Syracuse University
... the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice). Regular study time should include a review of the lecture notes and time for working problems. Strive not to fall behind in this class as it is very difficult, if not impossi ...
... the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice). Regular study time should include a review of the lecture notes and time for working problems. Strive not to fall behind in this class as it is very difficult, if not impossi ...
Ch. 02 - HCC Learning Web
... • Valence electrons are those in the outermost shell, or valence shell • The chemical behavior of an atom is mostly determined by the valence electrons • Elements with a full valence shell are ...
... • Valence electrons are those in the outermost shell, or valence shell • The chemical behavior of an atom is mostly determined by the valence electrons • Elements with a full valence shell are ...
AP Chemistry Second Semester Notes
... b. shorter bond distance = stronger bond c. polar bond when electrons are not shared equally 1. electronegativity a. measures atom's attraction for bonding electron pair (higher # = stronger) b. relative scale where period 2 elements are 1.0 (Li) to 4.0 (F), with 0.5 intervals 1. noble gases are exc ...
... b. shorter bond distance = stronger bond c. polar bond when electrons are not shared equally 1. electronegativity a. measures atom's attraction for bonding electron pair (higher # = stronger) b. relative scale where period 2 elements are 1.0 (Li) to 4.0 (F), with 0.5 intervals 1. noble gases are exc ...
Syllabus
... The chapters assigned in the course schedule should be read prior to class. RESPONSIBILITIES: Each student will be expected to understand all the material covered in class, in the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice) ...
... The chapters assigned in the course schedule should be read prior to class. RESPONSIBILITIES: Each student will be expected to understand all the material covered in class, in the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice) ...
Syracuse University
... The chapters assigned in the course schedule should be read prior to class. RESPONSIBILITIES: Each student will be expected to understand all the material covered in class, in the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice) ...
... The chapters assigned in the course schedule should be read prior to class. RESPONSIBILITIES: Each student will be expected to understand all the material covered in class, in the texts, and in the problems. You should be able to solve all the problems efficiently (which comes only through practice) ...
Notes on QA - Scarsdale Public Schools
... Qualitative analysis involves separating a mixture of cations based on their solubilities. In the traditional QA scheme, the Group 1 cations (not the periodic table group 1) are separated from a mixture of dozens of cations based on their insolubility as chloride salts. Hence the Group 1 cations con ...
... Qualitative analysis involves separating a mixture of cations based on their solubilities. In the traditional QA scheme, the Group 1 cations (not the periodic table group 1) are separated from a mixture of dozens of cations based on their insolubility as chloride salts. Hence the Group 1 cations con ...
Introduction to SOLID STATE PHYSICS
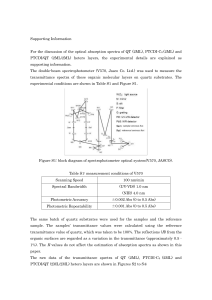
... When the Na+ and Cl- ions approach each other closely enough so that the orbits of the electron in the ions begin to overlap with each other, then the electron begins to repel each other by virtue of the repulsive electrostatic coulomb force. Of course the closer together the ions are, the greater t ...
... When the Na+ and Cl- ions approach each other closely enough so that the orbits of the electron in the ions begin to overlap with each other, then the electron begins to repel each other by virtue of the repulsive electrostatic coulomb force. Of course the closer together the ions are, the greater t ...
Click here to Ch 06.2 Covalent Bonding_Lewis Structures
... • Exceptions to the octet rule include those for atoms that cannot fit eight electrons, and for those that can fit more than eight electrons, into their outermost orbital. • Hydrogen forms bonds in which it is surrounded by only two electrons. • Boron has just three valence electrons, so it tends to ...
... • Exceptions to the octet rule include those for atoms that cannot fit eight electrons, and for those that can fit more than eight electrons, into their outermost orbital. • Hydrogen forms bonds in which it is surrounded by only two electrons. • Boron has just three valence electrons, so it tends to ...
Preview Sample 1
... 50) A nucleotide consists of A) a five-carbon sugar and a nitrogenous base. B) a five-carbon sugar, a nitrogenous base, and a phosphate group. C) a phosphate group and a nitrogenous base. D) a five-carbon sugar and an amino acid. E) a five-carbon sugar and phosphate group. ...
... 50) A nucleotide consists of A) a five-carbon sugar and a nitrogenous base. B) a five-carbon sugar, a nitrogenous base, and a phosphate group. C) a phosphate group and a nitrogenous base. D) a five-carbon sugar and an amino acid. E) a five-carbon sugar and phosphate group. ...
chapter-2 - HCC Learning Web
... • Valence electrons are those in the outermost shell, or valence shell • The chemical behavior of an atom is mostly determined by the valence electrons • Elements with a full valence shell are ...
... • Valence electrons are those in the outermost shell, or valence shell • The chemical behavior of an atom is mostly determined by the valence electrons • Elements with a full valence shell are ...
a level chemistry - some definitions to learn
... The number of protons in the nucleus of an atom The sum of the protons and neutrons in the nucleus of an atom The mass of an atom relative to that of the carbon 12 isotope having a value of 12.000 The simplest, whole number, ratio of elements in a compound The exact number of atoms of each element i ...
... The number of protons in the nucleus of an atom The sum of the protons and neutrons in the nucleus of an atom The mass of an atom relative to that of the carbon 12 isotope having a value of 12.000 The simplest, whole number, ratio of elements in a compound The exact number of atoms of each element i ...
Are diglycolamide ligands hard or soft Lewis bases?
... EuIII ions of the amide oxygen atoms in 2,9-diamide-1,10phenantroline than that of the ketone oxygen atoms in 2,9-diketone-1,10-phenantroline. Also, this difference can be interpreted in terms of the greater softness of amides than of ketones (see the Table above). New hypothesis have been formulate ...
... EuIII ions of the amide oxygen atoms in 2,9-diamide-1,10phenantroline than that of the ketone oxygen atoms in 2,9-diketone-1,10-phenantroline. Also, this difference can be interpreted in terms of the greater softness of amides than of ketones (see the Table above). New hypothesis have been formulate ...
Molecules & Condensed Matter
... • The energy band concept, introduced in 1928, looks at how the outer energy levels of states in an atom vary with distance. When atoms are close enough together, the bands from one atom can join with another to permit lots of states in a closely spaced band of energy. • Some bands are filled, some ...
... • The energy band concept, introduced in 1928, looks at how the outer energy levels of states in an atom vary with distance. When atoms are close enough together, the bands from one atom can join with another to permit lots of states in a closely spaced band of energy. • Some bands are filled, some ...
Most-likely Black Holes
... Case C Mass Transfer (in supergiant stage of BH progenitor) If BH formation through Case B (in giant stage) is possible, contrary to the observation, we should see about 10 times more BHs in our Galaxy. ...
... Case C Mass Transfer (in supergiant stage of BH progenitor) If BH formation through Case B (in giant stage) is possible, contrary to the observation, we should see about 10 times more BHs in our Galaxy. ...
Homework 1 - Devin Gatherwright IET 307 Portfolio
... 1. What is metallic bonding? How is it formed? What are its characteristics? Answer: According to our Materials Science textbook, metallic bonding can be defined as a “primary interatomic bond that involves the non-directional sharing of valence electrons that are shared mutually between all atoms i ...
... 1. What is metallic bonding? How is it formed? What are its characteristics? Answer: According to our Materials Science textbook, metallic bonding can be defined as a “primary interatomic bond that involves the non-directional sharing of valence electrons that are shared mutually between all atoms i ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.