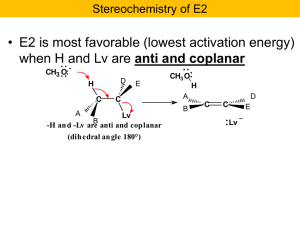
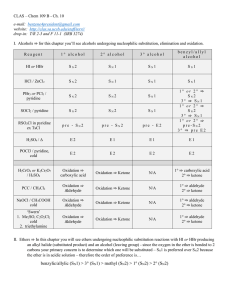
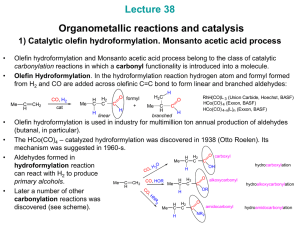
Alcohols
... • Primary: carbon with –OH is bonded to one other carbon. • Secondary: carbon with –OH is bonded to two other carbons. • Tertiary: carbon with –OH is bonded to ...
... • Primary: carbon with –OH is bonded to one other carbon. • Secondary: carbon with –OH is bonded to two other carbons. • Tertiary: carbon with –OH is bonded to ...
ALKANOLS (ALCOHOLS)
... and are neutral when liquid or in aqueous solution as the hydroxyl group does not readily donate or accept protons. Melting and boiling points are much higher than the corresponding alkanes because of the hydrogen bonding present in alcohols. Because hydrogen bonds can form with water and the hydrox ...
... and are neutral when liquid or in aqueous solution as the hydroxyl group does not readily donate or accept protons. Melting and boiling points are much higher than the corresponding alkanes because of the hydrogen bonding present in alcohols. Because hydrogen bonds can form with water and the hydrox ...
Lab 9 - Academic Computer Center
... addition of two H atoms. The first H atom comes from a hydride, H-, of NaBH4. The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the three hydride reagents and is easy to use in the lab, because it is soluble in ...
... addition of two H atoms. The first H atom comes from a hydride, H-, of NaBH4. The second comes from the workup of the reaction, which is normally conducted in aqueous acid. Sodium borohydride, NaBH4, is the mildest of the three hydride reagents and is easy to use in the lab, because it is soluble in ...
Nucleophilic Substitution and b
... Here is the crux of the matter: how can the non-reacting carbon change its configuration??? Further it does not always change but only if configuration of the reacting carbon changes!! We got a mixture of enantiomers, a racemic mixture. Something strange is happening!! Expect sulfur to attack the C- ...
... Here is the crux of the matter: how can the non-reacting carbon change its configuration??? Further it does not always change but only if configuration of the reacting carbon changes!! We got a mixture of enantiomers, a racemic mixture. Something strange is happening!! Expect sulfur to attack the C- ...
m4 phenol and diazo salts
... the OH group is electron releasing it increases the electron density of the delocalised system it makes substitution much easier compared to benzene the electron density is greatest at the 2,4 and 6 positions substitution takes place at the 2,4 and 6 positions ...
... the OH group is electron releasing it increases the electron density of the delocalised system it makes substitution much easier compared to benzene the electron density is greatest at the 2,4 and 6 positions substitution takes place at the 2,4 and 6 positions ...
WADE7Lecture10a
... The longest chain contains six carbon atoms, but it does not contain the carbon bonded to the hydroxyl group. The longest chain containing the carbon bonded to the —OH group is the one outlined by the green box, containing five carbon atoms. This chain is numbered from right to left in order to give ...
... The longest chain contains six carbon atoms, but it does not contain the carbon bonded to the hydroxyl group. The longest chain containing the carbon bonded to the —OH group is the one outlined by the green box, containing five carbon atoms. This chain is numbered from right to left in order to give ...
Chapter 10 - UCSB CLAS
... III. Epoxides ⇒ In this chapter you will see epoxides undergoing nucleophilic substitution (only SN2) reactions aka ring opening reactions - epoxides are ethers with lots of ring strain – the strain allows them to react with a large number of nucleophiles unlike typical ethers which only react with ...
... III. Epoxides ⇒ In this chapter you will see epoxides undergoing nucleophilic substitution (only SN2) reactions aka ring opening reactions - epoxides are ethers with lots of ring strain – the strain allows them to react with a large number of nucleophiles unlike typical ethers which only react with ...
Slide 1
... decomposition. becomes stronger Bulkier PR3 favor greater linear / branched becomes more hydride-like H aldehyde ratios ( up to 8 : 1). CO Finally, more hydride-like Co-H promotes aldehyde OC Co hydrogenation to alcohols: CO ...
... decomposition. becomes stronger Bulkier PR3 favor greater linear / branched becomes more hydride-like H aldehyde ratios ( up to 8 : 1). CO Finally, more hydride-like Co-H promotes aldehyde OC Co hydrogenation to alcohols: CO ...
Chapter 12 –Part 2 Reaction of Carbonyl Compounds with
... è They react readily with hydrogen atoms attached to oxygen, nitro gen or sulfur, or any compound with acidic hydrogens ...
... è They react readily with hydrogen atoms attached to oxygen, nitro gen or sulfur, or any compound with acidic hydrogens ...
A-level Chemistry Question paper Unit 4 - Further Physical
... (c) Propylamine, CH3CH2CH2NH2, can be formed either by nucleophilic substitution or by reduction. (i) Draw the structure of a compound which can undergo nucleophilic substitution to form propylamine. ...
... (c) Propylamine, CH3CH2CH2NH2, can be formed either by nucleophilic substitution or by reduction. (i) Draw the structure of a compound which can undergo nucleophilic substitution to form propylamine. ...
Chemistry in Action: Question paper - A
... 1 Sodium thiosulfate solution (Na2S2O3) reacts slowly with dilute hydrochloric acid to form a precipitate. The rate of this reaction can be studied by measuring the time (t) that it takes for a small fixed amount of precipitate to form under different conditions. The fixed amount of precipitate is t ...
... 1 Sodium thiosulfate solution (Na2S2O3) reacts slowly with dilute hydrochloric acid to form a precipitate. The rate of this reaction can be studied by measuring the time (t) that it takes for a small fixed amount of precipitate to form under different conditions. The fixed amount of precipitate is t ...
ppt
... The sulfur atom of sulfides is much more nucleophilic than the oxygen atom of ethers, and will react with alkyl halides to give stable sulfonium salts. H3C ...
... The sulfur atom of sulfides is much more nucleophilic than the oxygen atom of ethers, and will react with alkyl halides to give stable sulfonium salts. H3C ...
Functional groups and homologous series
... is added to an alkene the product is colourless. This decolorization of bromine solution provides a useful test to indicate the presence of an alkene group. 2. Hydration Ethene is an important product formed during the cracking of oil. Although ethanol can be made from the fermentation of starch and ...
... is added to an alkene the product is colourless. This decolorization of bromine solution provides a useful test to indicate the presence of an alkene group. 2. Hydration Ethene is an important product formed during the cracking of oil. Although ethanol can be made from the fermentation of starch and ...
List of Objectives for Chem52
... 1 Provide IUPAC names for alcohols and phenols. 2 Provide the products of alcohols (and phenols) acting as either acids or bases. 3 Provide products, or missing reagents if given one starting material and the products, for the deprotonation of alcohols/phenols to form alkoxides/phenoxides. (17.2) 4 ...
... 1 Provide IUPAC names for alcohols and phenols. 2 Provide the products of alcohols (and phenols) acting as either acids or bases. 3 Provide products, or missing reagents if given one starting material and the products, for the deprotonation of alcohols/phenols to form alkoxides/phenoxides. (17.2) 4 ...
Experiment #9 – Identification of Aldehydes and Ketones
... Aldehydes and ketones share the carbonyl functional group which features carbon doubly bonded to oxygen. In the case of ketones there are two carbon atoms bonded to the carbonyl carbon and no hydrogens. In the case of aldehydes there is at least one hydrogen bonded to the carbonyl carbon; the other ...
... Aldehydes and ketones share the carbonyl functional group which features carbon doubly bonded to oxygen. In the case of ketones there are two carbon atoms bonded to the carbonyl carbon and no hydrogens. In the case of aldehydes there is at least one hydrogen bonded to the carbonyl carbon; the other ...
Chapter 2: Nomenclature and Structure
... 2. if a tie, parent chain (two or more possible), parent is one that has more substituents (more substituents = easier to name) 3. name and number substituents a. substituent is prefix b. number from side that gives smaller number c. common names almost never have number 4. if more than one substitu ...
... 2. if a tie, parent chain (two or more possible), parent is one that has more substituents (more substituents = easier to name) 3. name and number substituents a. substituent is prefix b. number from side that gives smaller number c. common names almost never have number 4. if more than one substitu ...
16.2: Structure and Bonding in Ethers and Epoxides
... The sulfur atom of sulfides is much more nucleophilic than the oxygen atom of ethers, and will react with alkyl halides to give stable sulfonium salts. ...
... The sulfur atom of sulfides is much more nucleophilic than the oxygen atom of ethers, and will react with alkyl halides to give stable sulfonium salts. ...
Chapter 18
... Therefore an aldehyde or ketone can be determined by comparing the melting points ...
... Therefore an aldehyde or ketone can be determined by comparing the melting points ...
Microwave-Enhanced Sulphated Zirconia and SZ/MCM
... (initial power 50 W) for 10 min in a self tuning single mode CEM Labmate® microwave synthesizer. After completion of the reaction, the mixture was cooled rapidly to room temperature, passing compressed nitrogen through the microwave cavity for 5 min. The catalyst was then recovered by filtration and ...
... (initial power 50 W) for 10 min in a self tuning single mode CEM Labmate® microwave synthesizer. After completion of the reaction, the mixture was cooled rapidly to room temperature, passing compressed nitrogen through the microwave cavity for 5 min. The catalyst was then recovered by filtration and ...
Answers - Benjamin
... The peak at 1.0 is due to –C(CH3)3, that at 2.5 to –CH2– adjacent to the nitrogen and that at 4.5 to CH2– adjacent to the –OH group. (The other absorptions at 9.0, 4.9, 4.4 and 3.3 are due to the various –OH groups and the –NH group being much more difficult to assign as there will be exchanges occu ...
... The peak at 1.0 is due to –C(CH3)3, that at 2.5 to –CH2– adjacent to the nitrogen and that at 4.5 to CH2– adjacent to the –OH group. (The other absorptions at 9.0, 4.9, 4.4 and 3.3 are due to the various –OH groups and the –NH group being much more difficult to assign as there will be exchanges occu ...
ORGANIC CHEMISTRY - Alex Science Department
... Examples: methane reacting with chlorine ethane reacting with bromine pentane reacting with flourine ...
... Examples: methane reacting with chlorine ethane reacting with bromine pentane reacting with flourine ...
Identification of Ketones and Aldehydes
... Aldehydes and ketones share the carbonyl functional group which features carbon doubly bonded to oxygen. In the case of ketones there are two carbon atoms bonded to the carbonyl carbon and no hydrogens. In the case of aldehydes there is at least one hydrogen bonded to the carbonyl carbon, the other ...
... Aldehydes and ketones share the carbonyl functional group which features carbon doubly bonded to oxygen. In the case of ketones there are two carbon atoms bonded to the carbonyl carbon and no hydrogens. In the case of aldehydes there is at least one hydrogen bonded to the carbonyl carbon, the other ...
B. Reaction and Isolation of Products
... Background Information on Refractive Index Refractive Index is an important physical constant characteristic of liquids and transparent solids. The refractive index derives from the fact that light travels at a different velocity in condensed phases (liquids and solids) than it does in air. The refr ...
... Background Information on Refractive Index Refractive Index is an important physical constant characteristic of liquids and transparent solids. The refractive index derives from the fact that light travels at a different velocity in condensed phases (liquids and solids) than it does in air. The refr ...
Chapter 7: Alkene reactions
... Example: Compound A has the formula C10H16. On catalytic hydrogenation over palladium (H2, Pd) it reacts with only one molar equivalent of H2. Compound A also undergoes reaction with ozone (O3), followed by zinc treatment (Zn, H3O+) to yield a symmetrical diketone, B which has formula (C10H16O2). Pr ...
... Example: Compound A has the formula C10H16. On catalytic hydrogenation over palladium (H2, Pd) it reacts with only one molar equivalent of H2. Compound A also undergoes reaction with ozone (O3), followed by zinc treatment (Zn, H3O+) to yield a symmetrical diketone, B which has formula (C10H16O2). Pr ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.