Carbohydrates - De Anza College
... Sugar alcohols such as D-sorbitol, D-xylitol from D-xylose, and D-mannitol from D-mannose are used as sweeteners in many sugarfree products such as diet drinks and sugarless gum as well as products for people with diabetes. ...
... Sugar alcohols such as D-sorbitol, D-xylitol from D-xylose, and D-mannitol from D-mannose are used as sweeteners in many sugarfree products such as diet drinks and sugarless gum as well as products for people with diabetes. ...
Chem 30CL-Lecture 12.. - UCLA Chemistry and Biochemistry
... -OH (pKa~16-18), -NHx (pKa~35), -SH (pKa~9-12), -COOH (pKa~3-5) Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these groups is present, groups that are ...
... -OH (pKa~16-18), -NHx (pKa~35), -SH (pKa~9-12), -COOH (pKa~3-5) Some functional groups react with the reagent because they contain electrophilic atoms: -CHO, -COR, -CONR2, -COOR, -C≡N, -NO2, -SO2R, epoxides (ring opening) If more than one of these groups is present, groups that are ...
m3 isomerismintro
... There are two structural isomers of C4H10. One is a straight chain molecule where all the carbon atoms are in a single row. The other is a branched molecule where three carbon atoms are in a row and one carbon atom sticks out of the main chain. ...
... There are two structural isomers of C4H10. One is a straight chain molecule where all the carbon atoms are in a single row. The other is a branched molecule where three carbon atoms are in a row and one carbon atom sticks out of the main chain. ...
Sodium is an abundant metallic element with atomic number as 11
... -Sodium doesn’t react with nitrogen, not even at very high temperatures, but it can react with ammonia to form sodium amide. -Sodium and hydrogen react above 200ºC (390ºF) to form sodium hydride. -Sodium hardly reacts with carbon, but it does react with halogens. It also reacts with various metallic ...
... -Sodium doesn’t react with nitrogen, not even at very high temperatures, but it can react with ammonia to form sodium amide. -Sodium and hydrogen react above 200ºC (390ºF) to form sodium hydride. -Sodium hardly reacts with carbon, but it does react with halogens. It also reacts with various metallic ...
Chemistry 122 Chapter 9 Ketones and Aldehydes
... 1 C: formic acid, formaldehyde 2 C’s: acetic acid, acetaldehyde 3 C’s: propionic acid, propionaldehyde 4 C’s: butyric acid, butyraldehyde. 5 C’s: valeric acid, valeraldehyde Br CH3 γ ...
... 1 C: formic acid, formaldehyde 2 C’s: acetic acid, acetaldehyde 3 C’s: propionic acid, propionaldehyde 4 C’s: butyric acid, butyraldehyde. 5 C’s: valeric acid, valeraldehyde Br CH3 γ ...
Organic Chemistry Fifth Edition
... Halogen and alkyl groups are of equal rank when it comes to numbering the chain. ...
... Halogen and alkyl groups are of equal rank when it comes to numbering the chain. ...
Slides from Chapters 1,2
... Curved arrow notation is a convention that is used to show how electron position differs between the two resonance forms. Curved arrow notation shows the movement of an electron pair. The tail of the arrow always begins at the electron pair, either in a bond or lone pair. The head points to where th ...
... Curved arrow notation is a convention that is used to show how electron position differs between the two resonance forms. Curved arrow notation shows the movement of an electron pair. The tail of the arrow always begins at the electron pair, either in a bond or lone pair. The head points to where th ...
Alkyl Aryl Ether Bond Formation with PhenoFluor
... Abstract: An alkyl aryl ether bond formation reaction between phenols and primary and secondary alcohols with PhenoFluor has been developed. The reaction features a broad substrate scope and tolerates many functional groups, and substrates that are challenging for more conventional ether bond formin ...
... Abstract: An alkyl aryl ether bond formation reaction between phenols and primary and secondary alcohols with PhenoFluor has been developed. The reaction features a broad substrate scope and tolerates many functional groups, and substrates that are challenging for more conventional ether bond formin ...
© John Congleton, Orange Coast College Organic Chemistry 220
... o Where does oxidation stop? • Oxidation of alcohols with pyridinium chlorochromate (PCC) o Know mechanism o Where does oxidation stop? • Don’t worry about the other many methods of alcohol oxidation in section 11-3. • Biological oxidation o Fate of ethanol, methanol, ethylene glycol in the body… o ...
... o Where does oxidation stop? • Oxidation of alcohols with pyridinium chlorochromate (PCC) o Know mechanism o Where does oxidation stop? • Don’t worry about the other many methods of alcohol oxidation in section 11-3. • Biological oxidation o Fate of ethanol, methanol, ethylene glycol in the body… o ...
A Few More Notes on Acidity
... Weak inorganic acids (H2S, HCN, NH4+), amine salts (RNH3+), phenols (ArOH), thiols (RSH), aromatic amides (ArCONH2) H2O, alcohols, thiols (RSH), amides RCONH2 ketones (the alpha proton H-CH2COR) Esters (the alpha proton H-CH2CO2R), alkynes RCCH, nitriles (H-CH2CN) ...
... Weak inorganic acids (H2S, HCN, NH4+), amine salts (RNH3+), phenols (ArOH), thiols (RSH), aromatic amides (ArCONH2) H2O, alcohols, thiols (RSH), amides RCONH2 ketones (the alpha proton H-CH2COR) Esters (the alpha proton H-CH2CO2R), alkynes RCCH, nitriles (H-CH2CN) ...
Aldehydes and Ketones - Belle Vernon Area School District
... IUPAC - # chain from the end closest to the carbonyl, give the carbon #, drop –e add –one Name the groups on either side of the carbonyl and follow with ketone list groups by order of sixe of molar mass (smaller 1st) ...
... IUPAC - # chain from the end closest to the carbonyl, give the carbon #, drop –e add –one Name the groups on either side of the carbonyl and follow with ketone list groups by order of sixe of molar mass (smaller 1st) ...
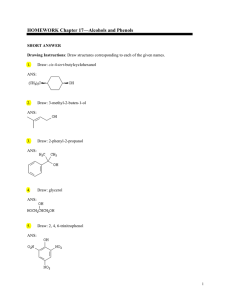
HOMEWORK Chapter 17—Alcohols and Phenols
... In m-nitrophenol, the inductive effect of the electron-withdrawing nitro group helps to stabilize the negative charge on oxygen. However, when the nitro group is para to the oxygen, direct conjugation of the negative charge on oxygen with the nitro group can occur. p-Nitrophenolate ion is, thus, mor ...
... In m-nitrophenol, the inductive effect of the electron-withdrawing nitro group helps to stabilize the negative charge on oxygen. However, when the nitro group is para to the oxygen, direct conjugation of the negative charge on oxygen with the nitro group can occur. p-Nitrophenolate ion is, thus, mor ...
2 Physical Properties of Aldehydes and Ketones GOB Structures
... • Aldehydes and ketones containing 3 to 10 carbon atoms are liquids. • The polar carbonyl group with a partially negative oxygen atom and a partially positive carbon atom has an influence on the boiling points and the solubility of aldehydes and ketones in water. ...
... • Aldehydes and ketones containing 3 to 10 carbon atoms are liquids. • The polar carbonyl group with a partially negative oxygen atom and a partially positive carbon atom has an influence on the boiling points and the solubility of aldehydes and ketones in water. ...
Slide 1 - Alfred State College intranet site
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
... Formaldehyde (CH2═O) is the simplest aldehyde: •It is sold as formalin, a 37% aqueous solution used to preserve biological specimens. ...
Organic Chemistry
... 21.1d Hydrocarbon Reactivity In general, alkenes and alkynes are more reactive than alkanes and aromatic compounds. Here we describe just a few of the more common reactions for these compounds. ...
... 21.1d Hydrocarbon Reactivity In general, alkenes and alkynes are more reactive than alkanes and aromatic compounds. Here we describe just a few of the more common reactions for these compounds. ...
Lipids lecture(1) by Prof.Dr.Moaed Al
... Chemistry of Lipids Lipids constitute a heterogeneous group of compounds of biochemical importance. Lipids may be defined as compounds which are relatively insoluble in water, but freely soluble in nonpolar ...
... Chemistry of Lipids Lipids constitute a heterogeneous group of compounds of biochemical importance. Lipids may be defined as compounds which are relatively insoluble in water, but freely soluble in nonpolar ...
Alcohol

In chemistry, an alcohol is any organic compound in which the hydroxyl functional group (–OH) is bound to a saturated carbon atom. The term alcohol originally referred to the primary alcohol ethyl alcohol (ethanol), the predominant alcohol in alcoholic beverages.The suffix -ol appears in the IUPAC chemical name of all substances where the hydroxyl group is the functional group with the highest priority; in substances where a higher priority group is present the prefix hydroxy- will appear in the IUPAC name. The suffix -ol in non-systematic names (such as paracetamol or cholesterol) also typically indicates that the substance includes a hydroxyl functional group and, so, can be termed an alcohol. But many substances, particularly sugars (examples glucose and sucrose) contain hydroxyl functional groups without using the suffix. An important class of alcohols, of which methanol and ethanol are the simplest members is the saturated straight chain alcohols, the general formula for which is CnH2n+1OH.