ORGANIC CHEMISTRY
... Locate the carbonyl group and name the parent carboxylic acid. Replace the “oic acid” with the ending –amide. A - Primary Amide – if there are two hydrogen atoms bonded to the nitrogen. This name needs no prefix. B – Secondary Amide – if there is one alkyl group attached to the nitrogen. Name and lo ...
... Locate the carbonyl group and name the parent carboxylic acid. Replace the “oic acid” with the ending –amide. A - Primary Amide – if there are two hydrogen atoms bonded to the nitrogen. This name needs no prefix. B – Secondary Amide – if there is one alkyl group attached to the nitrogen. Name and lo ...
amine
... The five carbon atoms and the sp2-hybridized nitrogen atom of pyridine contribute one p electron to the aromatic sextet • The lone-pair electrons of nitrogen atom occupy an sp2 orbital in the plane of the ring • Pyridine is less basic that alkylamines because the lone-pair electrons are in an sp2 or ...
... The five carbon atoms and the sp2-hybridized nitrogen atom of pyridine contribute one p electron to the aromatic sextet • The lone-pair electrons of nitrogen atom occupy an sp2 orbital in the plane of the ring • Pyridine is less basic that alkylamines because the lone-pair electrons are in an sp2 or ...
Higher-Coordinated Molecular Silicon Compounds
... which contain sixfold-coordinated silicon atoms, have been reported [8–10]. An even larger variety of molecular compounds containing higher-coordinated silicon atoms are known. These include neutral, anionic, as well as cationic species with mono-, bi-, and multidentate ligands. The probably best-kn ...
... which contain sixfold-coordinated silicon atoms, have been reported [8–10]. An even larger variety of molecular compounds containing higher-coordinated silicon atoms are known. These include neutral, anionic, as well as cationic species with mono-, bi-, and multidentate ligands. The probably best-kn ...
fundamental concept of transition and inner
... Mercury is the only metal amongst transition elements which is liquid at 0oC due to weak metallic bond. (e) Transition metals show variable oxidation states due to participation of ns and (n-1) d electrons in bonding. (f) First ionization energy of 5d elements is higher than those of 3d and 4d eleme ...
... Mercury is the only metal amongst transition elements which is liquid at 0oC due to weak metallic bond. (e) Transition metals show variable oxidation states due to participation of ns and (n-1) d electrons in bonding. (f) First ionization energy of 5d elements is higher than those of 3d and 4d eleme ...
5.5 Reactions of Inorganic Compounds in - A
... Pink solution blue precipitate [Co(H2O)4(OH)2](s) + 6NH3(aq) == [Co(NH3)6]2+(aq) + 4H2O(l) + 2OH-(aq) blue precipate straw-coloured solution The complex [Co(NH3)6]2+ is oxidised to [Co(NH3)6]3+ on standing in air: [Co(NH3)6]2+ [Co(NH3)6]3+ + e straw-coloured solution brown solution With Cu(H ...
... Pink solution blue precipitate [Co(H2O)4(OH)2](s) + 6NH3(aq) == [Co(NH3)6]2+(aq) + 4H2O(l) + 2OH-(aq) blue precipate straw-coloured solution The complex [Co(NH3)6]2+ is oxidised to [Co(NH3)6]3+ on standing in air: [Co(NH3)6]2+ [Co(NH3)6]3+ + e straw-coloured solution brown solution With Cu(H ...
Chapter 20 Organic Chemistry
... models not only show you the attachment pattern, but give you an idea about the shape of the molecule ...
... models not only show you the attachment pattern, but give you an idea about the shape of the molecule ...
Course Notes
... H + strength of the magnetic field r = radius of the path V = accelerating potential placing a detector at some point along the flight path of the ion allows its mass to charge ratio to be calculated. Since almost all the ions will have a charge of +1, the mass to charge ratio is also the mass. A ma ...
... H + strength of the magnetic field r = radius of the path V = accelerating potential placing a detector at some point along the flight path of the ion allows its mass to charge ratio to be calculated. Since almost all the ions will have a charge of +1, the mass to charge ratio is also the mass. A ma ...
Nucleophilic ring opening of aziridines
... 2.2. Enamines, enolates and olefins Indoles have been found to be good substrates for ring opening of aziridines under appropriate Lewis acid catalysis conditions. 2-Substituted indole 26 having enamine functionality embedded in the heteroaryl ring readily underwent nucleophilic addition to activate ...
... 2.2. Enamines, enolates and olefins Indoles have been found to be good substrates for ring opening of aziridines under appropriate Lewis acid catalysis conditions. 2-Substituted indole 26 having enamine functionality embedded in the heteroaryl ring readily underwent nucleophilic addition to activate ...
Theoretical Studies on Catalytic Bond Activation
... importantly, the offering of insights leading to new practical knowledge. This thesis concentrates on the subject of catalysis, in particular homogeneous catalysis, which is of prime importance in synthetic organic chemistry. A good choice of catalyst can decrease reaction barriers, thereby providin ...
... importantly, the offering of insights leading to new practical knowledge. This thesis concentrates on the subject of catalysis, in particular homogeneous catalysis, which is of prime importance in synthetic organic chemistry. A good choice of catalyst can decrease reaction barriers, thereby providin ...
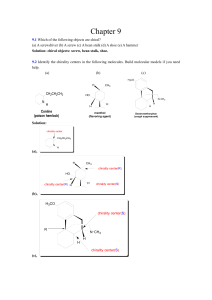
SCH4U TEXT BOOK
... from living things. In 1828, however, the German chemist Friedrich Wohler (1800–1882) made an organic compound called urea, CO(NH2)2 , out of an inorganic compound called ammonium cyanate, NH4CN. Urea is found in the urine of mammals. This was the first time in history that a compound normally made ...
... from living things. In 1828, however, the German chemist Friedrich Wohler (1800–1882) made an organic compound called urea, CO(NH2)2 , out of an inorganic compound called ammonium cyanate, NH4CN. Urea is found in the urine of mammals. This was the first time in history that a compound normally made ...
c8h18 isomers
... ♦ The molecules are non-polar or very weakly polar ♦ The forces holding together non-polar molecules are van der Waals forces. These intermolecular forces, which operate only over very small distances, result from induced polarization of the electron clouds in molecules. ♦ Within a family: The large ...
... ♦ The molecules are non-polar or very weakly polar ♦ The forces holding together non-polar molecules are van der Waals forces. These intermolecular forces, which operate only over very small distances, result from induced polarization of the electron clouds in molecules. ♦ Within a family: The large ...
Superoxide dismutases—a review of the metal
... In the former scenario, stable solutions of DMSO-O2•− can then be mixed in a 1:10 ratio (either with a single mixer or a triple mixer system) with water containing the enzyme. In the latter scenario, relatively stable high pH solutions of O2•− can be prepared as, at pH 12 for example, the half-life ...
... In the former scenario, stable solutions of DMSO-O2•− can then be mixed in a 1:10 ratio (either with a single mixer or a triple mixer system) with water containing the enzyme. In the latter scenario, relatively stable high pH solutions of O2•− can be prepared as, at pH 12 for example, the half-life ...
A Retrospective from Industrial and Engineering Chemistry Research
... purposes like knobs, buttons, knife handles, for which plastics are generally used. But its use for such fancy articles has not much appealed to my efforts as long as there are so many more important applications for engineering purposes.” However, the application of Bakelite, and the phenolformalde ...
... purposes like knobs, buttons, knife handles, for which plastics are generally used. But its use for such fancy articles has not much appealed to my efforts as long as there are so many more important applications for engineering purposes.” However, the application of Bakelite, and the phenolformalde ...
Aldehydes, Ketones, & Chiral Molecules
... With a common name by indicating the alkyl groups attached to the carbonyl group in alphabetical order followed by ketone. O O ...
... With a common name by indicating the alkyl groups attached to the carbonyl group in alphabetical order followed by ketone. O O ...
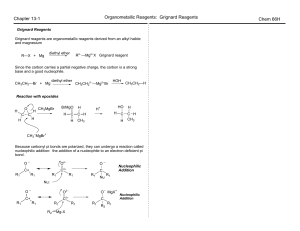
Chapter 13
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. Copyright © 2007 by Pearson Education, Inc. Publishing as Benjamin Cummings ...
Alcohols, Phenols, Thiols and Ethers
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. ...
... electronegative O in the OH groups. Form hydrogen bonds between alcohol molecules. Have higher boiling points than alkanes and ethers of similar mass. ...
September 11, 2007 - University of Idaho
... Trapping of the volatile gases using Tenax® showed no volatile organic carbon of molecular weight C4 and above released from the system during the course of the reaction ...
... Trapping of the volatile gases using Tenax® showed no volatile organic carbon of molecular weight C4 and above released from the system during the course of the reaction ...
File
... molecules are also present. There is an apparent increase in ordering when these ions are placed in water as compared to the separated state. The hydrating water molecules must be in a highly ordered arrangement when surrounding these anions. G = RTlnK = H TS; HX(aq) ⇌ H+(aq) + X(aq) Ka re ...
... molecules are also present. There is an apparent increase in ordering when these ions are placed in water as compared to the separated state. The hydrating water molecules must be in a highly ordered arrangement when surrounding these anions. G = RTlnK = H TS; HX(aq) ⇌ H+(aq) + X(aq) Ka re ...
Read Article - Roald Hoffmann
... in size going from C r to Mo to W allows more metal-metal interaction. I n Cr2C1g3- there are no d electrons paired in metal-metal bonds, and the complex is elongated from an ideal octahedron of 0 = M-X-M = 70.5’. In MO2C1g3- two electrons are paired in a metal-metal bond and the complex is now slig ...
... in size going from C r to Mo to W allows more metal-metal interaction. I n Cr2C1g3- there are no d electrons paired in metal-metal bonds, and the complex is elongated from an ideal octahedron of 0 = M-X-M = 70.5’. In MO2C1g3- two electrons are paired in a metal-metal bond and the complex is now slig ...
Hydroformylation

Hydroformylation, also known as oxo synthesis or oxo process, is an important homogeneously catalyzed industrial process for the production of aldehydes from alkenes. This chemical reaction entails the addition of a formyl group (CHO) and a hydrogen atom to a carbon-carbon double bond. This process has undergone continuous growth since its invention in 1938: Production capacity reached 6.6×106 tons in 1995. It is important because the resulting aldehydes are easily converted into many secondary products. For example, the resulting aldehydes are hydrogenated to alcohols that are converted to plasticizers or detergents. Hydroformylation is also used in specialty chemicals, relevant to the organic synthesis of fragrances and natural products. The development of hydroformylation, which originated within the German coal-based industry, is considered one of the premier achievements of 20th-century industrial chemistry.The process typically entails treatment of an alkene with high pressures (between 10 to 100 atmospheres) of carbon monoxide and hydrogen at temperatures between 40 and 200 °C. Transition metal catalysts are required.