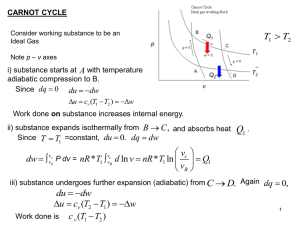
Maxwell Relations
... respect to its natural variables, we can refer back to the original equation of state and define, in this example, T and P. ...
... respect to its natural variables, we can refer back to the original equation of state and define, in this example, T and P. ...
NOTES on THERMODYNAMICS - University of Utah Physics
... ⋆ As a phenomenological description, it is based on a number of empirical observations which are summarized by the laws of thermodynamics. A coherent logical and mathe matical structure is then constructed on the basis of these observations, which leads to a variety of useful concepts, and to testa ...
... ⋆ As a phenomenological description, it is based on a number of empirical observations which are summarized by the laws of thermodynamics. A coherent logical and mathe matical structure is then constructed on the basis of these observations, which leads to a variety of useful concepts, and to testa ...
Chapter 1 Introduction and Basic Concepts Study Guide in PowerPoint
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
Chapter 1
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
dx cx dx and x - Cameron University
... gas 12.2 Apply heat, work, internal energy and enthalpy concepts to the reversible constant-P (or constant-V) process in a perfect gas 12.3 Apply heat, work, internal energy and enthalpy concepts to the reversible adiabatic P-V change in a perfect gas 12.4 Apply heat, work, internal energy and entha ...
... gas 12.2 Apply heat, work, internal energy and enthalpy concepts to the reversible constant-P (or constant-V) process in a perfect gas 12.3 Apply heat, work, internal energy and enthalpy concepts to the reversible adiabatic P-V change in a perfect gas 12.4 Apply heat, work, internal energy and entha ...
0 Quarter Three Assessment Review - SRHSchem
... the solid is completely dissolve, the water temperature drops to 15.2°C. a. Is this process endothermic or exothermic? Explain. – The temperature of the water decreases, so the dissolving process must have absorbed that heat, ...
... the solid is completely dissolve, the water temperature drops to 15.2°C. a. Is this process endothermic or exothermic? Explain. – The temperature of the water decreases, so the dissolving process must have absorbed that heat, ...
Student Study Guide PPT Chapter 01
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
Chapter 01 - McGraw Hill Higher Education
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
... The study of thermodynamics is concerned with the ways energy is stored within a body and how energy transformations, which involve heat and work, may take place. One of the most fundamental laws of nature is the conservation of energy principle. It simply states that during an energy interaction, e ...
Thermochemistry
... the car and the air around the car). – Without this limitation, cars could easily exceed 100 mpg. • Heat, denoted with the symbol q, will always flow when there is a temperature difference between the system and the surroundings (from high temp to low temp). ...
... the car and the air around the car). – Without this limitation, cars could easily exceed 100 mpg. • Heat, denoted with the symbol q, will always flow when there is a temperature difference between the system and the surroundings (from high temp to low temp). ...
Chapter 22-1 - UCF College of Sciences
... even if the system may have followed an irreversible path • The meaningful quantity is the change in entropy and not the entropy itself • For a finite process, f ...
... even if the system may have followed an irreversible path • The meaningful quantity is the change in entropy and not the entropy itself • For a finite process, f ...
Table of Content
... The word “Thermodynamics” originates from its Greek roots (therme, heat; dynamis, force). As a subject it is concerned with quantification of inter-relation between energy and the change of state of any real world system. The extent of such change of state due to transfer of energy to or from the sy ...
... The word “Thermodynamics” originates from its Greek roots (therme, heat; dynamis, force). As a subject it is concerned with quantification of inter-relation between energy and the change of state of any real world system. The extent of such change of state due to transfer of energy to or from the sy ...
GCE Physics - Thermodynamics Notes Word Document
... Has work been done or by the system? Not during the reaction. Almost immediately afterwards, the vessel may shatter and the pieces acquire kinetic energy as the expanding gas does work on them. So eq.3 (any version: 3a, 3b or 3c) shows that during the reaction there is no change in the internal ener ...
... Has work been done or by the system? Not during the reaction. Almost immediately afterwards, the vessel may shatter and the pieces acquire kinetic energy as the expanding gas does work on them. So eq.3 (any version: 3a, 3b or 3c) shows that during the reaction there is no change in the internal ener ...
Heat

In physics, heat is energy in a process of transfer between a system and its surroundings, other than as work or with the transfer of matter. When there is a suitable physical pathway, heat flows from a hotter body to a colder one. The pathway can be direct, as in conduction and radiation, or indirect, as in convective circulation.Because it refers to a process of transfer between two systems, the system of interest, and its surroundings considered as a system, heat is not a state or property of a single system. If heat transfer is slow and continuous, so that the temperature of the system of interest remains well defined, it can sometimes be described by a process function.Kinetic theory explains heat as a macroscopic manifestation of the motions and interactions of microscopic constituents such as molecules and photons.In calorimetry, sensible heat is defined with respect to a specific chosen state variable of the system, such as pressure or volume. Sensible heat transferred into or out of the system under study causes change of temperature while leaving the chosen state variable unchanged. Heat transfer that occurs with the system at constant temperature and that does change that particular state variable is called latent heat with respect to that variable. For infinitesimal changes, the total incremental heat transfer is then the sum of the latent and sensible heat increments. This is a basic paradigm for thermodynamics, and was important in the historical development of the subject.The quantity of energy transferred as heat is a scalar expressed in an energy unit such as the joule (J) (SI), with a sign that is customarily positive when a transfer adds to the energy of a system. It can be measured by calorimetry, or determined by calculations based on other quantities, relying on the first law of thermodynamics.