DOE Chemistry 1
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
Computational investigations of the electronic structure of molecular
... challenging computationally. The main result of the study confirms that the bonding is ionic and that there are few differences in the behaviour of the transition metals. In the fourth chapter the electronic spectrum of NpO22+, NpO2Cl42- and NpO2(OH)42- is calculated using time dependent DFT. TDDFT ...
... challenging computationally. The main result of the study confirms that the bonding is ionic and that there are few differences in the behaviour of the transition metals. In the fourth chapter the electronic spectrum of NpO22+, NpO2Cl42- and NpO2(OH)42- is calculated using time dependent DFT. TDDFT ...
Document
... flame test. When it was dissolved in water and reacted with NaOCl, a purple color appeared. The unknown is: a. b. c. d. ...
... flame test. When it was dissolved in water and reacted with NaOCl, a purple color appeared. The unknown is: a. b. c. d. ...
Oxidation numbers
... When some reactions occur, an exchange of electrons takes place. It is this exchange of electrons that leads to the change in charge that we noted in grade 10 (chapter 18, reactions in aqueous solution). When an atom gains electrons it becomes more negative and when it loses electrons it becomes mor ...
... When some reactions occur, an exchange of electrons takes place. It is this exchange of electrons that leads to the change in charge that we noted in grade 10 (chapter 18, reactions in aqueous solution). When an atom gains electrons it becomes more negative and when it loses electrons it becomes mor ...
Regents Review Live
... React violently with water to form hydrogen gas and a strong base: 2 Na (s) + H2O (l) 2 NaOH (aq) + H2 (g) 1 valence electron Form +1 ion by losing that valence electron Form oxides like Na2O, Li2O, K2O ...
... React violently with water to form hydrogen gas and a strong base: 2 Na (s) + H2O (l) 2 NaOH (aq) + H2 (g) 1 valence electron Form +1 ion by losing that valence electron Form oxides like Na2O, Li2O, K2O ...
Hybridization of atomic orbitals
... equivalent. The CH4 molecule is the most cited molecule to have a tetrahedral shape. Other molecules and ions having tetrahedral shapes are SiO44-, SO42-, As are the cases with sp2, hybrid orbitals, one or two of the sp3 hybrid orbitals may be occupied by non-bonding electrons. Water and ammonia are ...
... equivalent. The CH4 molecule is the most cited molecule to have a tetrahedral shape. Other molecules and ions having tetrahedral shapes are SiO44-, SO42-, As are the cases with sp2, hybrid orbitals, one or two of the sp3 hybrid orbitals may be occupied by non-bonding electrons. Water and ammonia are ...
chemistry - Ethiopian Ministry of Education
... 2. Why do soaps have a cleansing action? 3. What makes petrol/CNG (compressed natural gas) a better fuel than wood? 4. Is it possible to use wires made of rubber for conduction of electricity? 5. What properties of cement, iron and stone make them suitable for construction of houses, etc. but not fo ...
... 2. Why do soaps have a cleansing action? 3. What makes petrol/CNG (compressed natural gas) a better fuel than wood? 4. Is it possible to use wires made of rubber for conduction of electricity? 5. What properties of cement, iron and stone make them suitable for construction of houses, etc. but not fo ...
RedOx notes:
... Continue with elements picking their preferred charges (work from outside columns to the inner “valley of confusion”) until there is only one left; if the element is last to choose it must have the charge that makes everything else sum to zero. ...
... Continue with elements picking their preferred charges (work from outside columns to the inner “valley of confusion”) until there is only one left; if the element is last to choose it must have the charge that makes everything else sum to zero. ...
Chemistry 101L
... appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that the measurements were made. For example, it is easier to subtract the initial volume from the final vo ...
... appropriate, have room for classmates’ data. Now organize your list into things that are similar or data that should be compared. Tables columns/rows do not have to be listed in the same order that the measurements were made. For example, it is easier to subtract the initial volume from the final vo ...
BASIC CONCEPTS OF CHEMISTRY
... bodies extracted from the space, is called the system . If no mass and heat transfer exists between the system and the surrounding environment, the system is called isolated. If this condition is not met, then the system is called open. If the system is only possible for the heat transfer , it is ca ...
... bodies extracted from the space, is called the system . If no mass and heat transfer exists between the system and the surrounding environment, the system is called isolated. If this condition is not met, then the system is called open. If the system is only possible for the heat transfer , it is ca ...
Chemistry Essentials For Dummies
... John’s area of specialty is chemical education, especially at the pre-high school level. For the last several years, he has been the co-editor (along with one of his former students) of the Chemistry for Kids feature of The Journal of Chemical Education. He has authored Chemistry For Dummies and Che ...
... John’s area of specialty is chemical education, especially at the pre-high school level. For the last several years, he has been the co-editor (along with one of his former students) of the Chemistry for Kids feature of The Journal of Chemical Education. He has authored Chemistry For Dummies and Che ...
File
... 8 Positive ions are formed when the vaporised atom or molecule is bombarded with fast-moving electrons. The kinetic energy of these electrons is great enough to cause the removal of an electron from the outermost orbital of the atom or of one of the bonding electrons in the molecule. e ...
... 8 Positive ions are formed when the vaporised atom or molecule is bombarded with fast-moving electrons. The kinetic energy of these electrons is great enough to cause the removal of an electron from the outermost orbital of the atom or of one of the bonding electrons in the molecule. e ...
Ions
... Nonmetals, Group 5A (15), Group 6A (16), and Group 7A (17) • have high ionization energies, they don’t tend to lose electrons but instead gain electrons • readily gain one or more valence electrons to form ions with a negative charge. • gain electrons until they have the same number of valence elect ...
... Nonmetals, Group 5A (15), Group 6A (16), and Group 7A (17) • have high ionization energies, they don’t tend to lose electrons but instead gain electrons • readily gain one or more valence electrons to form ions with a negative charge. • gain electrons until they have the same number of valence elect ...
Alberta Chemistry 20-30 Sample CAB Questions - McGraw
... central atom is surrounded by three shared pairs of electrons. According to VSEPR theory, these electrons should be as far apart as possible, so that the electrostatic force of repulsion between them is the minimum. In such a case, the three electron pairs arrange themselves in trigonal planar geome ...
... central atom is surrounded by three shared pairs of electrons. According to VSEPR theory, these electrons should be as far apart as possible, so that the electrostatic force of repulsion between them is the minimum. In such a case, the three electron pairs arrange themselves in trigonal planar geome ...
Chapter 6 Ionic and Molecular Compounds
... the cause of this lack of reactivity? • Examination of their electron configurations reveals that the noble gases either have 1. an outermost electron energy level that is completely filled with electrons (He = 2 e- in the 1st energy level, Ne = 8 e- in the 2nd energy level) General, Organic, and Bi ...
... the cause of this lack of reactivity? • Examination of their electron configurations reveals that the noble gases either have 1. an outermost electron energy level that is completely filled with electrons (He = 2 e- in the 1st energy level, Ne = 8 e- in the 2nd energy level) General, Organic, and Bi ...
ChemQuest 1 Information: Qualitative vs. Quantitative Critical
... 5. How are compounds different from mixtures? Compounds are formed by a chemical change (i.e. two hydrogen and one oxygen atom bonding to form a water molecule), but mixtures are formed by a physical change (i.e. stirring salt and water together. 6. How are pure substances different from mixtures? P ...
... 5. How are compounds different from mixtures? Compounds are formed by a chemical change (i.e. two hydrogen and one oxygen atom bonding to form a water molecule), but mixtures are formed by a physical change (i.e. stirring salt and water together. 6. How are pure substances different from mixtures? P ...
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... its reactivity to the reactivity of other elements in the table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.2 – Predict characteristics of an atom or ...
... its reactivity to the reactivity of other elements in the table. Anchor: CHEM.A.2.2 – Describe the behavior of electrons in atoms. Eligible Content CHEM.A.2.2.2 – Predict characteristics of an atom or ...
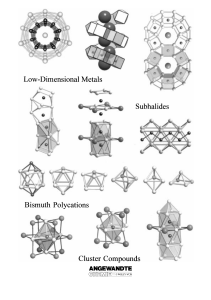
From the Metal to the Molecule
... establishing the rules and limitations of using localized bonding concepts to understand the binding in a large collection of atoms in the metallic state. Since practically all the typical physical properties of a metal depend on the almost free movement of electrons in the solid, the question arise ...
... establishing the rules and limitations of using localized bonding concepts to understand the binding in a large collection of atoms in the metallic state. Since practically all the typical physical properties of a metal depend on the almost free movement of electrons in the solid, the question arise ...
Introduction to Chemistry
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
... 2. I can calculate the pH of a solution. 3. I can write a neutralization reaction between an acid and base. 4. I can calculate the concentration of an acid or base from data collected in a titration. Unit 9: Energy of Chemical Changes Nature of Science Goal—Science provides technology to improve liv ...
Concept Development Studies in Chemistry
... In contrast, the compound we call iron rust bears little resemblance to elemental iron: iron rust does not exhibit elemental iron's color, density, hardness, magnetism, etc. Since the properties of the elements are not maintained by the compound, then the compound must not be a simple mixture of the ...
... In contrast, the compound we call iron rust bears little resemblance to elemental iron: iron rust does not exhibit elemental iron's color, density, hardness, magnetism, etc. Since the properties of the elements are not maintained by the compound, then the compound must not be a simple mixture of the ...
Table of Contents - slccscience`s Home Page
... and its compounds. Organic chemistry is the study of carbon and its compounds. Since there are 117 known elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a sepa ...
... and its compounds. Organic chemistry is the study of carbon and its compounds. Since there are 117 known elements, it often seems odd that an entire branch of chemistry is devoted to a single element and its compounds while the other 116 elements and their compounds are all lumped together in a sepa ...
Chapter 3: Ionic and Covalent Compounds Chapter 3: Ionic and
... 80. Anions are formed when a neutral atom gains one or more electrons. A) True B) False Ans: A Difficulty: Easy 81. The (II) in the name of the ionic compound lead (II) acetate specifically indicates that there are two lead ions present in the compound. A) True B) False Ans: B Difficulty: Medium 82. ...
... 80. Anions are formed when a neutral atom gains one or more electrons. A) True B) False Ans: A Difficulty: Easy 81. The (II) in the name of the ionic compound lead (II) acetate specifically indicates that there are two lead ions present in the compound. A) True B) False Ans: B Difficulty: Medium 82. ...
Oxidation-Reduction Reactions
... • How are cations and anions formed? • What determines whether a covalent bond between two atoms is represented by equal sharing or unequal sharing of the bonding electrons? An iron shovel left out in the rain will eventually begin to rust. What is happening to the shovel, and can anything be done t ...
... • How are cations and anions formed? • What determines whether a covalent bond between two atoms is represented by equal sharing or unequal sharing of the bonding electrons? An iron shovel left out in the rain will eventually begin to rust. What is happening to the shovel, and can anything be done t ...
PDF - mockies – Mockiesgateacademy
... Eg. Consider the following reaction 2 H2 + O2 → 2H2O In this reaction one molecule of oxygen reacts with two molecules of hydrogen. So it would be desirable to take the molecules of H2 and oxygen in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and mole ...
... Eg. Consider the following reaction 2 H2 + O2 → 2H2O In this reaction one molecule of oxygen reacts with two molecules of hydrogen. So it would be desirable to take the molecules of H2 and oxygen in the ratio 2:1, so that the reactants are completely consumed during the reaction. But atoms and mole ...
Review Study Guide for the Final
... What is it called when you have more electrons than protons? ...
... What is it called when you have more electrons than protons? ...
Electronegativity

Electronegativity, symbol χ, is a chemical property that describes the tendency of an atom or a functional group to attract electrons (or electron density) towards itself. An atom's electronegativity is affected by both its atomic number and the distance at which its valence electrons reside from the charged nucleus. The higher the associated electronegativity number, the more an element or compound attracts electrons towards it. The term ""electronegativity"" was introduced by Jöns Jacob Berzelius in 1811,though the concept was known even before that and was studied by many chemists including Avogadro.In spite of its long history, an accurate scale of electronegativity had to wait till 1932, when Linus Pauling proposed an electronegativity scale, which depends on bond energies, as a development of valence bond theory. It has been shown to correlate with a number of other chemical properties. Electronegativity cannot be directly measured and must be calculated from other atomic or molecular properties. Several methods of calculation have been proposed, and although there may be small differences in the numerical values of the electronegativity, all methods show the same periodic trends between elements. The most commonly used method of calculation is that originally proposed by Linus Pauling. This gives a dimensionless quantity, commonly referred to as the Pauling scale, on a relative scale running from around 0.7 to 3.98 (hydrogen = 2.20). When other methods of calculation are used, it is conventional (although not obligatory) to quote the results on a scale that covers the same range of numerical values: this is known as an electronegativity in Pauling units. As it is usually calculated, electronegativity is not a property of an atom alone, but rather a property of an atom in a molecule. Properties of a free atom include ionization energy and electron affinity. It is to be expected that the electronegativity of an element will vary with its chemical environment, but it is usually considered to be a transferable property, that is to say that similar values will be valid in a variety of situations.On the most basic level, electronegativity is determined by factors like the nuclear charge (the more protons an atom has, the more ""pull"" it will have on electrons) and the number/location of other electrons present in the atomic shells (the more electrons an atom has, the farther from the nucleus the valence electrons will be, and as a result the less positive charge they will experience—both because of their increased distance from the nucleus, and because the other electrons in the lower energy core orbitals will act to shield the valence electrons from the positively charged nucleus).The opposite of electronegativity is electropositivity: a measure of an element's ability to donate electrons.Caesium is the least electronegative element in the periodic table (=0.79), while fluorine is most electronegative (=3.98). (Francium and caesium were originally assigned both assigned 0.7; caesium's value was later refined to 0.79, but no experimental data allows a similar refinement for francium. However, francium's ionization energy is known to be slightly higher than caesium's, in accordance with the relativistic stabilization of the 7s orbital, and this in turn implies that caesium is in fact more electronegative than francium.)