Chemistry Syllabus - Madison County Schools
... Periodic properties (e.g., metal/nonmetal/metalloid behavior, electrical/heat conductivity, electronegativity, electron affinity, ionization energy, atomic/covalent/ionic radius) 2e. Compare the properties of compounds according to their type of bonding. (DOK 1) Covalent, ionic, and metallic bon ...
... Periodic properties (e.g., metal/nonmetal/metalloid behavior, electrical/heat conductivity, electronegativity, electron affinity, ionization energy, atomic/covalent/ionic radius) 2e. Compare the properties of compounds according to their type of bonding. (DOK 1) Covalent, ionic, and metallic bon ...
Feasibility Study of using FAIMS to Detect Carbonyl Sulfide in Propane
... wide variety of applications. The user requirements and sample matrix for each application define the sample preparation and introduction steps required. There are a wide variety of sample preparation, extraction and processing techniques each with their own advantages and disadvantages. It is not t ...
... wide variety of applications. The user requirements and sample matrix for each application define the sample preparation and introduction steps required. There are a wide variety of sample preparation, extraction and processing techniques each with their own advantages and disadvantages. It is not t ...
Ten Years Of XMM-Newton: Scientific Achievements And Future Prospects Norbert Schartel
... • Since 20 years QPO in X-ray binaries, but no one in AGNs (13y) • RE J1034+396 nearby (z=0.043) narrow-line Seyfert 1 • Black hole mass: 6.3 x 105 to 3.6 x 107 Msun XMM-Newton detection of a ~ 1 hour quasi periodic oscillation (QPO) Important cornerstone for generalization of accretion process ...
... • Since 20 years QPO in X-ray binaries, but no one in AGNs (13y) • RE J1034+396 nearby (z=0.043) narrow-line Seyfert 1 • Black hole mass: 6.3 x 105 to 3.6 x 107 Msun XMM-Newton detection of a ~ 1 hour quasi periodic oscillation (QPO) Important cornerstone for generalization of accretion process ...
ch6 - ChemistryVCE
... In a metallic lattice, the positive ions are surrounded by delocalised electrons; in an ionic lattice, negative ions alternate with the positive ions. Agree. In a metallic lattice, each positive ion attracts the delocalised electrons in its region, and each delocalised electron is attracted to all n ...
... In a metallic lattice, the positive ions are surrounded by delocalised electrons; in an ionic lattice, negative ions alternate with the positive ions. Agree. In a metallic lattice, each positive ion attracts the delocalised electrons in its region, and each delocalised electron is attracted to all n ...
Chapter 6. Electronic Structure of Atoms
... • Solve Schrödinger equation get wave functions and energies for the wave functions • Schrödinger’s equation requires three quantum numbers: • Principal quantum number, n • n becomes larger, the atom becomes larger and electron further from nucleus • Angular momentum quantum number, l • depends on t ...
... • Solve Schrödinger equation get wave functions and energies for the wave functions • Schrödinger’s equation requires three quantum numbers: • Principal quantum number, n • n becomes larger, the atom becomes larger and electron further from nucleus • Angular momentum quantum number, l • depends on t ...
Chemistry Unit Summaries - Oak Park Unified School District
... In the dimensional analysis technique, we keep track of units observing atomic spectra, which is the radiant energy emitted or as we carry measurements through calculations. The given units absorbed by matter. are multiplied by a series of conversion factors, which are ratios Equations for radiant e ...
... In the dimensional analysis technique, we keep track of units observing atomic spectra, which is the radiant energy emitted or as we carry measurements through calculations. The given units absorbed by matter. are multiplied by a series of conversion factors, which are ratios Equations for radiant e ...
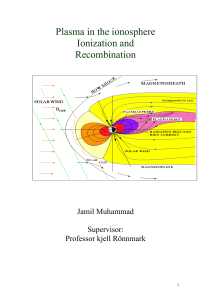
Plasma in ionosphere
... altitude the solar radiation is very strong but there are only few atoms are available to interact, so the ionization is small. As the latitude decreases, the more gas atoms or molecules are present to interact so the ionization is greater. At the same time an opposite process called recombination s ...
... altitude the solar radiation is very strong but there are only few atoms are available to interact, so the ionization is small. As the latitude decreases, the more gas atoms or molecules are present to interact so the ionization is greater. At the same time an opposite process called recombination s ...
Photoelectron spectroscopy of jet
... Pulsed UV laser light (hv=3.68 eV) is utilized to detach electrons from mass-identified cluster anion bunches. In Fig. 3 a, b we present the PE spectra of negatively charged A1, in the size range of 225 atoms. As usually, the data are plotted as a function of electron binding energy, given by the ph ...
... Pulsed UV laser light (hv=3.68 eV) is utilized to detach electrons from mass-identified cluster anion bunches. In Fig. 3 a, b we present the PE spectra of negatively charged A1, in the size range of 225 atoms. As usually, the data are plotted as a function of electron binding energy, given by the ph ...
Advanced Chemistry Midterm
... a. ultraviolet radiation b. infrared radiation c. X-rays d. gamma rays 11. The probability of finding electrons in certain regions of an atom is described by a. orbits b. orbitals c. quanta d. photons 12. How many periods are in the periodic table? a. 7 b. 8 c. 18 d. 109 13. The elements in group 18 ...
... a. ultraviolet radiation b. infrared radiation c. X-rays d. gamma rays 11. The probability of finding electrons in certain regions of an atom is described by a. orbits b. orbitals c. quanta d. photons 12. How many periods are in the periodic table? a. 7 b. 8 c. 18 d. 109 13. The elements in group 18 ...
1. Which terms describe components of atomic structure? • Proton
... 6. Which terms are used to classify rocks and to describe rock properties? • porosity - proportion of holes or cracks in a material which are not filled by solid material. • permeability - measure of how easily fluids can pass through a rock • tensile stress - forces acting on a rock to pull it apar ...
... 6. Which terms are used to classify rocks and to describe rock properties? • porosity - proportion of holes or cracks in a material which are not filled by solid material. • permeability - measure of how easily fluids can pass through a rock • tensile stress - forces acting on a rock to pull it apar ...
IB Chemistry Online EQ_Ans
... protons and hence a greater nuclear charge: 35+ versus 34+. Hence the outer or valence electrons are attracted more strongly. The bromide ion is formed by the addition of one electron to the bromine atom; the selenide ion is formed by the addition of two electrons to the selenium atom. The additiona ...
... protons and hence a greater nuclear charge: 35+ versus 34+. Hence the outer or valence electrons are attracted more strongly. The bromide ion is formed by the addition of one electron to the bromine atom; the selenide ion is formed by the addition of two electrons to the selenium atom. The additiona ...
document
... 1. Electrons occupy the lowest-energy orbitals available, beginning with 1s and continuing in order shown in the fig. 3.5. 2. Each orbital can hold only two electrons, which must be oppositely spin. 3. Two or more orbitals with the same energy – each orbital gets one electron before any orbital gets ...
... 1. Electrons occupy the lowest-energy orbitals available, beginning with 1s and continuing in order shown in the fig. 3.5. 2. Each orbital can hold only two electrons, which must be oppositely spin. 3. Two or more orbitals with the same energy – each orbital gets one electron before any orbital gets ...
TEST on Atomic Structure
... a. They have mobile valence electrons. c. They have mobile cations. b. They have mobile protons. d. Their crystal structures can be rearranged easily. _C__ 38) Which of these elements does not exist as a diatomic molecule? a. H b. F c. Ar d. O (Argon is a noble gas and is stable- will not bond) _C__ ...
... a. They have mobile valence electrons. c. They have mobile cations. b. They have mobile protons. d. Their crystal structures can be rearranged easily. _C__ 38) Which of these elements does not exist as a diatomic molecule? a. H b. F c. Ar d. O (Argon is a noble gas and is stable- will not bond) _C__ ...
Honors Chemistry - Stout Middle School
... j. Oxidation number (charge) k. Polyatomic ion l. Electron sea model m. Octet n. Crystal lattice o. Metallic bond p. Lattice energy q. Alloy 2. Know how cations and anions are formed. 3. Know what elements form cations and anions. 4. Be able to predict oxidation numbers for elements in groups 1, 2, ...
... j. Oxidation number (charge) k. Polyatomic ion l. Electron sea model m. Octet n. Crystal lattice o. Metallic bond p. Lattice energy q. Alloy 2. Know how cations and anions are formed. 3. Know what elements form cations and anions. 4. Be able to predict oxidation numbers for elements in groups 1, 2, ...
Chemistry -- Oxidation
... most compounds. Exceptions are O2 (where O = 0) and peroxides, such as H2O2 or Na2O2, where O = -1. • For other elements, you can usually use If no other rules apply, assume ON is the same as the charge taken on in an ionic compound (“the charge it would like to be) ...
... most compounds. Exceptions are O2 (where O = 0) and peroxides, such as H2O2 or Na2O2, where O = -1. • For other elements, you can usually use If no other rules apply, assume ON is the same as the charge taken on in an ionic compound (“the charge it would like to be) ...
Chapter 3: The Structure of Crystalline Solids
... amorphous structures. • Common metallic crystal structures are FCC, BCC, and HCP. Coordination number and atomic packing factor are the same for both FCC and HCP crystal structures. • We can predict the density of a material, provided we know the atomic weight, atomic radius, and crystal geometry (e ...
... amorphous structures. • Common metallic crystal structures are FCC, BCC, and HCP. Coordination number and atomic packing factor are the same for both FCC and HCP crystal structures. • We can predict the density of a material, provided we know the atomic weight, atomic radius, and crystal geometry (e ...
Soft X-ray spectroscopy of single sized CdS nanocrystals: size
... confining potentials (IP-EMA) [19] which predicts the CBM shift to dominate the band gap opening. In contrast, the experimental results show that both, VBM and CBM shifts, contribute about equally to the gap opening. Using an EMA algorithm developed to account for finite potential heights (FP-EMA) i ...
... confining potentials (IP-EMA) [19] which predicts the CBM shift to dominate the band gap opening. In contrast, the experimental results show that both, VBM and CBM shifts, contribute about equally to the gap opening. Using an EMA algorithm developed to account for finite potential heights (FP-EMA) i ...
Hybridization of atomic orbitals In general VSEPR predicts the
... Hybridization of atomic orbitals In general VSEPR predicts the shape of molecules and ions accurately CH4 : tetrahedral Four equal bonds with equal HCH angles A covalent bond is formed by sharing two electrons by two atoms Imagine an orbital (containing 1 electron) from one atom overlaps with an orb ...
... Hybridization of atomic orbitals In general VSEPR predicts the shape of molecules and ions accurately CH4 : tetrahedral Four equal bonds with equal HCH angles A covalent bond is formed by sharing two electrons by two atoms Imagine an orbital (containing 1 electron) from one atom overlaps with an orb ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.