Chem I Review Part 2
... 92. Which of these substances will display an incomplete octet in its Lewis structure? A. CO2 B. Cl2 C. ICl D. NO E. SO2 93. Which of these elements is most likely to exhibit an expanded octet in its compounds? A. O B. S C. Na D. C E. N 94. According to the VSEPR theory, the shape of the SO3 molecu ...
... 92. Which of these substances will display an incomplete octet in its Lewis structure? A. CO2 B. Cl2 C. ICl D. NO E. SO2 93. Which of these elements is most likely to exhibit an expanded octet in its compounds? A. O B. S C. Na D. C E. N 94. According to the VSEPR theory, the shape of the SO3 molecu ...
File - jj-sct
... A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
... A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
Lecture Presentation Book - Pottstown School District
... An element is a substance that cannot be broken down to other substances by chemical reactions A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
... An element is a substance that cannot be broken down to other substances by chemical reactions A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
02_Lecture_Presentation
... An element is a substance that cannot be broken down to other substances by chemical reactions A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
... An element is a substance that cannot be broken down to other substances by chemical reactions A compound is a substance consisting of two or more elements in a fixed ratio A compound has characteristics different from those of its elements ...
Unit 1 Atoms, Molecules and Stoichiometry
... In 1897, J. J. Thomson investigated the conductivity of electricity by gases at very low pressure. At ordinary pressures gases are electrical insulators, but when they are subjected to very high voltages at very low pressures (below 0.01 atm) they break down and conduct electricity. When Thomson app ...
... In 1897, J. J. Thomson investigated the conductivity of electricity by gases at very low pressure. At ordinary pressures gases are electrical insulators, but when they are subjected to very high voltages at very low pressures (below 0.01 atm) they break down and conduct electricity. When Thomson app ...
WIPO IPC: Internet Publication
... and a non-chemical part or aspect, the general rule is that the chemical part or aspect is covered by section C. In some of these cases, the chemical part or aspect brings with it a non-chemical one, even though purely mechanical, because this latter aspect either is essential to the operation or tr ...
... and a non-chemical part or aspect, the general rule is that the chemical part or aspect is covered by section C. In some of these cases, the chemical part or aspect brings with it a non-chemical one, even though purely mechanical, because this latter aspect either is essential to the operation or tr ...
CBSE Living Science Chemistry Class X
... the total mass of all the products. Hence, the chemical equation, as written above, is inaccurate. In a chemical equation, we must have the equal number of atoms of each element in the reactants and the products. This is known as balancing a chemical equation. If in a chemical equation the number of ...
... the total mass of all the products. Hence, the chemical equation, as written above, is inaccurate. In a chemical equation, we must have the equal number of atoms of each element in the reactants and the products. This is known as balancing a chemical equation. If in a chemical equation the number of ...
An Introduction to Chemical Science
... For the benefit of the science and the added interest in the study, it is earnestly recommended that teachers encourage pupils to fit up laboratories of their own at home. This need not at first entail a large outlay. A small attic room with running water, a very few chemicals, and a little apparatu ...
... For the benefit of the science and the added interest in the study, it is earnestly recommended that teachers encourage pupils to fit up laboratories of their own at home. This need not at first entail a large outlay. A small attic room with running water, a very few chemicals, and a little apparatu ...
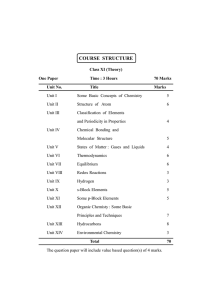
COURSE STRUCTURE
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
support material
... Avogadro Law (In 1811, Given by Avogadro) According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical ...
... Avogadro Law (In 1811, Given by Avogadro) According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory All substances are made up of tiny, indivisible particles called atoms. Atoms of the same element are identical ...
Chemical Compounds
... Start with “leftmost” (in periodic table) element. In cases of ties, go “downmost”. Use Greek prefixes to designate the number of each element – although a leading “mono” is usually dropped. End with “-ide” ...
... Start with “leftmost” (in periodic table) element. In cases of ties, go “downmost”. Use Greek prefixes to designate the number of each element – although a leading “mono” is usually dropped. End with “-ide” ...
Stereochemistry - Kantipur Engineering College
... 7. Sequence rule for assigning R or S configuration to the stereocenters This rule was developed by Cahn, Ingold, and Prelog to assign R and S configuration to the stereocenters. This system of nomenclature is also known as R, S-system or Cahn-Ingold-Prelog system and has been a part of IUPAC system ...
... 7. Sequence rule for assigning R or S configuration to the stereocenters This rule was developed by Cahn, Ingold, and Prelog to assign R and S configuration to the stereocenters. This system of nomenclature is also known as R, S-system or Cahn-Ingold-Prelog system and has been a part of IUPAC system ...
Ch. 20 - Chemical Bonds - Study Guide
... c. They have a slightly positive end and a slightly negative end. d. They contain at least two hydrogen atoms. ____ 38. What is the name of a binary compound made up of lithium and chlorine? a. chlorine lithium c. lithium chloride b. lithium chlorate d. chlorine lithiate ____ 39. How many electrons ...
... c. They have a slightly positive end and a slightly negative end. d. They contain at least two hydrogen atoms. ____ 38. What is the name of a binary compound made up of lithium and chlorine? a. chlorine lithium c. lithium chloride b. lithium chlorate d. chlorine lithiate ____ 39. How many electrons ...
Chemical Bonding
... There are many ways to classify substances. You are already familiar with several of them. For example, you know that iron is a solid at SATP (not a liquid or a gas), a metal (not a nonmetal), and an element (not a compound). Similarly, water is a liquid and a compound. In this section, we will look ...
... There are many ways to classify substances. You are already familiar with several of them. For example, you know that iron is a solid at SATP (not a liquid or a gas), a metal (not a nonmetal), and an element (not a compound). Similarly, water is a liquid and a compound. In this section, we will look ...
Study Material - Class- XI- Chemistry
... All reactants and products have simple ratio 1:1:2. Avogadro Law (In 1811, Given by Avogadro) According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory *All substances are made up of tiny, indivisible particles ...
... All reactants and products have simple ratio 1:1:2. Avogadro Law (In 1811, Given by Avogadro) According to this law equal volumes of gases at the same temperature and pressure should contain equal number of molecules. Dalton's Atomic Theory *All substances are made up of tiny, indivisible particles ...
PDF of Chapter 6 Foundations of Chemistry
... Before you read, decide if you agree or disagree with each of these statements. As you read this chapter, see if you change your mind about any of the statements. 1 The atoms in all objects are the same. 2 You cannot always tell by an object’s appearance whether it is made of more than one type of a ...
... Before you read, decide if you agree or disagree with each of these statements. As you read this chapter, see if you change your mind about any of the statements. 1 The atoms in all objects are the same. 2 You cannot always tell by an object’s appearance whether it is made of more than one type of a ...
Physical Science e
... Each book in the Power Practice™ series contains over 100 ready-to-use activity pages to provide students with skill practice. The fun activities can be used to supplement and enhance what you are teaching in your classroom. Give an activity page to students as independent class work, or send the pa ...
... Each book in the Power Practice™ series contains over 100 ready-to-use activity pages to provide students with skill practice. The fun activities can be used to supplement and enhance what you are teaching in your classroom. Give an activity page to students as independent class work, or send the pa ...
1 of 52
... (a) A sample of ABS plastic contains 8.80% N by mass. It took 0.605 g of Br2 to react completely with a 1.20 g sample of ABS plastic. Bromine reacts 1:1 (by moles) with the butadiene molecules in the polymer and nothing else. What is the percent by mass of acrylonitrile and butadiene in this polymer ...
... (a) A sample of ABS plastic contains 8.80% N by mass. It took 0.605 g of Br2 to react completely with a 1.20 g sample of ABS plastic. Bromine reacts 1:1 (by moles) with the butadiene molecules in the polymer and nothing else. What is the percent by mass of acrylonitrile and butadiene in this polymer ...
chemistry writing team
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
... Law of conservation of mass : ‘Mass can neither be created nor destroyed.’ In all physical and chemical changes, the total mass of reactants is equal to that of products. Law of constant composition : A chemical compound is always found to be made of same elements combined together in the same fixed ...
physical setting chemistry
... questions in this examination. Some questions may require the use of the Reference Tables for Physical Setting/Chemistry. You are to answer all questions in all parts of this examination according to the directions provided in the examination booklet. Your answer sheet for Part A and Part B–1 is the ...
... questions in this examination. Some questions may require the use of the Reference Tables for Physical Setting/Chemistry. You are to answer all questions in all parts of this examination according to the directions provided in the examination booklet. Your answer sheet for Part A and Part B–1 is the ...
17.2 The Avogadro Number
... The large number in front of H2 tells how many molecules of H2 are required for the reaction to proceed. The large number in front of H2O tells how many molecules of water are formed by the reaction. These numbers are called coefficients. Using coefficients, we can balance chemical equations so that ...
... The large number in front of H2 tells how many molecules of H2 are required for the reaction to proceed. The large number in front of H2O tells how many molecules of water are formed by the reaction. These numbers are called coefficients. Using coefficients, we can balance chemical equations so that ...
chapter 3
... 3.4 The Chemical Mole The mole is defined as the # of C atoms in exactly 12 grams of Carbon-12: 1 mole C atoms = 6.02x1023 C atoms ...
... 3.4 The Chemical Mole The mole is defined as the # of C atoms in exactly 12 grams of Carbon-12: 1 mole C atoms = 6.02x1023 C atoms ...
Atomic Masses
... 9.5 trillion kilometers!!!!!!!!!! Why do you think we measure distances to nearby stars in light years and not kilometers????? • Consider the reaction below, In order to make one molecule of CO2 (g), each carbon atom needs how many molecules of oxygen gas???? C(s) + ...
... 9.5 trillion kilometers!!!!!!!!!! Why do you think we measure distances to nearby stars in light years and not kilometers????? • Consider the reaction below, In order to make one molecule of CO2 (g), each carbon atom needs how many molecules of oxygen gas???? C(s) + ...