Power Point over chemistry
... oxygen gas using an electric current. When water molecules change chemically into hydrogen gas and oxygen gas, we say that a chemical change has occurred. Hydrogen gas and oxygen gas each have a different set of properties. Substances change into different substances through TAKS Need to Know chemic ...
... oxygen gas using an electric current. When water molecules change chemically into hydrogen gas and oxygen gas, we say that a chemical change has occurred. Hydrogen gas and oxygen gas each have a different set of properties. Substances change into different substances through TAKS Need to Know chemic ...
9.1 REDOX Introduction to Oxidation and Reduction
... oxygen was discovered by Joseph Priestley (17331804) Quickly realized that oxygen forms oxides so the word oxidation was created to describe the addition of oxygen When oxygen is removed “reduction” is used Now oxidation and reduction refer to transfer of ...
... oxygen was discovered by Joseph Priestley (17331804) Quickly realized that oxygen forms oxides so the word oxidation was created to describe the addition of oxygen When oxygen is removed “reduction” is used Now oxidation and reduction refer to transfer of ...
oxidation–reduction reaction
... compound is the number of electrons lost or gained by the atom when it forms ions. • Oxidation numbers are tools that scientists use in written chemical equations to help them keep track of the movement of electrons in a redox reaction. ...
... compound is the number of electrons lost or gained by the atom when it forms ions. • Oxidation numbers are tools that scientists use in written chemical equations to help them keep track of the movement of electrons in a redox reaction. ...
Chemistry Standards Clarification
... Given the structural formula of a compound, indicate all the intermolecular forces present (dispersion, dipolar, hydrogen bonding). Explain properties of various solids such as malleability, conductivity, and melting point in terms of the solid’s structure and bonding. Explain why ionic solids have ...
... Given the structural formula of a compound, indicate all the intermolecular forces present (dispersion, dipolar, hydrogen bonding). Explain properties of various solids such as malleability, conductivity, and melting point in terms of the solid’s structure and bonding. Explain why ionic solids have ...
GCE Getting Started - Edexcel
... Atoms of metallic elements in Groups 1,2 and 3 can form positive ions when they take part in reactions since they are readily able to lose electrons. Atoms of Group 1 metals lose one electron and form ions with a 1+ charge, e.g. Na+ Atoms of Group 2 metals lose two electrons and form ions with a 2+ ...
... Atoms of metallic elements in Groups 1,2 and 3 can form positive ions when they take part in reactions since they are readily able to lose electrons. Atoms of Group 1 metals lose one electron and form ions with a 1+ charge, e.g. Na+ Atoms of Group 2 metals lose two electrons and form ions with a 2+ ...
Full text
... state antisymmetric with respect to the exchange of the coordinates of any two electrons in order to satisfy the Pauli principle. If we neglect magnetic interactions, energies of members of the same multiplet will be the same since the total wave function will be simply a product of an orbital funct ...
... state antisymmetric with respect to the exchange of the coordinates of any two electrons in order to satisfy the Pauli principle. If we neglect magnetic interactions, energies of members of the same multiplet will be the same since the total wave function will be simply a product of an orbital funct ...
support material
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
... Kendriya Vidyalaya Sangathan is a pioneer organization which caters to the all round development of the students. Time to time various strategies have been adopted to adorn the students with academic excellence. This support material is one such effort by Kendriya Vidyalaya Sangathan, an empirical e ...
1b-Redox FIB notes and practice
... Ex) 2NaBr(aq) + Hg(NO3)2(aq) --> 2NaNO3(aq) + HgBr2(s) 3. The oxidation number of hydrogen in most of its compound is +1. The exception is when hydrogen bonds to metals to make a metal hydride. Ie. In NaH, H would be have an oxidation number of ____. This is because hydrogen attracts electrons more ...
... Ex) 2NaBr(aq) + Hg(NO3)2(aq) --> 2NaNO3(aq) + HgBr2(s) 3. The oxidation number of hydrogen in most of its compound is +1. The exception is when hydrogen bonds to metals to make a metal hydride. Ie. In NaH, H would be have an oxidation number of ____. This is because hydrogen attracts electrons more ...
chemistry
... juice, washing powder, shampoo, soap, salt water. Use either test strips or universal indicator. Your teacher will explain the meaning of the results. ...
... juice, washing powder, shampoo, soap, salt water. Use either test strips or universal indicator. Your teacher will explain the meaning of the results. ...
GCE Getting Started - Edexcel
... atoms, given the atomic number, Z, up to Z = 36 ii. ions, given the atomic number, Z, and the ionic charge, for s and p block ions only, up to Z = 36. Know that elements can be classified as s-, p- and d-block elements. Understand that electronic configuration determines the chemical properties of a ...
... atoms, given the atomic number, Z, up to Z = 36 ii. ions, given the atomic number, Z, and the ionic charge, for s and p block ions only, up to Z = 36. Know that elements can be classified as s-, p- and d-block elements. Understand that electronic configuration determines the chemical properties of a ...
Answers to Selected Exercises
... other. b. When wood burns, most of the solid material in wood is converted to gases, which escape. The gases produced are most likely CO2 and H2O. c. The atom is not an indivisible particle, but is instead composed of other smaller particles—electrons, neutrons, and protons. d. The two hydride sampl ...
... other. b. When wood burns, most of the solid material in wood is converted to gases, which escape. The gases produced are most likely CO2 and H2O. c. The atom is not an indivisible particle, but is instead composed of other smaller particles—electrons, neutrons, and protons. d. The two hydride sampl ...
Basic Agricultural Chemistry - Macmillan Education South Africa
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
... group share similar chemical properties. The Roman numerals listed above each group are the usual number of valence electrons. For example: l Group IA is the alkali metals where the element has one electron in the orbital, for example sodium, potassium and rubidium. Alkali metals form salts and man ...
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... 1. State the meaning of relative atomic mass based on C- 12 scale 2. State why C 12 is used as a standard for determining relative atomic mass and relative molecular mass 3. Caculate the relative molecular mass of substances. A student is able to 1. Define a mole as the amount of matter that contain ...
... 1. State the meaning of relative atomic mass based on C- 12 scale 2. State why C 12 is used as a standard for determining relative atomic mass and relative molecular mass 3. Caculate the relative molecular mass of substances. A student is able to 1. Define a mole as the amount of matter that contain ...
lecture slides file
... Specific gravity is the ratio of densities of a substance to that of water. It is dimensionless. When immersed in water, substances with density greater than 1 kg/L (e.g. most metals) sink, and those having density less 1 (ice, most liquids, wood, etc) float. Density depends on the way atoms are pac ...
... Specific gravity is the ratio of densities of a substance to that of water. It is dimensionless. When immersed in water, substances with density greater than 1 kg/L (e.g. most metals) sink, and those having density less 1 (ice, most liquids, wood, etc) float. Density depends on the way atoms are pac ...
KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
DEPARTMENT OF CHEMISTRY, CFS, IIUM
... Based on your answer to the question above, suggest the name for atom X. Sulphur (2i) ...
... Based on your answer to the question above, suggest the name for atom X. Sulphur (2i) ...
Lecture 2
... SALC can now be treated similarly to the atomic orbitals and combined with appropriate AO’s from H 1s(H) is Ag so it matches two SALC. The interaction can be bonding or antibonding. ...
... SALC can now be treated similarly to the atomic orbitals and combined with appropriate AO’s from H 1s(H) is Ag so it matches two SALC. The interaction can be bonding or antibonding. ...
Chapter 20
... No ionic charges are associated with atoms of molecular compounds. However, oxygen is reduced in the formation of water for example. In water the two shared e- in the H – O bond are shifted toward the O and away from the H. ...
... No ionic charges are associated with atoms of molecular compounds. However, oxygen is reduced in the formation of water for example. In water the two shared e- in the H – O bond are shifted toward the O and away from the H. ...
Topic 7b Redox notes
... sign followed by a number, or it is zero. Oxidation states of metals are usually written in roman numerals eg Iron (III) chloride. Atoms of elements have no overall charge and are therefore given an oxidation state of zero. When two elements combine, the atoms or ions of the more electropositive ele ...
... sign followed by a number, or it is zero. Oxidation states of metals are usually written in roman numerals eg Iron (III) chloride. Atoms of elements have no overall charge and are therefore given an oxidation state of zero. When two elements combine, the atoms or ions of the more electropositive ele ...
Chapter 8: Ionic Compounds
... in group 3A, [noble gas]ns2np1, also lose electrons and form positive ions. What is the charge on these ions? What is the formula for the aluminum ion? Recall that, in general, transition metals have an outer energy level of ns2. Going from left to right across a period, atoms of each element are fi ...
... in group 3A, [noble gas]ns2np1, also lose electrons and form positive ions. What is the charge on these ions? What is the formula for the aluminum ion? Recall that, in general, transition metals have an outer energy level of ns2. Going from left to right across a period, atoms of each element are fi ...
SQA CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
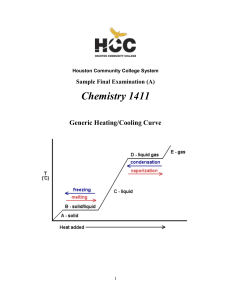
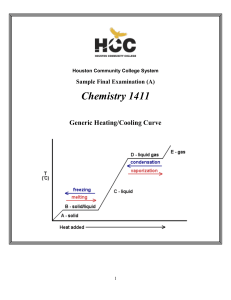
1411FINALSAMPLE+KEY - Houston Community College
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
CHEM-1411 Final Practice Exam
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
... sulfur atom in the first structure is therefore sp3. However, the sulfur is not simply sp3 hybridized in the second structure, which has an “expanded octet” around the sulfur atom. Hybridizations that allow more than an octet of electrons around an atom are sp3d (10 electrons) and sp3d2 (12 electron ...
Electronegativity

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. The term ""electronegativity"" was introduced by Jöns Jacob Berzelius in 1811,though the concept was known even before that and was studied by many chemists including Avogadro.In spite of its long history, an accurate scale of electronegativity had to wait till 1932, when Linus Pauling proposed an electronegativity scale, which depends on bond energies, as a development of valence bond theory. It has been shown to correlate with a number of other chemical properties. Electronegativity cannot be directly measured and must be calculated from other atomic or molecular properties. Several methods of calculation have been proposed, and although there may be small differences in the numerical values of the electronegativity, all methods show the same periodic trends between elements. The most commonly used method of calculation is that originally proposed by Linus Pauling. This gives a dimensionless quantity, commonly referred to as the Pauling scale, on a relative scale running from around 0.7 to 3.98 (hydrogen = 2.20). When other methods of calculation are used, it is conventional (although not obligatory) to quote the results on a scale that covers the same range of numerical values: this is known as an electronegativity in Pauling units. As it is usually calculated, electronegativity is not a property of an atom alone, but rather a property of an atom in a molecule. Properties of a free atom include ionization energy and electron affinity. It is to be expected that the electronegativity of an element will vary with its chemical environment, but it is usually considered to be a transferable property, that is to say that similar values will be valid in a variety of situations.On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more ""pull"" it will have on electrons) and the number/location of other electrons present in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result the less positive charge they will experience—both because of their increased distance from the nucleus, and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).The opposite of electronegativity is electropositivity: a measure of an element's ability to donate electrons.Caesium is the least electronegative element in the periodic table (=0.79), while fluorine is most electronegative (=3.98). (Francium and caesium were originally assigned both assigned 0.7; caesium's value was later refined to 0.79, but no experimental data allows a similar refinement for francium. However, francium's ionization energy is known to be slightly higher than caesium's, in accordance with the relativistic stabilization of the 7s orbital, and this in turn implies that caesium is in fact more electronegative than francium.)