98 pts
... Dehydration: It occurs via E1 mechanism. A carbocation is formed as an intermediate and it generates the alkene products according to the Zaitsev Rule, yielding predominantly the most substituted, 1-methylcyclohexene. CH3 ...
... Dehydration: It occurs via E1 mechanism. A carbocation is formed as an intermediate and it generates the alkene products according to the Zaitsev Rule, yielding predominantly the most substituted, 1-methylcyclohexene. CH3 ...
General Sciences Sample First Exercise Propanoic Acid Solution
... Isoamyl acetate (isopentyl acetate) is an ester that has a strong odor (similar to juicy fruit) which is also described as similar to both banana and pear. It is released by a honey bee's sting apparatus where it serves as a pheromone beacon to attract other bees and provoke them to sting. Isoamyl a ...
... Isoamyl acetate (isopentyl acetate) is an ester that has a strong odor (similar to juicy fruit) which is also described as similar to both banana and pear. It is released by a honey bee's sting apparatus where it serves as a pheromone beacon to attract other bees and provoke them to sting. Isoamyl a ...
The aim of this exercise is to study the acid... prepare a buffer solution. General Sciences Sample
... Isoamyl acetate (isopentyl acetate) is an ester that has a strong odor (similar to juicy fruit) which is also described as similar to both banana and pear. It is released by a honey bee's sting apparatus where it serves as a pheromone beacon to attract other bees and provoke them to sting. Isoamyl a ...
... Isoamyl acetate (isopentyl acetate) is an ester that has a strong odor (similar to juicy fruit) which is also described as similar to both banana and pear. It is released by a honey bee's sting apparatus where it serves as a pheromone beacon to attract other bees and provoke them to sting. Isoamyl a ...
Reactions involving HCl and their Evaporation
... has to be removed post reaction. The paper further discusses evaporation solutions to the problems which routine use of concentrated HCl can create. Deprotection using Hydrochloric Acid Di-tert-butyl dicarbonate is a widely used reagent in organic synthesis. The carbonate ester group in this reagent ...
... has to be removed post reaction. The paper further discusses evaporation solutions to the problems which routine use of concentrated HCl can create. Deprotection using Hydrochloric Acid Di-tert-butyl dicarbonate is a widely used reagent in organic synthesis. The carbonate ester group in this reagent ...
Chapter 9
... equilibrium will react to counteract any disturbance to the equilibrium. • One consequence of this is that removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alkene, which usually has a lower boiling point than the starti ...
... equilibrium will react to counteract any disturbance to the equilibrium. • One consequence of this is that removing a product from a reaction mixture as it is formed drives the equilibrium to the right, forming more product. • Thus, the alkene, which usually has a lower boiling point than the starti ...
(Z)-Tamoxifen and Tetrasubstituted Alkenes and Dienes via a Regio
... Skipped dienes were generated with allyl substituents (entries 3 and 4). In these cases the allyl functionality may be introduced as either the magnesium or palladium component. Dienes were prepared efficiently as demonstrated by entries 5, 6, and 7. In theses examples also, depending upon the synth ...
... Skipped dienes were generated with allyl substituents (entries 3 and 4). In these cases the allyl functionality may be introduced as either the magnesium or palladium component. Dienes were prepared efficiently as demonstrated by entries 5, 6, and 7. In theses examples also, depending upon the synth ...
Redox Biocatalysis. Fundamentals and Applications Brochure
... high–throughput screening, and medium engineering - Case studies tracing the development of industrial applications using biocatalytic redox reactions Each chapter ends with concluding remarks, underscoring the key scientific principles and processes. Extensive references serve as a gateway to the g ...
... high–throughput screening, and medium engineering - Case studies tracing the development of industrial applications using biocatalytic redox reactions Each chapter ends with concluding remarks, underscoring the key scientific principles and processes. Extensive references serve as a gateway to the g ...
Exam 2 Review A
... using an arrow-pushing mechanism to explain how the reaction works. a. Be able to explain the differences between this reaction and the reverse process, i.e., the acid-catalyzed dehydration of alcohols to form alkenes. 4. Be able to explain how alkenes can be hydrated in a Markovnikov fashion using ...
... using an arrow-pushing mechanism to explain how the reaction works. a. Be able to explain the differences between this reaction and the reverse process, i.e., the acid-catalyzed dehydration of alcohols to form alkenes. 4. Be able to explain how alkenes can be hydrated in a Markovnikov fashion using ...
Slide 1
... • Alkanes are hydrocarbons with only single bonds. – A straight-chain alkane has a long chain of carbons bonded to each other with hydrogen atoms around the outside. • Example: propane has three carbon atoms and 8 ...
... • Alkanes are hydrocarbons with only single bonds. – A straight-chain alkane has a long chain of carbons bonded to each other with hydrogen atoms around the outside. • Example: propane has three carbon atoms and 8 ...
Drawing Organic Structures Functional Groups Constitutional Isomers
... • Mixture of products from both SN1 vs. E1 will result anytime a unimolecular mechanism is predicted ...
... • Mixture of products from both SN1 vs. E1 will result anytime a unimolecular mechanism is predicted ...
Chapter 8 Lecture
... 2. How does the rate-determining step of the mechanism respond to the properties of the solvent? ...
... 2. How does the rate-determining step of the mechanism respond to the properties of the solvent? ...
Hydrocarbon ions in fuel-rich, CH4-C2H2-0, flames
... acetylene. Different types of ion-molecule reactions, both positive and negative, which can contribute through chemical ionization (CI) processes are summarized including their dependence on temperature, pressure, and equivalence ratio 4. Extensive data were presented previously involving ion concen ...
... acetylene. Different types of ion-molecule reactions, both positive and negative, which can contribute through chemical ionization (CI) processes are summarized including their dependence on temperature, pressure, and equivalence ratio 4. Extensive data were presented previously involving ion concen ...
Overview of the Reactions of Carbonyl Compounds
... the tetrahedral intermediate can either be protonated to form an alcohol (NaBH4, LiAlH4, or Grignard Reduction) or a non-bonded e- pair on the nucleophile can be used to form a second bond to the carbonyl carbon. The new bond formation causes expulsion of the carbonyl oxygen as H2O. ...
... the tetrahedral intermediate can either be protonated to form an alcohol (NaBH4, LiAlH4, or Grignard Reduction) or a non-bonded e- pair on the nucleophile can be used to form a second bond to the carbonyl carbon. The new bond formation causes expulsion of the carbonyl oxygen as H2O. ...
Preface (PDF, 24 Pages, 5.7 MB)
... permission, in this textbook appear on the appropriate page within the text or on p. P-1. Copyright © 2016, 2010, 2006 Pearson Education, Inc. All rights reserved. Manufactured in the United States of America. This publication is protected by Copyright, and p ermission should be obtained from the ...
... permission, in this textbook appear on the appropriate page within the text or on p. P-1. Copyright © 2016, 2010, 2006 Pearson Education, Inc. All rights reserved. Manufactured in the United States of America. This publication is protected by Copyright, and p ermission should be obtained from the ...
Chapter 23: Substituted Hydrocarbons and Their Reactions
... hydrocarbons of similar shape and size. Also, because of polarity and hydrogen bonding, ethanol is completely miscible with water. In fact, once they are mixed, it is difficult to separate water and ethanol completely. Distillation is used to remove ethanol from water, but even after that process is ...
... hydrocarbons of similar shape and size. Also, because of polarity and hydrogen bonding, ethanol is completely miscible with water. In fact, once they are mixed, it is difficult to separate water and ethanol completely. Distillation is used to remove ethanol from water, but even after that process is ...
reactions of alcohols with alkenes over an aluminum
... Figure 1. Mechanism for the acid-catalyzed reaction of 2-methyl pent-2-ene with alcohols (Al-montmorillonite catalyst). methyl t-butyl ether when using a clay catalyst (Bylina et al.. 1980: Adams et al.. 1981b). At this temperature methanol is the only alcohol to form a di-alkyl ether. whereas Balla ...
... Figure 1. Mechanism for the acid-catalyzed reaction of 2-methyl pent-2-ene with alcohols (Al-montmorillonite catalyst). methyl t-butyl ether when using a clay catalyst (Bylina et al.. 1980: Adams et al.. 1981b). At this temperature methanol is the only alcohol to form a di-alkyl ether. whereas Balla ...
REAKSI SENYAWA KOMPLEKS
... thermodynamically unstable in acid and can decompose to the equilibrium mixture on the right but it reacts very slowly (has a very high activation energy) and is therefore called inert or robust. ...
... thermodynamically unstable in acid and can decompose to the equilibrium mixture on the right but it reacts very slowly (has a very high activation energy) and is therefore called inert or robust. ...
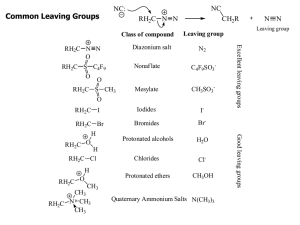
Common Leaving Groups
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
... Whenever substitution reactions are possible, we must also consider whether or not elimination reactions might occur under the same reaction conditions. In elimination reactions, a “neutral” molecule is „eliminated‟ from the substrate to form a π bond. The π bond is formed between the two carbon ato ...
Phosphine-Catalyzed Additions of Nucleophiles and Electrophiles to
... Key characteristics required for successful nucleophilic catalysis lie in the balance of leaving group ability, nucleophilicity, and ease of ylid formation. Increasing leaving group ability can often be correlated with decreasing basicity. Whereas phosphines are less basic than amines (pKa values: H ...
... Key characteristics required for successful nucleophilic catalysis lie in the balance of leaving group ability, nucleophilicity, and ease of ylid formation. Increasing leaving group ability can often be correlated with decreasing basicity. Whereas phosphines are less basic than amines (pKa values: H ...
Chapter_Sixteen_lecture
... Be able to specify where aldehydes and ketones are found & list their major applications Be able to describe and predict the products of the oxidation and reduction of aldehydes and ketones. Be able to recognize hemiacetals and acetals, describe the conditions under which they are formed, and predic ...
... Be able to specify where aldehydes and ketones are found & list their major applications Be able to describe and predict the products of the oxidation and reduction of aldehydes and ketones. Be able to recognize hemiacetals and acetals, describe the conditions under which they are formed, and predic ...
Chem 30CL-Lecture 12.. - UCLA Chemistry and Biochemistry
... Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these groups is present, groups that are ...
... Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these groups is present, groups that are ...
9: Formation of Alkenes and Alkynes. Elimination Reactions
... They also show that the relative amounts of elimination and substitution products vary significantly depending on the structure of the substrate. Nucleophile versus Base. When ethoxide ion displaces a leaving group in a S N2 reaction, we call it a nucleophile. When ethoxide ion removes a β-H in an E ...
... They also show that the relative amounts of elimination and substitution products vary significantly depending on the structure of the substrate. Nucleophile versus Base. When ethoxide ion displaces a leaving group in a S N2 reaction, we call it a nucleophile. When ethoxide ion removes a β-H in an E ...
Enantioselective Synthesis of Cyclic Ethers through a Vanadium
... olefin. Unsaturated a-hydroxyester 7, with two double bonds, provided the possibility of carrying out a double cyclization. Unfortunately, the dioxacyclic compound was obtained as a mixture of two diastereoisomers (44 % yield, d.r. = 1:1; CHCl3, TBHP (2.2 equiv), 40 8C, 48 h), but tandem reaction co ...
... olefin. Unsaturated a-hydroxyester 7, with two double bonds, provided the possibility of carrying out a double cyclization. Unfortunately, the dioxacyclic compound was obtained as a mixture of two diastereoisomers (44 % yield, d.r. = 1:1; CHCl3, TBHP (2.2 equiv), 40 8C, 48 h), but tandem reaction co ...
11_chapter 1
... reactors for the testing of various ammonia catalysts which could be operated at high pressure and temperature were designed by Bosch [9]. The ammonia synthesis was commercialized in 1913 by Badische Anilin-und Soda- Fabrik (BASF) as the Haber – Bosch [9] process. Mittasch [1] at BASF developed and ...
... reactors for the testing of various ammonia catalysts which could be operated at high pressure and temperature were designed by Bosch [9]. The ammonia synthesis was commercialized in 1913 by Badische Anilin-und Soda- Fabrik (BASF) as the Haber – Bosch [9] process. Mittasch [1] at BASF developed and ...
Ene reaction

The ene reaction (also known as the Alder-ene reaction) is a chemical reaction between an alkene with an allylic hydrogen (the ene) and a compound containing a multiple bond (the enophile), in order to form a new σ-bond with migration of the ene double bond and 1,5 hydrogen shift. The product is a substituted alkene with the double bond shifted to the allylic position.This transformation is a group transfer pericyclic reaction, and therefore, usually requires highly activated substrates and/or high temperatures. Nonetheless, the reaction is compatible with a wide variety of functional groups that can be appended to the ene and enophile moieties. Also,many useful Lewis acid-catalyzed ene reactions have been developed which can afford high yields and selectivities at significantly lower temperatures, making the ene reaction a useful C–C forming tool for the synthesis of complex molecules and natural products.