MSWord_examle - Magnetic Resonance in Solids
... Our work was initially stimulated by investigation of heavy-fermion Kondo lattice compounds. Very peculiar magnetic, thermal and transport properties of 4f-electron based heavy-fermion systems are determined by the interplay of the strong repulsion of 4f-electrons on the rare-earth ion sites, their ...
... Our work was initially stimulated by investigation of heavy-fermion Kondo lattice compounds. Very peculiar magnetic, thermal and transport properties of 4f-electron based heavy-fermion systems are determined by the interplay of the strong repulsion of 4f-electrons on the rare-earth ion sites, their ...
Slajd 1 - Uniwersytet Warszawski
... Verification of the hypothesis was done in two ways 1) using the quantum chemical method by which thermodynamic functions, enthalpy, and free enthalpy were calculated, 2) using MS method applying ‘soft’ ionization techniques APCI and ESI, which allow samples to be analysed in a liquid mobile phase, ...
... Verification of the hypothesis was done in two ways 1) using the quantum chemical method by which thermodynamic functions, enthalpy, and free enthalpy were calculated, 2) using MS method applying ‘soft’ ionization techniques APCI and ESI, which allow samples to be analysed in a liquid mobile phase, ...
Chapter 2
... The salt calcium fluoride is an ionic substance with formula CaF2, meaning that, while the atoms of Ca and F are in a 1-to-2 ratio, it does not exist as molecules of CaF2, but as a crystal lattice containing 1 Ca for every 2 F atoms. ...
... The salt calcium fluoride is an ionic substance with formula CaF2, meaning that, while the atoms of Ca and F are in a 1-to-2 ratio, it does not exist as molecules of CaF2, but as a crystal lattice containing 1 Ca for every 2 F atoms. ...
-Atomic Bonding in Solids
... Metallic bonding, the final primary bonding type, is found in metals and their alloys. A relatively simple model has been proposed that very nearly approximates the bonding scheme. Metallic materials have one, two, or at most, three valence electrons. With this model, these valence electrons are not ...
... Metallic bonding, the final primary bonding type, is found in metals and their alloys. A relatively simple model has been proposed that very nearly approximates the bonding scheme. Metallic materials have one, two, or at most, three valence electrons. With this model, these valence electrons are not ...
Basics of material sciece - E
... the valence electrons are those that occupy the outermost filled shell. These electrons are extremely important; as will be seen, they participate in the bonding between atoms to form atomic and molecular aggregates. Furthermore, many of the physical and chemical properties of solids are based on th ...
... the valence electrons are those that occupy the outermost filled shell. These electrons are extremely important; as will be seen, they participate in the bonding between atoms to form atomic and molecular aggregates. Furthermore, many of the physical and chemical properties of solids are based on th ...
Honors Chemistry
... b. matter, its properties and composition, and the changes that matter undergoes c. energy and its transformation from one form to another d. the structure of the earth’s crust and the formation and development of its various layers. ...
... b. matter, its properties and composition, and the changes that matter undergoes c. energy and its transformation from one form to another d. the structure of the earth’s crust and the formation and development of its various layers. ...
Advanced Characterization methods lectures
... IR spectra have similar selection rules as for optical spectroscopy – this means that many bands can not be measured with usual IR absorption. Raman spectroscopy (scattering) offers a way around this - since the interactions involve more than one photon, the selection rules are different than in sta ...
... IR spectra have similar selection rules as for optical spectroscopy – this means that many bands can not be measured with usual IR absorption. Raman spectroscopy (scattering) offers a way around this - since the interactions involve more than one photon, the selection rules are different than in sta ...
Atoms and Molecules - Wilson School District
... Chemical Bonds, Molecular Shapes, and Molecular Models Offers tutorials on chemical bonding and VSEPR structures, utilizing static images of molecular models, as well as three-dimensional, animated molecules that can be manipulated by students. The CHIME plug-in, which is a free download available a ...
... Chemical Bonds, Molecular Shapes, and Molecular Models Offers tutorials on chemical bonding and VSEPR structures, utilizing static images of molecular models, as well as three-dimensional, animated molecules that can be manipulated by students. The CHIME plug-in, which is a free download available a ...
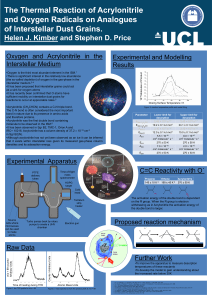
Oxygen is the third most abundant element in the ISM.1
... •There is significant interest in the relatively low abundance (the so-called depletion) of oxygen in the gas-phase in the interstellar medium.2-4 •It has been proposed that interstellar grains could act as a sink for oxygen atoms •It has recently been confirmed that O atoms have sufficient mobility ...
... •There is significant interest in the relatively low abundance (the so-called depletion) of oxygen in the gas-phase in the interstellar medium.2-4 •It has been proposed that interstellar grains could act as a sink for oxygen atoms •It has recently been confirmed that O atoms have sufficient mobility ...
Informational Brochure "Enlightening Science"
... new opportunities for science. Research groups from around the world will investigate nanometre-scale structures, fast processes, and extreme states; take three-dimensional (3D) images of viruses and proteins; and film chemical reactions. The construction and operation of the facility is entrusted t ...
... new opportunities for science. Research groups from around the world will investigate nanometre-scale structures, fast processes, and extreme states; take three-dimensional (3D) images of viruses and proteins; and film chemical reactions. The construction and operation of the facility is entrusted t ...
Know (main topic)
... - -memorize the 3 subatomic particles of an atom -determine atomic number, mass number, protons, electrons, & neutrons from the periodic table -explain why the nucleus of the atom is much smaller than the atom yet contains most of its mass -relate the position of an element in the Periodic Table to ...
... - -memorize the 3 subatomic particles of an atom -determine atomic number, mass number, protons, electrons, & neutrons from the periodic table -explain why the nucleus of the atom is much smaller than the atom yet contains most of its mass -relate the position of an element in the Periodic Table to ...
follow up solids
... Diamond is the metastable form of carbon. The stable form is graphite! Diamond can be synthesized from graphite at high pressure Memorial diamonds from carbonized human remains by companies such as ...
... Diamond is the metastable form of carbon. The stable form is graphite! Diamond can be synthesized from graphite at high pressure Memorial diamonds from carbonized human remains by companies such as ...
File
... atomic emission spectra; Bohr model of the hydrogen atom including explanation of H line spectrum and orbits, electron cloud and the probability model, wave/particle duality of electrons revisited, relate electron configurations of atoms to the Bohr and electron cloud models, describe the concepts o ...
... atomic emission spectra; Bohr model of the hydrogen atom including explanation of H line spectrum and orbits, electron cloud and the probability model, wave/particle duality of electrons revisited, relate electron configurations of atoms to the Bohr and electron cloud models, describe the concepts o ...
Nucleon number
... Define relative atomic mass and relative molecular mass based on the C-12 scale. Sketch and explain the function of the following main components of a simple mass spectrum: Analyze mass spectrum of an element. Name cations, anions and salt according to the ...
... Define relative atomic mass and relative molecular mass based on the C-12 scale. Sketch and explain the function of the following main components of a simple mass spectrum: Analyze mass spectrum of an element. Name cations, anions and salt according to the ...
Quarterly 1 Review Trupia - Trupia
... ____53. Which element has a total of 5 valence electrons present in the fifth energy level (shell)? ____60. Which element forms an ion that is larger (1) Sb (3) I than its atom? (2) Bi (4) Br (1) aluminum (3) magnesium (2) chlorine (4) sodium ____54. Lithium and potassium have similar chemical prope ...
... ____53. Which element has a total of 5 valence electrons present in the fifth energy level (shell)? ____60. Which element forms an ion that is larger (1) Sb (3) I than its atom? (2) Bi (4) Br (1) aluminum (3) magnesium (2) chlorine (4) sodium ____54. Lithium and potassium have similar chemical prope ...
Chemistry Readings
... "k, l, m, n, o, p, and q". The "k" shell is the one closest to the nucleus and "q" is the farthest away. Not all shells hold the same number of electrons. The general rule for calculating the number of electrons for the first four shells is 2n2 where n represents the shell number. For the first eigh ...
... "k, l, m, n, o, p, and q". The "k" shell is the one closest to the nucleus and "q" is the farthest away. Not all shells hold the same number of electrons. The general rule for calculating the number of electrons for the first four shells is 2n2 where n represents the shell number. For the first eigh ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.