Introduction to Magnetism - Appoquinimink High School
... This lead to a world-wide search for the links between electricity and magnetism. We will be discussing all of these links later this week ...
... This lead to a world-wide search for the links between electricity and magnetism. We will be discussing all of these links later this week ...
Magnetic Materials Background: 12. Other Materials
... commercially. This section will look at magnetostrictive and magnetoresistive materials. Magnetostriction: Most magnetic materials exhibit magnetostriction, which is a change in physical dimension as a result of magnetic ordering. There are two types of magnetostriction: spontaneous magnetostriction ...
... commercially. This section will look at magnetostrictive and magnetoresistive materials. Magnetostriction: Most magnetic materials exhibit magnetostriction, which is a change in physical dimension as a result of magnetic ordering. There are two types of magnetostriction: spontaneous magnetostriction ...
Chapter 8: Electromagnetism End of Chapter Questions
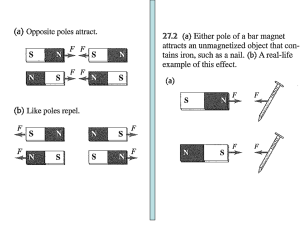
... 2. The force between electrically charged particles depends on the magnitude of charge, the distance of separation, and what else? 3. What is the source of magnetic force? 4. Is the rule for the interaction between magnetic poles similar to the rule for the interaction between electrically charged p ...
... 2. The force between electrically charged particles depends on the magnitude of charge, the distance of separation, and what else? 3. What is the source of magnetic force? 4. Is the rule for the interaction between magnetic poles similar to the rule for the interaction between electrically charged p ...
Unit 6 Magnetism
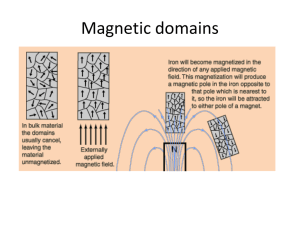
... • Groups of atoms with aligned magnetic poles are called magnetic domains • This allows the object to behave like a magnetic with a north and south pole • Ex: Placing a magnet against an iron nail will cause all the atoms to orient themselves in the same direction and temporarily cause the nail to a ...
... • Groups of atoms with aligned magnetic poles are called magnetic domains • This allows the object to behave like a magnetic with a north and south pole • Ex: Placing a magnet against an iron nail will cause all the atoms to orient themselves in the same direction and temporarily cause the nail to a ...
Magnetochemistry

Magnetochemistry is concerned with the magnetic properties of chemical compounds. Magnetic properties arise from the spin and orbital angular momentum of the electrons contained in a compound. Compounds are diamagnetic when they contain no unpaired electrons. Molecular compounds that contain one or more unpaired electrons are paramagnetic. The magnitude of the paramagnetism is expressed as an effective magnetic moment, μeff. For first-row transition metals the magnitude of μeff is, to a first approximation, a simple function of the number of unpaired electrons, the spin-only formula. In general, spin-orbit coupling causes μeff to deviate from the spin-only formula. For the heavier transition metals, lanthanides and actinides, spin-orbit coupling cannot be ignored. Exchange interaction can occur in clusters and infinite lattices, resulting in ferromagnetism, antiferromagnetism or ferrimagnetism depending on the relative orientations of the individual spins.