ATOMS, MOLECULES, AND IONS
... The Law of Multiple Proportions — if two elements A and B combine to form more than one compound, then the mass of B that combines with the mass of A is a ratio of small whole numbers. (e.g. CO, CO2 ) ...
... The Law of Multiple Proportions — if two elements A and B combine to form more than one compound, then the mass of B that combines with the mass of A is a ratio of small whole numbers. (e.g. CO, CO2 ) ...
Science 10 Chem - Holy Trinity Academy
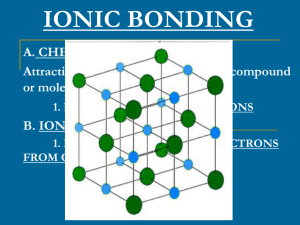
... To see if an ionic compound will be solid look at “Solubility of Ionic Compounds in Water” in Appendix D o (-) ion on top o (+) ion on bottom - highly solubility = compound dissolve o low solubility = compound form solid * Practice problems on page 278 ...
... To see if an ionic compound will be solid look at “Solubility of Ionic Compounds in Water” in Appendix D o (-) ion on top o (+) ion on bottom - highly solubility = compound dissolve o low solubility = compound form solid * Practice problems on page 278 ...
X-rays, Laser
... diameter rod. The ruby has very broad absorption band in the visual spectrum, at 400 and 550 nm. The E3 level is excited by optical pumping, typically by a xenon flashtube. After that from E3 level, to E2 level there is a non radiative transition. The E2 level is a metastable state its life time is ...
... diameter rod. The ruby has very broad absorption band in the visual spectrum, at 400 and 550 nm. The E3 level is excited by optical pumping, typically by a xenon flashtube. After that from E3 level, to E2 level there is a non radiative transition. The E2 level is a metastable state its life time is ...
LN_atoms_etc
... Experiments by Thomson and Millikan confirmed the existence of electrons as the negatively charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the center of an atom is a positively ...
... Experiments by Thomson and Millikan confirmed the existence of electrons as the negatively charged particles within an atom. Electrons have a charge of e = 1.6021773 10–19 C and a mass of 9.109390 10–31 kg. Later experiments by Rutherford determined that at the center of an atom is a positively ...
HW / Unit 2
... a. The physical properties of Ti are expected to be intermediate between those of Sc and V. b. The formula of the chloride of Ti is expected to be the same as those of Sc and V. c. The formula of the oxide of Ti is expected to be the same as those of Zr and Hf. d. There are fewer metallic elements t ...
... a. The physical properties of Ti are expected to be intermediate between those of Sc and V. b. The formula of the chloride of Ti is expected to be the same as those of Sc and V. c. The formula of the oxide of Ti is expected to be the same as those of Zr and Hf. d. There are fewer metallic elements t ...
PAP Chemistry - Fall Final Review
... 7. When is a bright-line spectrum produced by an atom? IE – How does an atom give off color (especially when burned)? The resting state or the ground state is when the electron is closest to the nucleus. It will jump to outer levels gaining energy and this is called the excited state. When the elect ...
... 7. When is a bright-line spectrum produced by an atom? IE – How does an atom give off color (especially when burned)? The resting state or the ground state is when the electron is closest to the nucleus. It will jump to outer levels gaining energy and this is called the excited state. When the elect ...
Bonding
... 6. Also, check the number of electrons in your drawing with the number of electrons from step 2. If you have more electrons in the drawing than in step 2, you must make double or triple bonds. If you have less electrons in the drawing than in step 2, you made a mistake! ...
... 6. Also, check the number of electrons in your drawing with the number of electrons from step 2. If you have more electrons in the drawing than in step 2, you must make double or triple bonds. If you have less electrons in the drawing than in step 2, you made a mistake! ...
The Periodic table
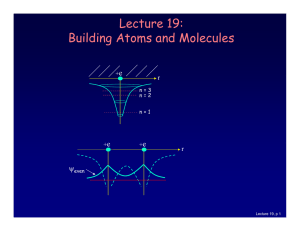
... A quantized property is a property that can have only certain values. The energy of an electron is quantized, only certain behavior patterns are allowed. ...
... A quantized property is a property that can have only certain values. The energy of an electron is quantized, only certain behavior patterns are allowed. ...
IONIZATION METHODS IN MASS SPECTROMETRY
... Chemical ionisation has become very popular for structural elucidation since it was first developed in the mid-1960s. Here, the concept of ionisation relies on the interaction of ions with neutral molecules and the further production of new ions. In general, the amount of fragments ions produced is ...
... Chemical ionisation has become very popular for structural elucidation since it was first developed in the mid-1960s. Here, the concept of ionisation relies on the interaction of ions with neutral molecules and the further production of new ions. In general, the amount of fragments ions produced is ...
Unit 2 - Biochemistry Notes
... Molecule – when two or more atoms bond. CO2 , O2 , H2 and H2O are all molecules. Compound – when different elements combine. CO2 and H2O are molecules, but they are also compounds because they are molecules containing more than one element. ...
... Molecule – when two or more atoms bond. CO2 , O2 , H2 and H2O are all molecules. Compound – when different elements combine. CO2 and H2O are molecules, but they are also compounds because they are molecules containing more than one element. ...
Common Chemical Formula List
... with chemical symbols, as then you will be able to see how many atoms of each type are in each chemical. Example 1 Unbalanced Equation:- C3H8 + O2 ---> H2O + CO2 There are three carbons on the left, but only one on the right. There are eight hydrogens on the left but only two on the right. There are ...
... with chemical symbols, as then you will be able to see how many atoms of each type are in each chemical. Example 1 Unbalanced Equation:- C3H8 + O2 ---> H2O + CO2 There are three carbons on the left, but only one on the right. There are eight hydrogens on the left but only two on the right. There are ...
+1/2 and
... The sample is here passive, a light source is necessary for the working. A part of the light will be absorbed by the sample. The monochromator resolve the light, the detector transforms the optical signal to electric one. The speciality of the absorption spectrometer is the necessity of a reference. ...
... The sample is here passive, a light source is necessary for the working. A part of the light will be absorbed by the sample. The monochromator resolve the light, the detector transforms the optical signal to electric one. The speciality of the absorption spectrometer is the necessity of a reference. ...
Acrobat Distiller, Job 21
... In summary, an “empirical” analysis of the covalent bond in H2 has been presented based on the virial theorem and a Morse molecular energy function parameterized using spectroscopic data. This analysis has shown that the role of electron kinetic energy in covalent bond formation runs counter to conv ...
... In summary, an “empirical” analysis of the covalent bond in H2 has been presented based on the virial theorem and a Morse molecular energy function parameterized using spectroscopic data. This analysis has shown that the role of electron kinetic energy in covalent bond formation runs counter to conv ...
الرقم الجامعي
... an s orbital on atom A and a px orbital on atom B ---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------- ...
... an s orbital on atom A and a px orbital on atom B ---------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------------- ...
TDR XFEL workshop series Atomic, molecular and cluster physics
... are very important for modern fundamental and applied physics. So, the explorations of non-linear processes of the x-ray photons interaction with atom, particularly, have direct relation to solving the problem of x-ray laser design [3]. In this paper we consider the problem of theoretical descriptio ...
... are very important for modern fundamental and applied physics. So, the explorations of non-linear processes of the x-ray photons interaction with atom, particularly, have direct relation to solving the problem of x-ray laser design [3]. In this paper we consider the problem of theoretical descriptio ...
Topic 1 Test - A-Level Chemistry
... An atom has half as many protons as an atom of 28Si and also has six fewer neutrons than an atom of 28Si. Give the symbol, including the mass number and the atomic number, of this atom. ...
... An atom has half as many protons as an atom of 28Si and also has six fewer neutrons than an atom of 28Si. Give the symbol, including the mass number and the atomic number, of this atom. ...
PowerPoint - Astronomy at Swarthmore College
... the magnetic star 1 Ori C is very young (<400,000 yr). It has a relatively strong wind (dM/dt 10-7 M, v 2500 km s-1) in addition to a strong dipole field (Donati et al. 2002). Its strong, hard, variable X-ray emission suggests it may be the prototype of a new class of stellar X-ray source: an ...
... the magnetic star 1 Ori C is very young (<400,000 yr). It has a relatively strong wind (dM/dt 10-7 M, v 2500 km s-1) in addition to a strong dipole field (Donati et al. 2002). Its strong, hard, variable X-ray emission suggests it may be the prototype of a new class of stellar X-ray source: an ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.