Biology\Ch 2 Chemistry
... Mixtures, Solutions and Suspensions A mixture - 2 or more elements or compounds physically mixed together but not chemically combined. Ex: salt mixed with sand When something like NaCl (salt) is mixed in water and the salt breaks into Na+ and Cl- ions, they equally disperse. This is a solution. The ...
... Mixtures, Solutions and Suspensions A mixture - 2 or more elements or compounds physically mixed together but not chemically combined. Ex: salt mixed with sand When something like NaCl (salt) is mixed in water and the salt breaks into Na+ and Cl- ions, they equally disperse. This is a solution. The ...
Chemistry EOC Review
... 34. How are frequency and wavelength related? 35. Calculate the wavelength of a yellow light by a sodium lamp if the frequency of the radiation is 3.34 x 10 14 Hz. ...
... 34. How are frequency and wavelength related? 35. Calculate the wavelength of a yellow light by a sodium lamp if the frequency of the radiation is 3.34 x 10 14 Hz. ...
File
... B) Energy is released as bonds are broken, only. C) Energy is absorbed as bonds are broken, and energy is released as bonds are formed. D) Energy is absorbed as bonds are formed, and energy is released as bonds are broken. 4. What occurs in order to break the bond in a Cl2 molecule? A) B) C) D) ...
... B) Energy is released as bonds are broken, only. C) Energy is absorbed as bonds are broken, and energy is released as bonds are formed. D) Energy is absorbed as bonds are formed, and energy is released as bonds are broken. 4. What occurs in order to break the bond in a Cl2 molecule? A) B) C) D) ...
How do you tell if a molecule is paramagnetic or diamagnetic
... that all of electrons in the molecule are paired up and therefore the molecule should be diamagnetic. It turns out that molecular oxygen is paramagnetic. VB theory is not complex enough to account for this physical property and here is where molecular orbital theory comes in. The other thing that MO ...
... that all of electrons in the molecule are paired up and therefore the molecule should be diamagnetic. It turns out that molecular oxygen is paramagnetic. VB theory is not complex enough to account for this physical property and here is where molecular orbital theory comes in. The other thing that MO ...
Midterm Review Answers
... Questions 52-53nrefer to the following types of energy A) Activation energy B) Free energy C) Ionization energy D) Kinetic energy E) Lattice energy 52. The energy required to convert a ground-state atom in the gas phase to a gaseous positive ion. C 53. The energy released when gas phase ions bond t ...
... Questions 52-53nrefer to the following types of energy A) Activation energy B) Free energy C) Ionization energy D) Kinetic energy E) Lattice energy 52. The energy required to convert a ground-state atom in the gas phase to a gaseous positive ion. C 53. The energy released when gas phase ions bond t ...
Name: Date: Period: _____ Unit 2 Notes, Part 1 – The Basics of
... (think H-NOF). These bonds are usually depicted with a dotted line. Because they occur between two different molecules and not within one molecule (like ionic or covalent bonds) and they occur between partial (not full) charges, hydrogen bonds are weaker than ionic or covalent bonds. 16. Chemical re ...
... (think H-NOF). These bonds are usually depicted with a dotted line. Because they occur between two different molecules and not within one molecule (like ionic or covalent bonds) and they occur between partial (not full) charges, hydrogen bonds are weaker than ionic or covalent bonds. 16. Chemical re ...
Spectral-Type Trends: Absorption
... stellar luminosity or surface temperature. So, an X-ray emission trend would be very surprising. But stellar winds are stronger and denser for hotter and more luminous stars, so an X-ray hardness trend governed by wind attenuation is expected.) To explore the effect of wind absorption, we have devel ...
... stellar luminosity or surface temperature. So, an X-ray emission trend would be very surprising. But stellar winds are stronger and denser for hotter and more luminous stars, so an X-ray hardness trend governed by wind attenuation is expected.) To explore the effect of wind absorption, we have devel ...
Chemistry EOC Review Spring 2013
... 12. Calculate the density of a 5.0 g object that has a volume of 2.0 cm3. 13. Convert the following temperatures: a. 34C to K b. 50 K to C ...
... 12. Calculate the density of a 5.0 g object that has a volume of 2.0 cm3. 13. Convert the following temperatures: a. 34C to K b. 50 K to C ...
Partially Ionized Plasmas - Harvard
... The magnetic field sweeps excess ions into the current sheet The overabundant ions recombine to become neutrals which reduces the bottleneck associated with ion mass conservation ...
... The magnetic field sweeps excess ions into the current sheet The overabundant ions recombine to become neutrals which reduces the bottleneck associated with ion mass conservation ...
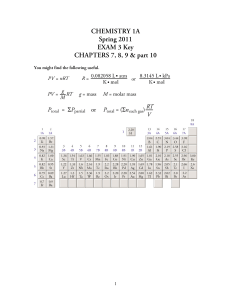
Exam 3 Key
... For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. The condition of an atom that has at least one of its electrons in orbitals that do not represent the lowest possible potential energy is called a(n) excited state. 2. A(n ...
... For each of the following, write the word, words, or number in each blank that best completes each sentence. (2 points each) 1. The condition of an atom that has at least one of its electrons in orbitals that do not represent the lowest possible potential energy is called a(n) excited state. 2. A(n ...
4. bonding - New Hartford Central Schools
... Either absorb or release energy when formed Create new species with chemical and physical properties unlike constituent atoms Form because atoms want to have complete outer shells ...
... Either absorb or release energy when formed Create new species with chemical and physical properties unlike constituent atoms Form because atoms want to have complete outer shells ...
Atomic structure
... 1.In 1897 JJ.Thomson devised an experiment with cathode rays that resulted in the discovery of the electron. 2.Up to this time it was thought that the hydrogen atom was the smallest particle in existence. Thomson demonstrated that electrons comprising cathode rays were nearly 2,000 times smaller in ...
... 1.In 1897 JJ.Thomson devised an experiment with cathode rays that resulted in the discovery of the electron. 2.Up to this time it was thought that the hydrogen atom was the smallest particle in existence. Thomson demonstrated that electrons comprising cathode rays were nearly 2,000 times smaller in ...
UNIT 3 VOCABULARY MATCHING and mole problems
... ____ 7.) part of the atom that surrounds the center; accounts for almost all of the volume of an atom ____ 8.) discovered the nucleus with the gold foil experiment ____ 9.) positively-charged subatomic particle ____ 10.) established that atoms of one element are not changed into atoms of another ele ...
... ____ 7.) part of the atom that surrounds the center; accounts for almost all of the volume of an atom ____ 8.) discovered the nucleus with the gold foil experiment ____ 9.) positively-charged subatomic particle ____ 10.) established that atoms of one element are not changed into atoms of another ele ...
Chemistry - StudyTime NZ
... must each lose or gain electrons in order to become stable. Oxygen has 8 electrons and hence an electron arrangement of 2, 6. This means it has 6 electrons in its valence shell. It must hence ...
... must each lose or gain electrons in order to become stable. Oxygen has 8 electrons and hence an electron arrangement of 2, 6. This means it has 6 electrons in its valence shell. It must hence ...
Summer Resources - mvhs
... subtracting the sum of masses of C and H from total g present of that substance. From the mass of C,H and O, we can calculate the moles of C,H and O. Then the smallest whole number ratios of these moles will give the empirical formula. Ex. A 0.6349 g sample of the unknown produced 1.603 g of CO2 and ...
... subtracting the sum of masses of C and H from total g present of that substance. From the mass of C,H and O, we can calculate the moles of C,H and O. Then the smallest whole number ratios of these moles will give the empirical formula. Ex. A 0.6349 g sample of the unknown produced 1.603 g of CO2 and ...
Gr 10 Review sheet chemistry
... The coefficient (2) applies to all elements: Therefore, 2 Ca atoms total 2 N atoms 2 O atoms The subscript 3 only follows O so we have 2 x 3 O = 6 O The subscript 2 is outside brackets, so it applies to N and O Therefore, 2 x 6 O = 12 oxygen atoms total 2 x 2 N = 4 nitrogen atoms total 2. Give the n ...
... The coefficient (2) applies to all elements: Therefore, 2 Ca atoms total 2 N atoms 2 O atoms The subscript 3 only follows O so we have 2 x 3 O = 6 O The subscript 2 is outside brackets, so it applies to N and O Therefore, 2 x 6 O = 12 oxygen atoms total 2 x 2 N = 4 nitrogen atoms total 2. Give the n ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.