Handbook of Inorganic Chemicals
... Mohs hardness is based on a scale from 1 to 10 units in which diamond, the hardest substance, is given a value of 10 Mohs and talc given a value of 0.5. Vapor pressure is exerted by a solid or liquid in equilibrium with its own vapor. All liquids have vapor pressures. Vapor pressure depends on tempe ...
... Mohs hardness is based on a scale from 1 to 10 units in which diamond, the hardest substance, is given a value of 10 Mohs and talc given a value of 0.5. Vapor pressure is exerted by a solid or liquid in equilibrium with its own vapor. All liquids have vapor pressures. Vapor pressure depends on tempe ...
infrared spectroscopy
... C-O stretches also occur in alcohols and esters, so look for O-H or C=O stretches before concluding a compound is an ether ...
... C-O stretches also occur in alcohols and esters, so look for O-H or C=O stretches before concluding a compound is an ether ...
Elementary Principles of Chemical Processes, 3rd Update Edition
... the studies complement the formal text material. An additional benefit occurs if the assignments are made to groups, an approach we regularly use in our classes. We invariably see the groups starting out in a state of semi-anarchy and then developing cohesiveness as the weeks go by. By the end of the ...
... the studies complement the formal text material. An additional benefit occurs if the assignments are made to groups, an approach we regularly use in our classes. We invariably see the groups starting out in a state of semi-anarchy and then developing cohesiveness as the weeks go by. By the end of the ...
C07C - Cooperative Patent Classification
... C10G 2/00; of synthetic natural gas C10L 3/06) . . from carbon monoxide with hydrogen . . . {Apparatus} . . . . {Reactors} . . . . . {with moving catalysts} . . . . {Temperature controlling devices; Heat exchangers} . . . {Catalysts; their physical properties} . . . . {characterised by the compositi ...
... C10G 2/00; of synthetic natural gas C10L 3/06) . . from carbon monoxide with hydrogen . . . {Apparatus} . . . . {Reactors} . . . . . {with moving catalysts} . . . . {Temperature controlling devices; Heat exchangers} . . . {Catalysts; their physical properties} . . . . {characterised by the compositi ...
Lecture 8
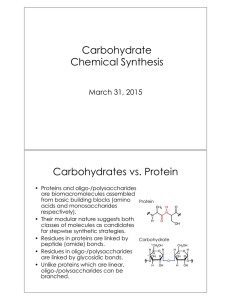
... • While glycals were originally used in the synthesis of 2deoxyglycosides, Danishefsky and coworkers devised a glycal strategy that afforded 2-hydroxyglycosides. • Glycals are initially treated with dimethyldioxirane which transfers an oxygen bond to the glycal resulting in an epoxide. • The epox ...
... • While glycals were originally used in the synthesis of 2deoxyglycosides, Danishefsky and coworkers devised a glycal strategy that afforded 2-hydroxyglycosides. • Glycals are initially treated with dimethyldioxirane which transfers an oxygen bond to the glycal resulting in an epoxide. • The epox ...
Chapter 12 Organic Compounds with Oxygen and Sulfur Alcohols
... Oxidation of Primary (1°) Alcohols When a primary alcohol is oxidized, [O], • one H is removed from the –OH. • another H is removed from the carbon bonded to the OH. • an aldehyde is produced. [O] Primary alcohol ...
... Oxidation of Primary (1°) Alcohols When a primary alcohol is oxidized, [O], • one H is removed from the –OH. • another H is removed from the carbon bonded to the OH. • an aldehyde is produced. [O] Primary alcohol ...
Theoretical studies of nitrilotriacetic acid and nitrilotripropionic acid geometries for
... Nitrilotriacetic Acid (NTA) is an organic ligand which has been extensively studied due to its biological significance and excellent chelating properties. Nitrilotripropionic Acid (NTPA) is a ligand that is believed to possess similar properties to NTA, but has not been as extensively studied. It ha ...
... Nitrilotriacetic Acid (NTA) is an organic ligand which has been extensively studied due to its biological significance and excellent chelating properties. Nitrilotripropionic Acid (NTPA) is a ligand that is believed to possess similar properties to NTA, but has not been as extensively studied. It ha ...
Infrared - ResearchGate
... Number of bands can be used to decide on symmetry of a molecule Tha task of assignment is complicated by presence of low intensity bands and presence of forbidden overtone and combination bands. There are different levels at which information from IR can be analyzed to allow identification of sample ...
... Number of bands can be used to decide on symmetry of a molecule Tha task of assignment is complicated by presence of low intensity bands and presence of forbidden overtone and combination bands. There are different levels at which information from IR can be analyzed to allow identification of sample ...
Organic – Nomenclature – III
... group having highest priority(see above table) is carboxylic acid(-COOH). So the parent compound is a carboxylic acid(oic acid), not alcohol(ol), nitrile or a ketone(-one). The carboxyl carbon is numbered 1 as per rule. All functional groups other than the highest priority group(COOH) excepting the ...
... group having highest priority(see above table) is carboxylic acid(-COOH). So the parent compound is a carboxylic acid(oic acid), not alcohol(ol), nitrile or a ketone(-one). The carboxyl carbon is numbered 1 as per rule. All functional groups other than the highest priority group(COOH) excepting the ...
Alcohols, Phenols, Thiols, & Ethers
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
Cyanide detoxification process
... cyanide compounds, and there are more than 64 oxidation states of these metals capable of forming complex cyanides under certain conditions (Smith A. and Mudder T., The Chemistry and Treatment of Cyanidation Wastes, Mining Journal Books Limited, London, 1991). Two factors influencing the rate of dis ...
... cyanide compounds, and there are more than 64 oxidation states of these metals capable of forming complex cyanides under certain conditions (Smith A. and Mudder T., The Chemistry and Treatment of Cyanidation Wastes, Mining Journal Books Limited, London, 1991). Two factors influencing the rate of dis ...
CH 2
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
... 1-propanol would have the higher boiling point because an alcohol can form hydrogen bonds, but the ether cannot. ...
The Results from a Canadian National Field Trial Comparing 1,8-
... [8]. For this reason, and the fact that DFO has proven to be more sensitive to amino acids than ninhydrin, it is recommended that DFO be used in a sequence prior to ninhydrin [3]. In contrast to ninhydrin, the reaction mechanism and analogues of DFO have not received much attention in the ten years ...
... [8]. For this reason, and the fact that DFO has proven to be more sensitive to amino acids than ninhydrin, it is recommended that DFO be used in a sequence prior to ninhydrin [3]. In contrast to ninhydrin, the reaction mechanism and analogues of DFO have not received much attention in the ten years ...
Organic Chemistry introduction
... Assign numbers to each C of the parent chain. For alkenes and alkynes the first carbon of the multiple bond should have the smallest number. For alkanes the first branch (or first point of difference) should have the lowest #. Carbons in a multiple bond must be numbered consecutively. Attach a prefi ...
... Assign numbers to each C of the parent chain. For alkenes and alkynes the first carbon of the multiple bond should have the smallest number. For alkanes the first branch (or first point of difference) should have the lowest #. Carbons in a multiple bond must be numbered consecutively. Attach a prefi ...
What is Organic Chemistry?
... (valence electrons) or one half the bondlength of a diatomic molecule H—H bond length is 0.74Å, atomic radius is 0.37Å Atomic radius increases with increasing number of electron shells within an atom and decreases with the increase in the number of protons within an atom Thus atomic radius decreases ...
... (valence electrons) or one half the bondlength of a diatomic molecule H—H bond length is 0.74Å, atomic radius is 0.37Å Atomic radius increases with increasing number of electron shells within an atom and decreases with the increase in the number of protons within an atom Thus atomic radius decreases ...
Chapter 14
... yes, alcohols with 1-4 carbon atoms hydrogen bond with water yes; the water can hydrogen bond to the O in ether no; a carbon chain longer than 4 carbon atoms diminishes the effect of the OH group. no; alkanes are nonpolar and do not hydrogen bond yes; the OH in phenol ionizes in water, which makes ...
... yes, alcohols with 1-4 carbon atoms hydrogen bond with water yes; the water can hydrogen bond to the O in ether no; a carbon chain longer than 4 carbon atoms diminishes the effect of the OH group. no; alkanes are nonpolar and do not hydrogen bond yes; the OH in phenol ionizes in water, which makes ...
Chapter 10 Chemical Calculations and Chemical Equations
... 7. For some chemical reactions, chemists want to mix reactants in amounts that are as close as possible to the ratio that would lead to the complete reaction of each. This ratio is sometimes called the stoichiometric ratio. 9. Sometimes one product is more important than others are, and the amounts ...
... 7. For some chemical reactions, chemists want to mix reactants in amounts that are as close as possible to the ratio that would lead to the complete reaction of each. This ratio is sometimes called the stoichiometric ratio. 9. Sometimes one product is more important than others are, and the amounts ...
Outline
... Nanoscale Structures The self-assembly process mainly includes non-covalent interactions such as electrostatic, hydrogen-bonding,π-π stacking, and metal-coordination approaches. ...
... Nanoscale Structures The self-assembly process mainly includes non-covalent interactions such as electrostatic, hydrogen-bonding,π-π stacking, and metal-coordination approaches. ...
Derivatization reactions for the determination of amines by gas
... pathways are uncertain, it has also been demonstrated in model experiments that amines react with NO x and O H radicals in air to form nitrosamines and nitramines [13,27,34]. ...
... pathways are uncertain, it has also been demonstrated in model experiments that amines react with NO x and O H radicals in air to form nitrosamines and nitramines [13,27,34]. ...
Alcohol, Ethers, and Thiols
... Ethers as Anesthetics Anesthetics • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F F Cl F H ...
... Ethers as Anesthetics Anesthetics • inhibit pain signals to the brain. • like diethyl ether CH3─CH2─O─CH2─CH3 were used for over a century, but caused nausea and were flammable. • developed by the 1960’s were nonflammable. Cl F F Cl F H ...
Coordination Chemistry of Sugar
... Diese Dissertation wurde im Sinne von § 7 der Promotionsordnung vom 28. November 2011 von Herrn Prof. Dr. Peter Klüfers betreut. ...
... Diese Dissertation wurde im Sinne von § 7 der Promotionsordnung vom 28. November 2011 von Herrn Prof. Dr. Peter Klüfers betreut. ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.