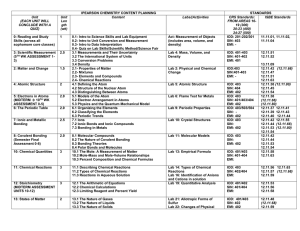
85 Q.1 A substance X melts at 1600oC. Its does
... 35 Br and 35 Br . The relative atomic mass of bromine is 79.9. Which of the following statements is/are correct? (1) The relative abundance of each isotopic form is about the same. (2) The two isotopes have different numbers of protons. (3) The two isotopes have different numbers of neutrons. ...
... 35 Br and 35 Br . The relative atomic mass of bromine is 79.9. Which of the following statements is/are correct? (1) The relative abundance of each isotopic form is about the same. (2) The two isotopes have different numbers of protons. (3) The two isotopes have different numbers of neutrons. ...
Hadronic Chemistry and Binding Energies
... obtained do not accurately match with the experimental values [1, 2]. The energy calculated for H2 molecule by LCAO-MO method is observed to be -1.0985 a.u. (Binding energy = - 2.681 eV) as against the experimental value of -1.174 a.u. (Binding energy = - 4.75 eV). By applying the valence-bond (VB) ...
... obtained do not accurately match with the experimental values [1, 2]. The energy calculated for H2 molecule by LCAO-MO method is observed to be -1.0985 a.u. (Binding energy = - 2.681 eV) as against the experimental value of -1.174 a.u. (Binding energy = - 4.75 eV). By applying the valence-bond (VB) ...
P-BLOCK ELEMENTS
... * * Thalium, Lead and Bismuth show valency two unit less than their group valency due to inert pair effect. (v) P-block elements have high ionization enthalpy, high electron gain enthalpy and high electronegativity hence, they have strong tendency to form anions (vi) Metallic character increases dow ...
... * * Thalium, Lead and Bismuth show valency two unit less than their group valency due to inert pair effect. (v) P-block elements have high ionization enthalpy, high electron gain enthalpy and high electronegativity hence, they have strong tendency to form anions (vi) Metallic character increases dow ...
Answers to Selected Exercises
... (b) Atomic weights are average atomic masses, the sum of the mass of each naturally occurring isotope of an element times its fractional abundance. Each B atom will have the mass of one of the naturally occurring isotopes, while the ‘atomic weight’ is an average value. 1.55 (a) Cr (metal) (b) He (no ...
... (b) Atomic weights are average atomic masses, the sum of the mass of each naturally occurring isotope of an element times its fractional abundance. Each B atom will have the mass of one of the naturally occurring isotopes, while the ‘atomic weight’ is an average value. 1.55 (a) Cr (metal) (b) He (no ...
Oxidation
... Other nonmetals: the element closest to fluorine on the PT gets to keep its “usual” O.S. ...
... Other nonmetals: the element closest to fluorine on the PT gets to keep its “usual” O.S. ...
Course Map_2011-2012 - Kenwood Academy High School
... 12.11.74 Understand that the magnitude of a force F is defined as F = ma (Force equals Mass times Acceleration). Know how to perform such calculations. Understand that whenever one object exerts force on another, a force equal in magnitude and opposite in direction is exerted on the first object. Un ...
... 12.11.74 Understand that the magnitude of a force F is defined as F = ma (Force equals Mass times Acceleration). Know how to perform such calculations. Understand that whenever one object exerts force on another, a force equal in magnitude and opposite in direction is exerted on the first object. Un ...
chemical bonding i: basic concepts
... The Lewis theory of bonding describes a covalent bond as the sharing of a pair of electrons, but this does not necessarily mean that each atom contributes an electron to the bond. A covalent bond in which a single atom contributes both of the electrons to a shared pair is called a coordinate covalen ...
... The Lewis theory of bonding describes a covalent bond as the sharing of a pair of electrons, but this does not necessarily mean that each atom contributes an electron to the bond. A covalent bond in which a single atom contributes both of the electrons to a shared pair is called a coordinate covalen ...
Chapter One
... began excavations at an archaeological site on Dragon-Bone Hill, near the town of Chou-k’outien, 34 miles southwest of Beijing, China. Fossils found at this site were assigned to a new species, Homo erectus pekinensis, commonly known as Peking man. These excavations suggest that for at least 500,000 ...
... began excavations at an archaeological site on Dragon-Bone Hill, near the town of Chou-k’outien, 34 miles southwest of Beijing, China. Fossils found at this site were assigned to a new species, Homo erectus pekinensis, commonly known as Peking man. These excavations suggest that for at least 500,000 ...
Final Exam Review Notes
... Some numbers are very large or very small difficult to express. For example, Avogadro’s number = 602,000,000,000,000,000,000,000 an electron’s mass = 0.000 000 000 000 000 000 000 000 000 91 kg Also, it's not clear how many sig figs there are in some measurements. For example, Express 100.0 g to 3 ...
... Some numbers are very large or very small difficult to express. For example, Avogadro’s number = 602,000,000,000,000,000,000,000 an electron’s mass = 0.000 000 000 000 000 000 000 000 000 91 kg Also, it's not clear how many sig figs there are in some measurements. For example, Express 100.0 g to 3 ...
Chapter 2 - San Joaquin Memorial High School
... compounds to replace the awkward symbolic representations of the alchemists. Although some chemists, including Dalton, objected to the new system, it was gradually adopted and forms the basis of the system we use today. In addition to these accomplishments, Berzelius discovered the elements cerium, ...
... compounds to replace the awkward symbolic representations of the alchemists. Although some chemists, including Dalton, objected to the new system, it was gradually adopted and forms the basis of the system we use today. In addition to these accomplishments, Berzelius discovered the elements cerium, ...
Chem I Review Part 2
... 69. Which of these ionic solids would have the largest lattice energy? A. NaCl B. NaF C. CaBr2 D. CsI E. CaCl2 70. Which of these solids would have the highest melting point? A. NaF B. NaCl C. NaBr D. NaI 71. Which of these elements has the greatest electronegativity? A. Na B. As C. Ga D. Cs E. Sb ...
... 69. Which of these ionic solids would have the largest lattice energy? A. NaCl B. NaF C. CaBr2 D. CsI E. CaCl2 70. Which of these solids would have the highest melting point? A. NaF B. NaCl C. NaBr D. NaI 71. Which of these elements has the greatest electronegativity? A. Na B. As C. Ga D. Cs E. Sb ...
Final Exam - KFUPM Faculty List
... and Eea = electron affinity) A) an element with a large Ei and an element with a small negative Eea no, because the cation formation needs a large Ei and the anion formation provides only a small negative Eea B) an element with a small Ei and an element with a small negative Eea no, the cation form ...
... and Eea = electron affinity) A) an element with a large Ei and an element with a small negative Eea no, because the cation formation needs a large Ei and the anion formation provides only a small negative Eea B) an element with a small Ei and an element with a small negative Eea no, the cation form ...
12 - einstein classes
... Reasons : (a) small size (b) high EN, IE (c) absence of d-orbitals and hence cannot expand its covalency more than 4. (d) has tendency to form p-p multiple bonds with itself and other elements having small size and high EN like carbon and oxygen. ...
... Reasons : (a) small size (b) high EN, IE (c) absence of d-orbitals and hence cannot expand its covalency more than 4. (d) has tendency to form p-p multiple bonds with itself and other elements having small size and high EN like carbon and oxygen. ...
Worksheet 1 - Oxidation/Reduction Reactions Oxidation number
... Balancing Redox Reactions Oxidation/Reduction (Redox) reactions can be balanced using the oxidation state changes, as seen in the previous example. However, there is an easier method, which involves breaking a redox reaction into two half- reactions. This is best shown by working an example. Hydrob ...
... Balancing Redox Reactions Oxidation/Reduction (Redox) reactions can be balanced using the oxidation state changes, as seen in the previous example. However, there is an easier method, which involves breaking a redox reaction into two half- reactions. This is best shown by working an example. Hydrob ...
Chemistry Entrance Material for Grade 11 to 12 Answer Key
... [-D-] it boils when heated in air Effect of temperature on vapour pressure 05. How does the vapour pressure of a liquid vary with temperature( T)? As T increase the vapor pressure increase. When a liquid boils it absorbs heat at constant temperature 06. When a liquid at its boiling point is heated, ...
... [-D-] it boils when heated in air Effect of temperature on vapour pressure 05. How does the vapour pressure of a liquid vary with temperature( T)? As T increase the vapor pressure increase. When a liquid boils it absorbs heat at constant temperature 06. When a liquid at its boiling point is heated, ...
jyvaskla2 - School of Chemistry
... via their associated bcp’s, and indicate their is no direct chemical bonding along the B-B vectors which are triply bridged by the H atom. ...
... via their associated bcp’s, and indicate their is no direct chemical bonding along the B-B vectors which are triply bridged by the H atom. ...
As a result of activities in grades 9
... water. Enthalpy of formation. "Special reactions" like combustion, electrolysis, photosynthesis, and respiration. Many other topics are covered in less detail. For example, the graduate will gain an understanding of the fundamentals of pH but not to the depths required for calculating it (because th ...
... water. Enthalpy of formation. "Special reactions" like combustion, electrolysis, photosynthesis, and respiration. Many other topics are covered in less detail. For example, the graduate will gain an understanding of the fundamentals of pH but not to the depths required for calculating it (because th ...
2 - TestBankTop
... The nuclear model of the atom is based on experiments of Geiger, Marsden, and Rutherford. Rutherford stated that most of the mass of an atom is concentrated in a positively charged center called the nucleus around which negatively charged electrons move. The nucleus, although it contains most of the ...
... The nuclear model of the atom is based on experiments of Geiger, Marsden, and Rutherford. Rutherford stated that most of the mass of an atom is concentrated in a positively charged center called the nucleus around which negatively charged electrons move. The nucleus, although it contains most of the ...
p Block Elements General Configuration: ns2 np1
... is the most basic whereas ASH3, SbH3, BiH3 do not show any basic properties. Thermal stability decreases down the group as the size of the central atom increases and the tendency to form stable M-H bond decreases. Reducing character increases down the group, because thermal stability of hydrides ...
... is the most basic whereas ASH3, SbH3, BiH3 do not show any basic properties. Thermal stability decreases down the group as the size of the central atom increases and the tendency to form stable M-H bond decreases. Reducing character increases down the group, because thermal stability of hydrides ...
Topological Analysis of Electron Density
... quite transferable and additive, but do not look very much like the balls and spheres of molecular models !!! The simple binary hydrides of the second period elements show that the relative volumes of space associated with each element is determined by their relative electronegativities. Surfaces ar ...
... quite transferable and additive, but do not look very much like the balls and spheres of molecular models !!! The simple binary hydrides of the second period elements show that the relative volumes of space associated with each element is determined by their relative electronegativities. Surfaces ar ...
Cleaning Up With Atom Economy
... as a cause than a solution to pollution, chemistry does offer unique solutions in the area of waste prevention. One of the most fundamental of these solutions is the application of the green chemistry principle of atom economy to chemical reactions. Atom economy moves the practice of minimizing wast ...
... as a cause than a solution to pollution, chemistry does offer unique solutions in the area of waste prevention. One of the most fundamental of these solutions is the application of the green chemistry principle of atom economy to chemical reactions. Atom economy moves the practice of minimizing wast ...
Atoms, Molecules, and Ions
... The nuclear model of the atom is based on experiments of Geiger, Marsden, and Rutherford. Rutherford stated that most of the mass of an atom is concentrated in a positively charged center called the nucleus around which negatively charged electrons move. The nucleus, although it contains most of the ...
... The nuclear model of the atom is based on experiments of Geiger, Marsden, and Rutherford. Rutherford stated that most of the mass of an atom is concentrated in a positively charged center called the nucleus around which negatively charged electrons move. The nucleus, although it contains most of the ...
Oxidation-Reduction Reactions
... Reduction is the gain of electrons by an atom, ion, or molecule. The atom, ion, or molecule that is reduced will become more negatively charged. Reduction may also involve the loss of oxygen or the gain of hydrogen. Tips for determining if an atom, ion, or compound has been Oxidized or Reduced: Reme ...
... Reduction is the gain of electrons by an atom, ion, or molecule. The atom, ion, or molecule that is reduced will become more negatively charged. Reduction may also involve the loss of oxygen or the gain of hydrogen. Tips for determining if an atom, ion, or compound has been Oxidized or Reduced: Reme ...
Chemistry Entrance Material for Grade 11 to 12
... Variation of molar heat of vaporization 10. Which of the following liquids has the lowest molar heat of vaporization? Which one has the highest molar heat of vaporization? Which one has the highest vapour pressure at its boiling point? ...
... Variation of molar heat of vaporization 10. Which of the following liquids has the lowest molar heat of vaporization? Which one has the highest molar heat of vaporization? Which one has the highest vapour pressure at its boiling point? ...
Electronegativity

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. The term ""electronegativity"" was introduced by Jöns Jacob Berzelius in 1811,though the concept was known even before that and was studied by many chemists including Avogadro.In spite of its long history, an accurate scale of electronegativity had to wait till 1932, when Linus Pauling proposed an electronegativity scale, which depends on bond energies, as a development of valence bond theory. It has been shown to correlate with a number of other chemical properties. Electronegativity cannot be directly measured and must be calculated from other atomic or molecular properties. Several methods of calculation have been proposed, and although there may be small differences in the numerical values of the electronegativity, all methods show the same periodic trends between elements. The most commonly used method of calculation is that originally proposed by Linus Pauling. This gives a dimensionless quantity, commonly referred to as the Pauling scale, on a relative scale running from around 0.7 to 3.98 (hydrogen = 2.20). When other methods of calculation are used, it is conventional (although not obligatory) to quote the results on a scale that covers the same range of numerical values: this is known as an electronegativity in Pauling units. As it is usually calculated, electronegativity is not a property of an atom alone, but rather a property of an atom in a molecule. Properties of a free atom include ionization energy and electron affinity. It is to be expected that the electronegativity of an element will vary with its chemical environment, but it is usually considered to be a transferable property, that is to say that similar values will be valid in a variety of situations.On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more ""pull"" it will have on electrons) and the number/location of other electrons present in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result the less positive charge they will experience—both because of their increased distance from the nucleus, and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).The opposite of electronegativity is electropositivity: a measure of an element's ability to donate electrons.Caesium is the least electronegative element in the periodic table (=0.79), while fluorine is most electronegative (=3.98). (Francium and caesium were originally assigned both assigned 0.7; caesium's value was later refined to 0.79, but no experimental data allows a similar refinement for francium. However, francium's ionization energy is known to be slightly higher than caesium's, in accordance with the relativistic stabilization of the 7s orbital, and this in turn implies that caesium is in fact more electronegative than francium.)