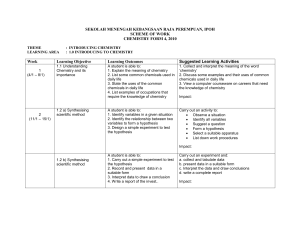
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... change in kinetic energy of particles. 2. Explain the inter- conversion of the states of matter in terms of kinetic theory of matter. ...
... change in kinetic energy of particles. 2. Explain the inter- conversion of the states of matter in terms of kinetic theory of matter. ...
2014 Exams
... excess of NaOH. The solution was centrifuged and decanted. The colorless decantate was treated with first HNO3, then NH3, then BaCl2 and no precipitate was observed upon any of those additions. b. One part was treated with NH4Cl and excess NH3 and gave a blue solution with no precipitate. Which ions ...
... excess of NaOH. The solution was centrifuged and decanted. The colorless decantate was treated with first HNO3, then NH3, then BaCl2 and no precipitate was observed upon any of those additions. b. One part was treated with NH4Cl and excess NH3 and gave a blue solution with no precipitate. Which ions ...
Measuring difficult reaction rates involving
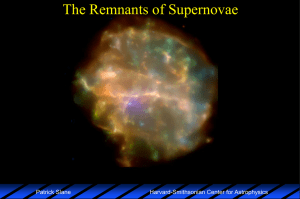
... Contributions from secondary sources (such as novae) is not well established. ...
... Contributions from secondary sources (such as novae) is not well established. ...
1.9 M - Thierry Karsenti
... If sodium hydroxide is added to hydrochloric acid a) The product is chlorine gas. b) The product is chlorine and hydrogen gases. c) The product would be sodium chloride only. d) The product would be sodium chloride and water. ...
... If sodium hydroxide is added to hydrochloric acid a) The product is chlorine gas. b) The product is chlorine and hydrogen gases. c) The product would be sodium chloride only. d) The product would be sodium chloride and water. ...
Articles - Theoretical and Computational Inorganic Chemistry
... similar geometries of (PH3)2Ni and (PH3)2Pt (see Table 1), this large difference in the reorganization energy seems not to be connected with geometry differences between the two fragments and is probably due to different dependencies of their total energy from the P-M-P angle. On the other hand, the ...
... similar geometries of (PH3)2Ni and (PH3)2Pt (see Table 1), this large difference in the reorganization energy seems not to be connected with geometry differences between the two fragments and is probably due to different dependencies of their total energy from the P-M-P angle. On the other hand, the ...
Textbook Answer Keys - Mr. Massey`s Chemistry Pages
... Opinions or conclusions should be presented clearly and supported by appropriate evidence. [Assessment Objective 3] The Dalton model of the atom as an indivisible particles was changed as result of Thomson’s experiment; without the use of high voltage cathode rays and magnetism, Thomson would not ha ...
... Opinions or conclusions should be presented clearly and supported by appropriate evidence. [Assessment Objective 3] The Dalton model of the atom as an indivisible particles was changed as result of Thomson’s experiment; without the use of high voltage cathode rays and magnetism, Thomson would not ha ...
Spectroscopic observation of helium-ion- and hydrogen
... go to an excited electronic state, but it cannot release a photon and go to a lower-energy electronic state). However, an electron transition from the ground state to a lower-energy state may be possible by a resonant nonradiative energy transfer such as multipole coupling or a resonant collision me ...
... go to an excited electronic state, but it cannot release a photon and go to a lower-energy electronic state). However, an electron transition from the ground state to a lower-energy state may be possible by a resonant nonradiative energy transfer such as multipole coupling or a resonant collision me ...
Ch a n d
... link”. Archival observations indicate that an accretion disk was present in this system as recently as 2001 but is absent now, suggesting the radio MSP has turned on after a recent LMXB accretion phase. I will present Chandra and XMM observations of this system and describe how they can offer unique ...
... link”. Archival observations indicate that an accretion disk was present in this system as recently as 2001 but is absent now, suggesting the radio MSP has turned on after a recent LMXB accretion phase. I will present Chandra and XMM observations of this system and describe how they can offer unique ...
Answer Key, Problem Set 6 – complete, with explanations
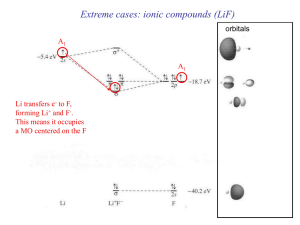
... ions, I have shown the ions as “touching” here—you could have shown them with a bit of space in between them as well, as long as the amount of space in between was roughly “equal” for all adjacent ions). To further ...
... ions, I have shown the ions as “touching” here—you could have shown them with a bit of space in between them as well, as long as the amount of space in between was roughly “equal” for all adjacent ions). To further ...
Principles of Chemistry: A Molecular Approach
... must propose a new model of the atom to replace the first statement in Dalton’s Atomic Theory. rest of Dalton’s theory still valid at this point ...
... must propose a new model of the atom to replace the first statement in Dalton’s Atomic Theory. rest of Dalton’s theory still valid at this point ...
Extraordinary Luminous Soft X-Ray Transient MAXI
... (=1650 V), while the other two (GSC_7 and GSC_8) were operated at the reduced voltage (=1550 V). We analyzed the GSC event data version 1.0 or later, which included the data taken by cameras operated at the nominal and reduced voltages. In these versions, the position and energy responses of the ano ...
... (=1650 V), while the other two (GSC_7 and GSC_8) were operated at the reduced voltage (=1550 V). We analyzed the GSC event data version 1.0 or later, which included the data taken by cameras operated at the nominal and reduced voltages. In these versions, the position and energy responses of the ano ...
Reaction of tungsten anion clusters with molecular and atomic
... whereas a sharp jump of the reactivity is detected for n ⫽15 at liquid-nitrogen temperature as well as room temperature.6,7 In many cases, correlations between electronic structures 共for example, ionization potentials兲 and chemisorption reactivities of metal clusters have been found.1,2,5,8 –10,13,1 ...
... whereas a sharp jump of the reactivity is detected for n ⫽15 at liquid-nitrogen temperature as well as room temperature.6,7 In many cases, correlations between electronic structures 共for example, ionization potentials兲 and chemisorption reactivities of metal clusters have been found.1,2,5,8 –10,13,1 ...
GCE Getting Started - Edexcel
... Atoms of metallic elements in Groups 1,2 and 3 can form positive ions when they take part in reactions since they are readily able to lose electrons. Atoms of Group 1 metals lose one electron and form ions with a 1+ charge, e.g. Na+ Atoms of Group 2 metals lose two electrons and form ions with a 2+ ...
... Atoms of metallic elements in Groups 1,2 and 3 can form positive ions when they take part in reactions since they are readily able to lose electrons. Atoms of Group 1 metals lose one electron and form ions with a 1+ charge, e.g. Na+ Atoms of Group 2 metals lose two electrons and form ions with a 2+ ...
as PDF - Heriot
... [Cr(C2 O4 )3 ]3− is known to exist in two optical isomers. Experimental studies on the racemization reaction of this system show that its activation energy in water is 13.3 kcal mol−1 , and the racemization rate increases with the presence of an acid, alkali, or inert salt, and decreases in organic ...
... [Cr(C2 O4 )3 ]3− is known to exist in two optical isomers. Experimental studies on the racemization reaction of this system show that its activation energy in water is 13.3 kcal mol−1 , and the racemization rate increases with the presence of an acid, alkali, or inert salt, and decreases in organic ...
s_block - ilc.edu.hk
... 2. Group II hydroxides Ca(s) + 2H2O(l) Ca(OH)2(aq) + H2(g) Sr(s) + 2H2O(l) Sr(OH)2(aq) + H2(g) Ba(s) + 2H2O(l) Ba(OH)2(aq) + H2(g) Mg reacts with steam but not water. Mg(s) + H2O(g) MgO(s) + H2(g) Be does not react with water and steam. ...
... 2. Group II hydroxides Ca(s) + 2H2O(l) Ca(OH)2(aq) + H2(g) Sr(s) + 2H2O(l) Sr(OH)2(aq) + H2(g) Ba(s) + 2H2O(l) Ba(OH)2(aq) + H2(g) Mg reacts with steam but not water. Mg(s) + H2O(g) MgO(s) + H2(g) Be does not react with water and steam. ...
Metastable inner-shell molecular state

Metastable Innershell Molecular State (MIMS) is a class of ultra-high-energy short-lived molecules have the binding energy up to 1,000 times larger and bond length up to 100 times smaller than typical molecules. MIMS is formed by inner-shell electrons that are normally resistant to molecular formation. However, in stellar conditions, the inner-shell electrons become reactive to form molecular structures (MIMS) from combinations of all elements in the periodic table. MIMS upon dissociation can emit x-ray photons with energies up to 100 keV at extremely high conversion efficiencies from compression energy to photon energy. MIMS is predicted to exist and dominate radiation processes in extreme astrophysical environments, such as large planet cores, star interiors, and black hole and neutron star surroundings. There, MIMS is predicted to enable highly energy-efficient transformation of the stellar compression energy into the radiation energy.The right schematic illustration shows the proposed four stages of the K-shell MIMS (K-MIMS) formation and x-ray generation process. Stage I: Individual atoms are subjected to the stellar compression and ready for absorbing the compression energy. Stage II: The outer electron shells fuse together under increasing ""stellar"" pressure. Stage III: At the peak pressure, via pressure ionization K-shell orbits form the K-MIMS, which is vibrationally hot and encapsulated by a Rydberg-like pseudo-L-Shell structure. Stage IV: The K-MIMS cools down by ionizing (""boiling-off"") a number of pseudo-L-shell electrons and subsequent optical decay by emitting an x-ray photon. The dissociated atoms return their original atoms states and are ready for absorbing the compression energy.MIMS also can be readily produced in laboratory and industrial environments, such as hypervelocity particle impact, laser fusion and z-machine. MIMS can be exploited for highly energy-efficient production of high intensity x-ray beams for a wide range of innovative applications, such as photolithography, x-ray lasers, and inertial fusion.