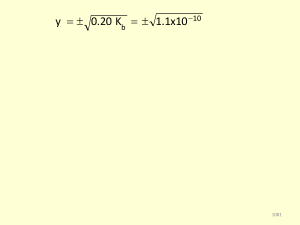
Programme
... photoredox reaction to produce 3-formylbenzhydrol. Extensive investigation into the mechanistic behaviour of 3-(hydroxymethyl)benzophenone produced evidence of a unique solvent mediated, acid catalysed photoreaction. A mechanism has been proposed for the intramolecular photoredox reaction that proce ...
... photoredox reaction to produce 3-formylbenzhydrol. Extensive investigation into the mechanistic behaviour of 3-(hydroxymethyl)benzophenone produced evidence of a unique solvent mediated, acid catalysed photoreaction. A mechanism has been proposed for the intramolecular photoredox reaction that proce ...
PHASE TRANSFER CATALYSIS IN PHARMACEUTICAL
... Organic synthesis is still the main way to produce pharmaceuticals. Among the reactions used for transformations of substrates into final products, especially important are those in which the abstraction of a proton from C, O, N, S, etc. acids, under the action of bases, results in the formation of ...
... Organic synthesis is still the main way to produce pharmaceuticals. Among the reactions used for transformations of substrates into final products, especially important are those in which the abstraction of a proton from C, O, N, S, etc. acids, under the action of bases, results in the formation of ...
5H2O → CuSO4 + 5H2O(g)
... Complete the equation below (with phases) and balance it. K2S(aq) + AgNO3(g) If you combine 10.21 mL of 0.152 M K2S with 1.0092 g AgNO3, how much solid product can be formed? If 0.2744 g of product were actually formed, what is the percent yield? ...
... Complete the equation below (with phases) and balance it. K2S(aq) + AgNO3(g) If you combine 10.21 mL of 0.152 M K2S with 1.0092 g AgNO3, how much solid product can be formed? If 0.2744 g of product were actually formed, what is the percent yield? ...
Introduction
... Electrolytes are ionic and strong acid solutions (e.g., GatoradeTM); Nonelectrolytes are covalent compounds (e.g., sugar); weak electrolytes are in between. ...
... Electrolytes are ionic and strong acid solutions (e.g., GatoradeTM); Nonelectrolytes are covalent compounds (e.g., sugar); weak electrolytes are in between. ...
4. Amines Amides and Amino Acids
... Polyesters can be hydrolysed by acid and alkali With HCl a polyester will be hydrolysed and split up in to the original dicarboxylic acid and diol With NaOH an polyester will be hydrolysed and split up into the diol and dicarboxylic acid salt. Polyamides can be hydrolysed by aqueous acids or alkalis ...
... Polyesters can be hydrolysed by acid and alkali With HCl a polyester will be hydrolysed and split up in to the original dicarboxylic acid and diol With NaOH an polyester will be hydrolysed and split up into the diol and dicarboxylic acid salt. Polyamides can be hydrolysed by aqueous acids or alkalis ...
Chem 2A Final Review
... 60. The oxidation number (oxidation state) of sulfur in the following are? K2SO2, K2S2O3, K2S 61. According to Le Chatelier’s principle what effects will take place on the equilibrium of the following reaction: CO2 + H2 H2O + CO a) Increase [H2] b) Increase [H2O] c) remove H2O and CO ...
... 60. The oxidation number (oxidation state) of sulfur in the following are? K2SO2, K2S2O3, K2S 61. According to Le Chatelier’s principle what effects will take place on the equilibrium of the following reaction: CO2 + H2 H2O + CO a) Increase [H2] b) Increase [H2O] c) remove H2O and CO ...
Carbon Compounds
... Organic compounds, such as alcohols, esters, and others, can be linked together to build huge molecules with thousands or even millions of atoms. A very large molecule made of a chain of many smaller molecules bonded together is called a polymer. The smaller molecules- the links that make up t ...
... Organic compounds, such as alcohols, esters, and others, can be linked together to build huge molecules with thousands or even millions of atoms. A very large molecule made of a chain of many smaller molecules bonded together is called a polymer. The smaller molecules- the links that make up t ...
Nucleophilic Aromatic Substitution, General Corrected Mechanism
... Nucleophilic aromatic substitution in electron-deficient arenas, particularly nitroarenes is an efficient tool in synthesis and manufacturing of pharmaceuticals [1-3]. It is therefore of crucial importance to know how exactly these reactions proceed. For many years it was considered that these react ...
... Nucleophilic aromatic substitution in electron-deficient arenas, particularly nitroarenes is an efficient tool in synthesis and manufacturing of pharmaceuticals [1-3]. It is therefore of crucial importance to know how exactly these reactions proceed. For many years it was considered that these react ...
Carboxylic Acids
... apples, rhubarb, grapes and other fruits with high acidity Carboxylic acids are also produced when an alcohol is oxidized accordingly. This creates the double bond between the oxygen, creating the –COOH group. An example of this is when red wine is oxidized to make red wine vinegar, gaining the more ...
... apples, rhubarb, grapes and other fruits with high acidity Carboxylic acids are also produced when an alcohol is oxidized accordingly. This creates the double bond between the oxygen, creating the –COOH group. An example of this is when red wine is oxidized to make red wine vinegar, gaining the more ...
Document
... Electrochemistry is the study of interchange of chemical and electrical energy. Oxidation/Reduction involves the exchange of electrons from one chemical species to another. Normally, this is done when the two chemicals contact each other in the activated complex (when two species bump into each othe ...
... Electrochemistry is the study of interchange of chemical and electrical energy. Oxidation/Reduction involves the exchange of electrons from one chemical species to another. Normally, this is done when the two chemicals contact each other in the activated complex (when two species bump into each othe ...
JF Physical Chemistry 2010-2011. JF CH 1101: Introduction to
... a. What is the internal energy U and the enthalpy H of a system? Write down an expression for the First Law of Thermodynamics which relates the change in internal energy of a system to the work done on the system and the heat absorbed by the system. Hence derive a relationship between the change in ...
... a. What is the internal energy U and the enthalpy H of a system? Write down an expression for the First Law of Thermodynamics which relates the change in internal energy of a system to the work done on the system and the heat absorbed by the system. Hence derive a relationship between the change in ...
Naming Substituted Hydrocarbons
... element other than hydrogen attached somewhere along the hydrocarbon chain. It is named in a similar fashion to a hydrocarbon. This can be illustrated with alcohols as an example. The compounds pictured to the lower left are alcohols. They look like alkanes with –OH at one end where a H hydrogen wou ...
... element other than hydrogen attached somewhere along the hydrocarbon chain. It is named in a similar fashion to a hydrocarbon. This can be illustrated with alcohols as an example. The compounds pictured to the lower left are alcohols. They look like alkanes with –OH at one end where a H hydrogen wou ...
EXPERIMENT 6: Reactions of Carbonyl Compounds: Qualitative
... steam distillation in the presence of dilute acid. The most commonly used for identification are the 2,4-dinitrophenylhydrazones since simple carbonyl compounds give very colourful, highly crystalline solids. These solid derivatives can also be prepared very rapidly, which makes them an extremely us ...
... steam distillation in the presence of dilute acid. The most commonly used for identification are the 2,4-dinitrophenylhydrazones since simple carbonyl compounds give very colourful, highly crystalline solids. These solid derivatives can also be prepared very rapidly, which makes them an extremely us ...
Insertion of SO2 into the Metal−Carbon Bonds of Rhodium and
... more stable toward disproportionation. Indeed, one way to synthesize this compound is to react 1 equiv of Cp*Ir(PMe3)(CH3)2 with 1 equiv of Cp*Ir(PMe3)(OTf)2.4 When a solution of 4 in CH2Cl2 is put under an atmosphere of SO2 at room temperature, the orange solution immediately turns yellow. The 1H N ...
... more stable toward disproportionation. Indeed, one way to synthesize this compound is to react 1 equiv of Cp*Ir(PMe3)(CH3)2 with 1 equiv of Cp*Ir(PMe3)(OTf)2.4 When a solution of 4 in CH2Cl2 is put under an atmosphere of SO2 at room temperature, the orange solution immediately turns yellow. The 1H N ...
Reaction of Organometallic Reagents with Aldehydes and Ketones.
... • The reaction follows the same two-step process as opening of epoxide rings with other negatively charged nucleophiles—that is, nucleophilic attack from the back side of the epoxide, followed by protonation of the resulting alkoxide. • In unsymmetrical epoxides, nucleophilic attack occurs at the le ...
... • The reaction follows the same two-step process as opening of epoxide rings with other negatively charged nucleophiles—that is, nucleophilic attack from the back side of the epoxide, followed by protonation of the resulting alkoxide. • In unsymmetrical epoxides, nucleophilic attack occurs at the le ...
Chemical Equilibrium Stress? What stress? 1
... Molarity is a measure of how much “stuff” is dissolved in water. ̵ The more stuff dissolved, the more concentrated the solution. ̵ The higher the Molarity. ...
... Molarity is a measure of how much “stuff” is dissolved in water. ̵ The more stuff dissolved, the more concentrated the solution. ̵ The higher the Molarity. ...
Redox Balancing Worksheet
... Oxidation-reduction (redox) reactions are reactions in which oxidation numbers change. Oxidation numbers are either real charges or formal charges which help chemists keep track of electron transfer. In practice, oxidation numbers are best viewed as a bookkeeping device. Oxidation cannot occur witho ...
... Oxidation-reduction (redox) reactions are reactions in which oxidation numbers change. Oxidation numbers are either real charges or formal charges which help chemists keep track of electron transfer. In practice, oxidation numbers are best viewed as a bookkeeping device. Oxidation cannot occur witho ...
main types and mechanisms of the reactions in organic chemistry
... not kept to the full extent. Hydrogen atoms can be substituted at any carbon atoms under the chlorination, but quantity of substitution product at the tertiary carbon will be the greatest. Task №2. Describe the mechanism of oleic acid peroxidation which takes place at radiation sickness as a result ...
... not kept to the full extent. Hydrogen atoms can be substituted at any carbon atoms under the chlorination, but quantity of substitution product at the tertiary carbon will be the greatest. Task №2. Describe the mechanism of oleic acid peroxidation which takes place at radiation sickness as a result ...
Chapter 3 - Stoichiometry
... Balancing word equations: The key here is to understand that the oxidation number of a free element is _____. But certain free elements occur in pairs under normal conditions. They are the 7 diatomics. They are: more problems: 1] potassium chlorate decomposes into potassium chloride and oxygen 2] ph ...
... Balancing word equations: The key here is to understand that the oxidation number of a free element is _____. But certain free elements occur in pairs under normal conditions. They are the 7 diatomics. They are: more problems: 1] potassium chlorate decomposes into potassium chloride and oxygen 2] ph ...
AP Chem – Unit 1 Part 2 AP Chemistry 2016-‐2017 Unit 1
... After completion of unit 1 I will be able to… • Identify an element or determine its purity using mass percent calculations. • Use mole relationships to convert between moles, mass and particles. • ...
... After completion of unit 1 I will be able to… • Identify an element or determine its purity using mass percent calculations. • Use mole relationships to convert between moles, mass and particles. • ...
Ethers and Epoxides
... For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
... For a pure alkylthiol use thiourea (NH2(C=S)NH2) as the nucleophile This gives an intermediate alkylisothiourea salt, which is hydrolyzed cleanly to the alkyl thiourea ...
Strychnine total synthesis

Strychnine total synthesis in chemistry describes the total synthesis of the complex biomolecule strychnine. The first reported method by the group of Robert Burns Woodward in 1954 is considered a classic in this research field. At the time it formed the natural conclusion to an elaborate process of molecular structure elucidation that started with the isolation of strychnine from the beans of Strychnos ignatii by Pierre Joseph Pelletier and Joseph Bienaimé Caventou in 1818. Major contributors to the entire effort were Sir Robert Robinson with over 250 publications and Hermann Leuchs with another 125 papers in a time span of 40 years. Robinson was awarded the Nobel Prize in Chemistry in 1947 for his work on alkaloids, strychnine included. The process of chemical identification was completed with publications in 1946 by Robinson and later confirmed by Woodward in 1947. X-ray structures establishing the absolute configuration became available between 1947 and 1951 with publications from J. M. Bijvoet and J.H. Robertson .Woodward published a very brief account on the strychnine synthesis in 1954 (just 3 pages) and a lengthy one (42 pages) in 1963.Many more methods exist and reported by the research groups of Magnus, Overman, Kuehne, Rawal, Bosch, Vollhardt, Mori, Shibasaki, Li, Fukuyama Vanderwal and MacMillan. Synthetic (+)-strychnine is also known. Racemic synthesises were published by Padwa in 2007 and in 2010 by Andrade and by Reissig.In his 1963 publication Woodward quoted Sir Robert Robinson who said for its molecular size it is the most complex substance known.























