product in the overall transformation (reaction (6)), HBr actually exists in water as H3 O+ and Br- that we see are products of reactions (7) and (9). Solvent Stabilizes the Intermediate Ions. The carbocation formed by ioniz ...
Heterogeneous Catalysis and Solid Catalysts
... In photocatalysis light is absorbed by the catalyst or a reactant during the reaction. This can take place in a homogeneous or heterogeneous system. One example is the utilization of semiconductor catalysts (titanium, zinc, and iron oxides) for photochemical degradation of organic substances, e.g., ...
... In photocatalysis light is absorbed by the catalyst or a reactant during the reaction. This can take place in a homogeneous or heterogeneous system. One example is the utilization of semiconductor catalysts (titanium, zinc, and iron oxides) for photochemical degradation of organic substances, e.g., ...
Improved Synthesis, Separation, Transition Metal Coordination and
... three reactions of 1-hexene, 1-octene, and a mixture of 1- hexene/1octene and Ni2Cl4(meso-et,ph-P4) in a H2O/acetone solvent mixture (70°C) .............................................................................................. 26 ...
... three reactions of 1-hexene, 1-octene, and a mixture of 1- hexene/1octene and Ni2Cl4(meso-et,ph-P4) in a H2O/acetone solvent mixture (70°C) .............................................................................................. 26 ...
Handout 3
... CH3 CH3 OH OH CH3 This is due to 1,2-Methyl shift that is similar to the hydride shift. In 1,2-Methyl shift a methyl group could move to an adjacent carbon atom to increase the stability of intermediate. CH3 CH3 H CH3C CHCH3 carbocation intermediate CH3CCH CH2 o + ve charge on (2 ) carbon atom CH3 C ...
... CH3 CH3 OH OH CH3 This is due to 1,2-Methyl shift that is similar to the hydride shift. In 1,2-Methyl shift a methyl group could move to an adjacent carbon atom to increase the stability of intermediate. CH3 CH3 H CH3C CHCH3 carbocation intermediate CH3CCH CH2 o + ve charge on (2 ) carbon atom CH3 C ...
nucleophilic addition on ketones and ketimines - ISI
... by the groups of Shibasaki22 and Yamamoto23 using soft transition metal–chiral diphosphine complexes as catalysts (Scheme 9). Shibasaki et al. used allyl boronates as nucleophiles for the allyl transfer on aliphatic and aromatic ketones with fair to high enantioselectivities when a chiral copper com ...
... by the groups of Shibasaki22 and Yamamoto23 using soft transition metal–chiral diphosphine complexes as catalysts (Scheme 9). Shibasaki et al. used allyl boronates as nucleophiles for the allyl transfer on aliphatic and aromatic ketones with fair to high enantioselectivities when a chiral copper com ...
Cracking (chemistry)

In petroleum geology and chemistry, cracking is the process whereby complex organic molecules such as kerogens or heavy hydrocarbons are broken down into simpler molecules such as light hydrocarbons, by the breaking of carbon-carbon bonds in the precursors. The rate of cracking and the end products are strongly dependent on the temperature and presence of catalysts. Cracking is the breakdown of a large alkane into smaller, more useful alkanes and alkenes. Simply put, hydrocarbon cracking is the process of breaking a long-chain of hydrocarbons into short ones. More loosely, outside the field of petroleum chemistry, the term ""cracking"" is used to describe any type of splitting of molecules under the influence of heat, catalysts and solvents, such as in processes of destructive distillation or pyrolysis. Fluid catalytic cracking produces a high yield of petrol and LPG, while hydrocracking is a major source of jet fuel, Diesel fuel, naphtha, and again yields LPG.