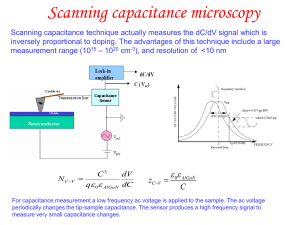
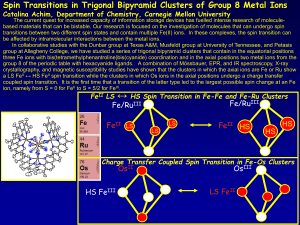
Elements, basic principles, periodic table
... Ionization and Ionization Energy (aka ionization potential): Ionization energy measures how easy or hard it is to remove an electron from an element or ion. Energies of filled electronic orbitals give rise to common oxidation states for individual elements. Electronic structure determines ionic char ...
... Ionization and Ionization Energy (aka ionization potential): Ionization energy measures how easy or hard it is to remove an electron from an element or ion. Energies of filled electronic orbitals give rise to common oxidation states for individual elements. Electronic structure determines ionic char ...
CHEM 1211K Test IV MULTIPLE CHOICE. (3 points each) 1) The
... among different I2 molecules in the solid? A) London dispersion forces B) ionic-dipole interactions C) dipole-dipole attractions D) dipole-dipole rejections E) covalent-ionic interactions 22) The heat of fusion of water is 6.01 kJ/mol. The heat capacity of liquid water is 75.2 J/mol(K) the conversio ...
... among different I2 molecules in the solid? A) London dispersion forces B) ionic-dipole interactions C) dipole-dipole attractions D) dipole-dipole rejections E) covalent-ionic interactions 22) The heat of fusion of water is 6.01 kJ/mol. The heat capacity of liquid water is 75.2 J/mol(K) the conversio ...
Atomic Systems and Bonding
... & therefore use ATOMIC Structure Primary & secondary Electron configurations BONDING ...
... & therefore use ATOMIC Structure Primary & secondary Electron configurations BONDING ...
Fall Exam 4 - Chemistry - University of Kentucky
... The total number of molecular orbitals formed does not always equal the number of atomic orbitals combined. In H2 molecules, the two 1s orbitals combine constructively, which results in one bonding orbital and one nonbonding orbital Electrons placed in antibonding orbitals stabilize the species. Whe ...
... The total number of molecular orbitals formed does not always equal the number of atomic orbitals combined. In H2 molecules, the two 1s orbitals combine constructively, which results in one bonding orbital and one nonbonding orbital Electrons placed in antibonding orbitals stabilize the species. Whe ...
Section 3.6
... quantum number might be n = 2, l = 1, ml = –1, and ms = +1/2 . This might describe an electron in a hydrogen atom in an “excited” state. 7. For each principal quantum number from n = 1 to n = 3 (see Table 4), there can be 2, 8, and 18 different electron descriptions. 8. In the development of scienti ...
... quantum number might be n = 2, l = 1, ml = –1, and ms = +1/2 . This might describe an electron in a hydrogen atom in an “excited” state. 7. For each principal quantum number from n = 1 to n = 3 (see Table 4), there can be 2, 8, and 18 different electron descriptions. 8. In the development of scienti ...
Chapter 2 (Hill/Petrucci/McCreary/Perry This chapter deals with
... Chemical compounds have constant, unvarying compositions with respect to weights of elements which make them up. Law of Constant Composition (Definite Proportions) "In a compound, elements are always present in a definite proportion by weight." Thus, water, a compound is always 11 parts H and 89 par ...
... Chemical compounds have constant, unvarying compositions with respect to weights of elements which make them up. Law of Constant Composition (Definite Proportions) "In a compound, elements are always present in a definite proportion by weight." Thus, water, a compound is always 11 parts H and 89 par ...
Total view of the AFM
... that is heated to an extremely high temperature of ~2500 C to make electrons have high enough energy to overcome the surface work function of ~4.5 eV – To get higher electron current stable materials with lower work function is preferred. LaB6 as polycrystalline powder is used to reduce the work fun ...
... that is heated to an extremely high temperature of ~2500 C to make electrons have high enough energy to overcome the surface work function of ~4.5 eV – To get higher electron current stable materials with lower work function is preferred. LaB6 as polycrystalline powder is used to reduce the work fun ...
Project Title : X-RAY LASER RESEARCH
... A molecule is chiral, having left and right handed types, if one type cannot be rotated so that it is superposed on the other. These molecules have very similar chemical and physical properties such as having the same infrared(IR) and nuclear magnetic resonance (NMR) spectra. However, left and right ...
... A molecule is chiral, having left and right handed types, if one type cannot be rotated so that it is superposed on the other. These molecules have very similar chemical and physical properties such as having the same infrared(IR) and nuclear magnetic resonance (NMR) spectra. However, left and right ...
Standards Practice
... covalent or metallic bonds or by exchanging electrons to form ionic bonds. 1. Potassium (K) and chlorine (Cl) form a(n) A. covalent bond. B. hydrogen bond. C. ionic bond. D. metallic bond. 2. When atoms combine to form a molecule by sharing electrons, what type of bonds are ...
... covalent or metallic bonds or by exchanging electrons to form ionic bonds. 1. Potassium (K) and chlorine (Cl) form a(n) A. covalent bond. B. hydrogen bond. C. ionic bond. D. metallic bond. 2. When atoms combine to form a molecule by sharing electrons, what type of bonds are ...
Solar Wind - International School of Space Science
... energy tail can increase the solar wind speed and may account for the fast solar wind. • Other models predict that cyclotron resonance heating occurring at the source may account for the bulk acceleration of the solar wind. • Models have also included spiral interplanetary magnetic field. • A few pa ...
... energy tail can increase the solar wind speed and may account for the fast solar wind. • Other models predict that cyclotron resonance heating occurring at the source may account for the bulk acceleration of the solar wind. • Models have also included spiral interplanetary magnetic field. • A few pa ...
the effect of an soft X-ray source near the compact object in LS 5039
... infc by the large density of stellar radiation field GeV: IC anisotropy is dominant CS(superior) MS At supc, flux is larger than GeV supc by head-on collision of IC scattering X-ray: e± number variation by IC cooling At periastron, the e± number CS(superior) MS in steady state is smaller than X-ray ...
... infc by the large density of stellar radiation field GeV: IC anisotropy is dominant CS(superior) MS At supc, flux is larger than GeV supc by head-on collision of IC scattering X-ray: e± number variation by IC cooling At periastron, the e± number CS(superior) MS in steady state is smaller than X-ray ...
Ionic and Covalent Compounds: Naming, Formulas, Properties 1
... Fe2+). When such ions are formed, the transition metal atom loses its two 4s electrons (3d electrons are not lost). (In fact, whenever a positive ion is formed from an atom, electrons are always lost first from the subshell ...
... Fe2+). When such ions are formed, the transition metal atom loses its two 4s electrons (3d electrons are not lost). (In fact, whenever a positive ion is formed from an atom, electrons are always lost first from the subshell ...
4. bonding - New Hartford Central Schools
... Either absorb or release energy when formed Create new species with chemical and physical properties unlike constituent atoms Form because atoms want to have complete outer shells ...
... Either absorb or release energy when formed Create new species with chemical and physical properties unlike constituent atoms Form because atoms want to have complete outer shells ...
complete outlines
... Alkali metals are highly reactive with water (due to low ionization energies). Alkaline Earth metals are not nearly as reactive with water (increasing reactivity going down). Halogens have high (very negative) electron affinities. Metal oxides are basic (examples: Na2O, BaO) Non-metal oxides are aci ...
... Alkali metals are highly reactive with water (due to low ionization energies). Alkaline Earth metals are not nearly as reactive with water (increasing reactivity going down). Halogens have high (very negative) electron affinities. Metal oxides are basic (examples: Na2O, BaO) Non-metal oxides are aci ...
synoptic - chemnotes.org.uk
... Note the following definitions: Bond enthalpy – enthalpy needed to break a covalent bond (applies to a bond that only occurs in 1 molecule e.g. H-H) Mean bond enthalpy – enthalpy needed to break a covalent bond averaged over many compounds (applies to a bond that occurs in many compounds e.g. C-H) I ...
... Note the following definitions: Bond enthalpy – enthalpy needed to break a covalent bond (applies to a bond that only occurs in 1 molecule e.g. H-H) Mean bond enthalpy – enthalpy needed to break a covalent bond averaged over many compounds (applies to a bond that occurs in many compounds e.g. C-H) I ...
Chem 115 POGIL Worksheet - Week 10 Periodic Trends Why? The
... rationalized on that basis. The most commonly used measure of size of an atom is its bonding atomic radius, also called the covalent radius.1 The bonding atomic radius of an element is taken as one half the distance between the nuclei when two identical atoms are bonded together. For example, the in ...
... rationalized on that basis. The most commonly used measure of size of an atom is its bonding atomic radius, also called the covalent radius.1 The bonding atomic radius of an element is taken as one half the distance between the nuclei when two identical atoms are bonded together. For example, the in ...
Review for Chapter 6: Thermochemistry
... 17. The molecular orbital diagrams for Li2, B2, C2, N2, O2, and F2 are shown in Table 10.5. Note that 2p orbitals are lower in energy than 2p orbitals for elements with an atomic number of 7 or less. 18. Molecular orbital theory helps explain why O2 shows paramagnetic properties. This occurs becau ...
... 17. The molecular orbital diagrams for Li2, B2, C2, N2, O2, and F2 are shown in Table 10.5. Note that 2p orbitals are lower in energy than 2p orbitals for elements with an atomic number of 7 or less. 18. Molecular orbital theory helps explain why O2 shows paramagnetic properties. This occurs becau ...
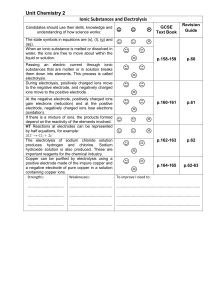
Unit_Chemistry_2_Ionic_Substances_and_Electrolysis
... Ionic Substances and Electrolysis Candidates should use their skills, knowledge and understanding of how science works: ...
... Ionic Substances and Electrolysis Candidates should use their skills, knowledge and understanding of how science works: ...
Chemical Bonding I: Lewis Theory
... This is easy to do (I hope) since I have rounded things! We show this now→→→ ...
... This is easy to do (I hope) since I have rounded things! We show this now→→→ ...
Ionic Bonding - petersonORHS
... Step 3 Add subscripts to make the charge total = zero, i.e. “criss-cross”. Note: Use parentheses if you need more than one of a polyatomic ion on other formulas. ...
... Step 3 Add subscripts to make the charge total = zero, i.e. “criss-cross”. Note: Use parentheses if you need more than one of a polyatomic ion on other formulas. ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.