Polarity of Molecules
... Valence bond (VB) model Combines the notion of electron-pair bonding (Lewis valence) with the idea of atomic orbitals Review atomic orbitals from Ch 6! ...
... Valence bond (VB) model Combines the notion of electron-pair bonding (Lewis valence) with the idea of atomic orbitals Review atomic orbitals from Ch 6! ...
Question 2
... 1. Small quantities of hydrogen gas can be prepared in the laboratory by the following reaction: Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) Assume you carried out this experiment and collected 653 mL of hydrogen gas over water. The gas mixture collected includes hydrogen and water vapor. The temperature ...
... 1. Small quantities of hydrogen gas can be prepared in the laboratory by the following reaction: Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) Assume you carried out this experiment and collected 653 mL of hydrogen gas over water. The gas mixture collected includes hydrogen and water vapor. The temperature ...
Document
... I guess the reason is the same one as that causing hydrogen bonding in chloroform “A hydrogen attached to carbon can also participate in hydrogen bonding when the carbon atom is bound to electronegative atoms, as is the case in chloroform, CHCl3” (from wikipedia http://en.wikipedia.org/wiki/Hydrogen ...
... I guess the reason is the same one as that causing hydrogen bonding in chloroform “A hydrogen attached to carbon can also participate in hydrogen bonding when the carbon atom is bound to electronegative atoms, as is the case in chloroform, CHCl3” (from wikipedia http://en.wikipedia.org/wiki/Hydrogen ...
幻灯片 1
... the electrons ll up the allowed orbits starting from the bottom. Each energy level only has room for a certain number of electrons so they cannot all go into the lowest level and they arrange themselves in shells, labelled by the principal quantum number, around the nucleus. This shell structure ari ...
... the electrons ll up the allowed orbits starting from the bottom. Each energy level only has room for a certain number of electrons so they cannot all go into the lowest level and they arrange themselves in shells, labelled by the principal quantum number, around the nucleus. This shell structure ari ...
Sample Exercise 2.1 Illustrating the Size of an Atom
... isotope of titanium is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the symbol for the ion is 48Ti3+. (b) By referring to a periodic table or a table of elements, we see that sulfur (S) has an atomic numbe ...
... isotope of titanium is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the symbol for the ion is 48Ti3+. (b) By referring to a periodic table or a table of elements, we see that sulfur (S) has an atomic numbe ...
Ch02-sample-and-practice-set-2
... isotope of titanium is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the symbol for the ion is 48Ti3+. (b) By referring to a periodic table or a table of elements, we see that sulfur (S) has an atomic numbe ...
... isotope of titanium is 22 + 26 = 48 (the sum of the protons and neutrons). Because the ion has three more protons than electrons, it has a net charge of 3+. Thus, the symbol for the ion is 48Ti3+. (b) By referring to a periodic table or a table of elements, we see that sulfur (S) has an atomic numbe ...
PERIODIC TABLE OF THE ELEMENTS
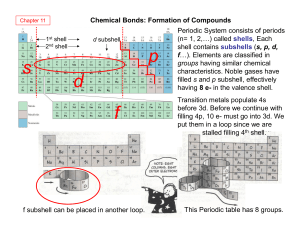
... • For our purposes, we’re only going to focus on the elements having an M SHELL (it is happiest with 8 electrons). ...
... • For our purposes, we’re only going to focus on the elements having an M SHELL (it is happiest with 8 electrons). ...
Basic Chemistry Lecture Notes - Roderick Biology
... electron to Cl. This fills Cl’s energy level and drops Na back to a full energy level. Because of the number of electrons is no longer the same as the protons there is a negative and positive charge. ...
... electron to Cl. This fills Cl’s energy level and drops Na back to a full energy level. Because of the number of electrons is no longer the same as the protons there is a negative and positive charge. ...
Webquest Review - Harrison High School
... 20. Explain why magnesium chloride should dissolve in water. Magnesium chloride is an ionic compound composed of a cation and two anions. Most ionic compounds with charges are easily pulled into solution with water which is polar (partial charges). 21. Which of the following would be the most diffic ...
... 20. Explain why magnesium chloride should dissolve in water. Magnesium chloride is an ionic compound composed of a cation and two anions. Most ionic compounds with charges are easily pulled into solution with water which is polar (partial charges). 21. Which of the following would be the most diffic ...
Molecules in Intense Laser Fields
... cycle laser pulses. in the atomic case new nonperturbative optical phenomena have been found such as above threshold ionization (ATI), tunnelling ionization which is the basis of recollision physics [1]. Another important nonlinear process, high order harmonic generation (HHG) is the current main so ...
... cycle laser pulses. in the atomic case new nonperturbative optical phenomena have been found such as above threshold ionization (ATI), tunnelling ionization which is the basis of recollision physics [1]. Another important nonlinear process, high order harmonic generation (HHG) is the current main so ...
PPT - Chandra X
... Spectral Fitting 1. Estimate column density (NH = 5.9*10^21*E(B-V)) if E(B-V) is known 2. Fit the ungrouped spectrum (CSTAT statistics, Powell optimization, kT1 [0.01-1 keV] and kT2 [1-10 kev], ...
... Spectral Fitting 1. Estimate column density (NH = 5.9*10^21*E(B-V)) if E(B-V) is known 2. Fit the ungrouped spectrum (CSTAT statistics, Powell optimization, kT1 [0.01-1 keV] and kT2 [1-10 kev], ...
ChESS: ChaMP Extended Stellar Survey
... Also shown are urves orresponding to the number predi ted based on a ...
... Also shown are urves orresponding to the number predi ted based on a ...
Unit 2 Part I PowerPoint
... nucleus surrounded by electrons that travel in circular orbits around the nucleus • Dalton’s Postulates - States that matter is composed of extremely small particles called atoms; atoms are invisible and indestructable; atoms of a given element are identical in size, mass, and chemical properties; a ...
... nucleus surrounded by electrons that travel in circular orbits around the nucleus • Dalton’s Postulates - States that matter is composed of extremely small particles called atoms; atoms are invisible and indestructable; atoms of a given element are identical in size, mass, and chemical properties; a ...
Semiconductor
... covalent bond structure and therefore a hole in the valence band of the energy level diagram. Every impurity atom will produce a hole in the valence band. These holes will drift to produce an electrical current if a voltage is applied to the material and the P type semiconductor is a much better con ...
... covalent bond structure and therefore a hole in the valence band of the energy level diagram. Every impurity atom will produce a hole in the valence band. These holes will drift to produce an electrical current if a voltage is applied to the material and the P type semiconductor is a much better con ...
IB Definitions
... Standard state is the accepted form of the substance at 298K and 101.3KPa (1atm) (eg graphite for carbon and not diamond, O2 for oxygen and not O3) Standard enthalpy change of formation is the enthalpy change when one mole of a substance is formed from its elements in their standard states. Reactant ...
... Standard state is the accepted form of the substance at 298K and 101.3KPa (1atm) (eg graphite for carbon and not diamond, O2 for oxygen and not O3) Standard enthalpy change of formation is the enthalpy change when one mole of a substance is formed from its elements in their standard states. Reactant ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.