final exam review packet
... C- Periodic Table-2301. I can differentiate between groups and periods on the periodic table and what is common about elements within a group. C- Periodic Table-2302. I can locate metals, non-metals and metalloids on the periodic table. C- Periodic Table-2303. I can list properties of metals, non me ...
... C- Periodic Table-2301. I can differentiate between groups and periods on the periodic table and what is common about elements within a group. C- Periodic Table-2302. I can locate metals, non-metals and metalloids on the periodic table. C- Periodic Table-2303. I can list properties of metals, non me ...
Here
... A. Definition – bonding that results from electrical attraction between (+) ions (cations) and (-) ions (anions) 1. Cations – 1+, 2+, 3+ - P.T. Trends – show valence e- and dots 2. Anions – 3-, 2-, 1- - Same as above B. Difference in e-neg >1.7 ex:// Na=.9 and Cl=3.0 C. Typically the joining of a me ...
... A. Definition – bonding that results from electrical attraction between (+) ions (cations) and (-) ions (anions) 1. Cations – 1+, 2+, 3+ - P.T. Trends – show valence e- and dots 2. Anions – 3-, 2-, 1- - Same as above B. Difference in e-neg >1.7 ex:// Na=.9 and Cl=3.0 C. Typically the joining of a me ...
The Periodic Table HL Page 1 of 3 G. Galvin Name: Periodic Table
... A sample of boron contains 18.7% 10B and 81.3% 11B. Calculate the relative atomic mass of boron. Solution: For 100 atoms, 18.7 of them have a mass of 10: 18.7 x 10 = 187 81.3 of them have a mass of 11: 81.3 x 11 = 894.3 Total mass of 100 atoms = 1081.3 Average mass of 1 atom = 1081.3 ÷ 100 = 10.813 ...
... A sample of boron contains 18.7% 10B and 81.3% 11B. Calculate the relative atomic mass of boron. Solution: For 100 atoms, 18.7 of them have a mass of 10: 18.7 x 10 = 187 81.3 of them have a mass of 11: 81.3 x 11 = 894.3 Total mass of 100 atoms = 1081.3 Average mass of 1 atom = 1081.3 ÷ 100 = 10.813 ...
CP Chemistry Final Exam Review Sheet
... 50. What is the octet rule? The octet rule states that atoms will gain, lose, or share electrons in order to get a full octet (8 e-) in the valence (outermost) shell of an atom. 51. An ion is a particle with an electrical charge created by the transfer (loss or gaining) of electrons. 52. What is a c ...
... 50. What is the octet rule? The octet rule states that atoms will gain, lose, or share electrons in order to get a full octet (8 e-) in the valence (outermost) shell of an atom. 51. An ion is a particle with an electrical charge created by the transfer (loss or gaining) of electrons. 52. What is a c ...
Molecules and Ions
... and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of each type of atom in a compound e.g. hydrogen peroxide = HO ...
... and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of each type of atom in a compound e.g. hydrogen peroxide = HO ...
Molecules and Ions
... and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of each type of atom in a compound e.g. hydrogen peroxide = HO ...
... and type of atoms) found inside any molecule: Molecular Formula: the actual number and type of atoms in a compound, e.g. hydrogen peroxide = H2O2 Empirical Formula: the lowest whole number ratio of each type of atom in a compound e.g. hydrogen peroxide = HO ...
File - Get Involved!
... – Since you are unable to use a calculator, try to eliminate choices by estimating a probable order of magnitude for the answer or simplifying the math involved ...
... – Since you are unable to use a calculator, try to eliminate choices by estimating a probable order of magnitude for the answer or simplifying the math involved ...
Inorganic Physical Methods
... NMR, interference is generated during the relaxation of nuclear spins, which gives beats of frequency which can be Fourier Transformed to generate a spectrum. In IR, the interference is generated by recombination of beams that have travelled different distances, and the path difference is Fourier Tr ...
... NMR, interference is generated during the relaxation of nuclear spins, which gives beats of frequency which can be Fourier Transformed to generate a spectrum. In IR, the interference is generated by recombination of beams that have travelled different distances, and the path difference is Fourier Tr ...
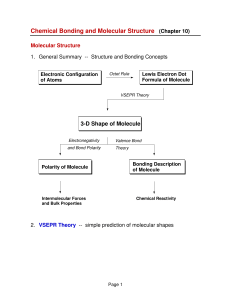
3-D Shape of Molecule
... Molecular Orbital Theory (more general but "complex") all e-'s in molecule fill up a set of molecular orbitals that are made up of linear combinations of atomic orbitals on two or more atoms MO's can be: "localized" -- ...
... Molecular Orbital Theory (more general but "complex") all e-'s in molecule fill up a set of molecular orbitals that are made up of linear combinations of atomic orbitals on two or more atoms MO's can be: "localized" -- ...
List Definition Chemistry - A Level / Secondary Chemistry Tuition
... change when 1 mole of an ionic compound is dissolved in a large excess of water under standard conditions 25C and 1 atm. Entropy (S) measures the degree of disorder in a system. The entropy of a system increases when the matter or energy in the system becomes more random in its arrangement. A syste ...
... change when 1 mole of an ionic compound is dissolved in a large excess of water under standard conditions 25C and 1 atm. Entropy (S) measures the degree of disorder in a system. The entropy of a system increases when the matter or energy in the system becomes more random in its arrangement. A syste ...
Chapter 4.1 and 4.2 - science-b
... In a chemical reaction, one substance changes to another by reorganizing the way the atoms are attached to each other ...
... In a chemical reaction, one substance changes to another by reorganizing the way the atoms are attached to each other ...
File
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
... Protons and neutrons are found in the center of the atom, called the nucleus. The electrons move about in the electron cloud that surrounds the nucleus. 46. Which subatomic particle(s) defines the identity of the atom? Protons 47. Which subatomic particle(s) determines chemical properties? electrons ...
Electrons in Atoms
... Cone-shaped cells in our eyes act as receivers tuned to the wavelengths in this narrow band of the spectrum A typical human eye will respond to wavelengths from about 380 to 750 nm The spectrum does not contain all the colors that the human eye and brain can distinguish Unsaturated colors such a ...
... Cone-shaped cells in our eyes act as receivers tuned to the wavelengths in this narrow band of the spectrum A typical human eye will respond to wavelengths from about 380 to 750 nm The spectrum does not contain all the colors that the human eye and brain can distinguish Unsaturated colors such a ...
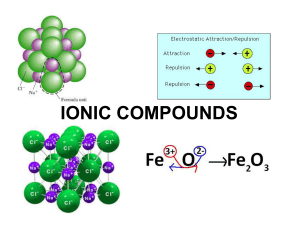
Section 8.3 Names and Formulas of Ionic Compounds Formula Unit
... • 3. Noble gases are relatively unreactive because they have eight electrone I n their outermost energy ...
... • 3. Noble gases are relatively unreactive because they have eight electrone I n their outermost energy ...
1. Interaction of x
... The progress of high energy physics, with the construction of powerful particle accelerators gave birth to what we now call First generation synchrotron sources (1970). Using the deflection of high energy electrons by a magnetic field for the production of x-rays proved so promising that a number of ...
... The progress of high energy physics, with the construction of powerful particle accelerators gave birth to what we now call First generation synchrotron sources (1970). Using the deflection of high energy electrons by a magnetic field for the production of x-rays proved so promising that a number of ...
vsepr_lite_oct_2011 - chemistry11crescentsummer
... 2. VSEPR theory treats double bonds and triple bonds the same as a single bond. That is, the space requirements of a double and triple bond are similar to that of a single bond. What is the total of the chemical bonds and lone pairs? How can three sets of electrons get as far from each other as poss ...
... 2. VSEPR theory treats double bonds and triple bonds the same as a single bond. That is, the space requirements of a double and triple bond are similar to that of a single bond. What is the total of the chemical bonds and lone pairs? How can three sets of electrons get as far from each other as poss ...
AP Chem Test 5-7 Practice Exam - mvhs
... the element with the electron configuration 1s22s22p63s23p3 the alkaline earth element with the smallest atomic radius the element with the largest ionization energy in Group 5A the element whose 2+ ion has the configuration [Kr] 4d5 the element with the most negative electron affinity in Group 7A t ...
... the element with the electron configuration 1s22s22p63s23p3 the alkaline earth element with the smallest atomic radius the element with the largest ionization energy in Group 5A the element whose 2+ ion has the configuration [Kr] 4d5 the element with the most negative electron affinity in Group 7A t ...
The Satellites of Uranus and Neptune: A New Astrometrie Programme
... X-ray flux is due to this radiating matter. So me evidence for such intra-cluster matter can be seen in figure 1. Since this interpretation was published in 1972 many new X-ray clusters have been discovered. So me of the more recent clusters contain relatively few galaxies, raising the question "Are ...
... X-ray flux is due to this radiating matter. So me evidence for such intra-cluster matter can be seen in figure 1. Since this interpretation was published in 1972 many new X-ray clusters have been discovered. So me of the more recent clusters contain relatively few galaxies, raising the question "Are ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.