Suggested Student Schedule (progress chart – to be
... This course is a study of the fundamental principles of chemistry with particular reference to chemical reactions and equations; energy in chemical systems; atomic and molecular structure; stoichiometry and chemical equilibrium; properties of gases, liquids, solids and solutions. This theory compone ...
... This course is a study of the fundamental principles of chemistry with particular reference to chemical reactions and equations; energy in chemical systems; atomic and molecular structure; stoichiometry and chemical equilibrium; properties of gases, liquids, solids and solutions. This theory compone ...
Thermally induced structural rearrangement of the Fe(ii)
... impinging X-ray photon is sufficient to excite an electron, it will probe the unoccupied electronic states present in the absorbing atom. This part of the spectrum, called X-ray absorption near-edge structure (XANES), gives information on the oxidation state, hybridization, changes in the coordination ...
... impinging X-ray photon is sufficient to excite an electron, it will probe the unoccupied electronic states present in the absorbing atom. This part of the spectrum, called X-ray absorption near-edge structure (XANES), gives information on the oxidation state, hybridization, changes in the coordination ...
Polymer Properties and Structure
... Two types of monomer required – each capable of making two bonds Bonding occurs between them by elimination of components of water ...
... Two types of monomer required – each capable of making two bonds Bonding occurs between them by elimination of components of water ...
Day 05- Matter and the Atom
... protons, but the number of neutrons can vary. For example, most of the oxygen atoms in nature have 8 neutrons in their atomic nuclei. Since all oxygen atoms have 8 protons, this means that most oxygen atoms have an atomic mass of 8+8 = 16. There are also two other stable forms of oxygen that o ...
... protons, but the number of neutrons can vary. For example, most of the oxygen atoms in nature have 8 neutrons in their atomic nuclei. Since all oxygen atoms have 8 protons, this means that most oxygen atoms have an atomic mass of 8+8 = 16. There are also two other stable forms of oxygen that o ...
Chapter 8: Ionic Compounds
... valence electrons are those in the second energy level, as can be seen in the electron-dot structure for carbon in the table. Recall from Chapter 6 that ionization energy refers to how easily an atom loses an electron. The term electron affinity indicates how much attraction an atom has for electron ...
... valence electrons are those in the second energy level, as can be seen in the electron-dot structure for carbon in the table. Recall from Chapter 6 that ionization energy refers to how easily an atom loses an electron. The term electron affinity indicates how much attraction an atom has for electron ...
Chem. 121, Sec 11 Name: Student I.D. Please Show Your Work
... 21. Using the orbital diagram showing the valence electron configuration of magnesium, Mg, aluminum, Al, phosphorous, P, and sulfur, S, explain the following exceptions: a) Why the first ionization energy of Al is smaller than that of Mg, and b) the first ionization energy of S is smaller than that ...
... 21. Using the orbital diagram showing the valence electron configuration of magnesium, Mg, aluminum, Al, phosphorous, P, and sulfur, S, explain the following exceptions: a) Why the first ionization energy of Al is smaller than that of Mg, and b) the first ionization energy of S is smaller than that ...
Introduction to theoretical chemistry 2 semesters
... • What can we learn about the stability and properties of a material by analyzing its electronic orbitals? Such knowledge will allow more powerful prediction. What was known before? • Why is it necessary to consider quantum mechanics? Why does classical mechanics fail? • What is an electronic orbita ...
... • What can we learn about the stability and properties of a material by analyzing its electronic orbitals? Such knowledge will allow more powerful prediction. What was known before? • Why is it necessary to consider quantum mechanics? Why does classical mechanics fail? • What is an electronic orbita ...
Chemistry_in_Parallel_Computing_old
... – Compute the derivative of the wavefunction with respect to the nuclear coordinates – Adjust the nuclear coordinates – Repeat until the derivative is within tolerance of zero in every dimension Note that this is a nested iteration: we’re iterating to build a wavefunction, ...
... – Compute the derivative of the wavefunction with respect to the nuclear coordinates – Adjust the nuclear coordinates – Repeat until the derivative is within tolerance of zero in every dimension Note that this is a nested iteration: we’re iterating to build a wavefunction, ...
Parallel Computing in Chemistry
... – Compute the derivative of the wavefunction with respect to the nuclear coordinates – Adjust the nuclear coordinates – Repeat until the derivative is within tolerance of zero in every dimension Note that this is a nested iteration: we’re iterating to build a wavefunction, ...
... – Compute the derivative of the wavefunction with respect to the nuclear coordinates – Adjust the nuclear coordinates – Repeat until the derivative is within tolerance of zero in every dimension Note that this is a nested iteration: we’re iterating to build a wavefunction, ...
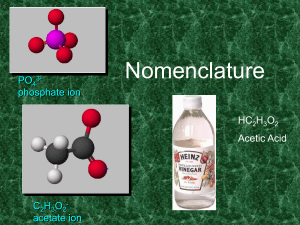
Class Presentation – Naming and Formula Writing
... calcium fluoride Charge on a calcium ion is +2 Charge on a fluorine ion is -1 So the formula is CaF2 ...
... calcium fluoride Charge on a calcium ion is +2 Charge on a fluorine ion is -1 So the formula is CaF2 ...
New Bohr model calculates Helium ground state energy
... Fig.3: The electrons have moved one quarter of their orbitals. Here we investigate how the electrons of the helium atom are moving by calculating the Coulomb force among the two electrons and the nucleus at short time intervals. Methods and results: The computer program (class filename: MathMethod) w ...
... Fig.3: The electrons have moved one quarter of their orbitals. Here we investigate how the electrons of the helium atom are moving by calculating the Coulomb force among the two electrons and the nucleus at short time intervals. Methods and results: The computer program (class filename: MathMethod) w ...
Confined Atoms - Frankfurt Institute for Advanced Studies
... potentials are used for different ranges. A typical example is α-particle decay, where a square well or Woods–Saxon potential is used at short distances, and a Coulombic potential is used for the outer reaches. The confined atom is a variation on such problems. It does, however, correspond to a rath ...
... potentials are used for different ranges. A typical example is α-particle decay, where a square well or Woods–Saxon potential is used at short distances, and a Coulombic potential is used for the outer reaches. The confined atom is a variation on such problems. It does, however, correspond to a rath ...
Newton’s first law
... move to the plate and the current rises accordingly. For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach the plate and thus reduce the current. As V is increas ...
... move to the plate and the current rises accordingly. For mercury atoms, when V=4.9V, the electrons make inelastic collision and leave the atom jump to a high orbit (n=2). The original electrons move off with little energy and could not reach the plate and thus reduce the current. As V is increas ...
8B31A38F-1279-3B00-CDA90244BEA11A7B
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the SECOND element (in this class, it’s NOT optional!). 3. Change the ending of the second element to -ide. ...
... 2. Add prefixes to indicate # of atoms. Omit mono- prefix on the FIRST element. Mono- is OPTIONAL on the SECOND element (in this class, it’s NOT optional!). 3. Change the ending of the second element to -ide. ...
120CH05 - Louisiana Tech University
... • Mass is neither created nor destroyed in an ordinary chemical rxn. • Or the sum of the masses of the reactants is equal to the sum of the masses of the products ...
... • Mass is neither created nor destroyed in an ordinary chemical rxn. • Or the sum of the masses of the reactants is equal to the sum of the masses of the products ...
chemistry
... and carefully, tear off the answer sheet. Then fill in the heading of your answer sheet. All of your answers are to be recorded on the separate answer sheet. For each question, decide which of the choices given is the best answer. Then on the answer sheet, in the row of numbers for that question, ci ...
... and carefully, tear off the answer sheet. Then fill in the heading of your answer sheet. All of your answers are to be recorded on the separate answer sheet. For each question, decide which of the choices given is the best answer. Then on the answer sheet, in the row of numbers for that question, ci ...
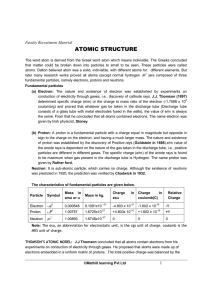
atomic structure
... the same. From that he concluded that all atoms contained electrons. The name electron was given by Irish physicist, Stoney (b) Proton: A proton is a fundamental particle with a charge equal in magnitude but opposite in sign to the charge on the electron, and having a much larger mass. The nature an ...
... the same. From that he concluded that all atoms contained electrons. The name electron was given by Irish physicist, Stoney (b) Proton: A proton is a fundamental particle with a charge equal in magnitude but opposite in sign to the charge on the electron, and having a much larger mass. The nature an ...
Ionization due to the interaction between two Rydberg atoms
... enough that the atoms can be treated as fixed in space over a time of approximately 1 µs. For principle quantum number, n, less than ∼100, the internal quantum states of the atoms can be described within an essential states model if the atoms are separated by more than 10n2 a0, approximately three a ...
... enough that the atoms can be treated as fixed in space over a time of approximately 1 µs. For principle quantum number, n, less than ∼100, the internal quantum states of the atoms can be described within an essential states model if the atoms are separated by more than 10n2 a0, approximately three a ...
AP Chemistry Unit 1 Essential Questions Screencast 1
... 2. What is molar mass? 3. Find the molar mass of water H2O? 4. How do you convert between grams and moles and moles and Avogadro’s number? ...
... 2. What is molar mass? 3. Find the molar mass of water H2O? 4. How do you convert between grams and moles and moles and Avogadro’s number? ...
Chemistry 3211 – Coordination Chemistry Part 4 Electronic Spectra
... −1, 0, +1) so is there a preference? Also, Hund only predicts ground states… what about higher energy excited states? Also, are the electrons paired or unpaired? Again, Hund says they should have parallel spins, but to have two electrons in different p orbitals with one being spin “up” and the other ...
... −1, 0, +1) so is there a preference? Also, Hund only predicts ground states… what about higher energy excited states? Also, are the electrons paired or unpaired? Again, Hund says they should have parallel spins, but to have two electrons in different p orbitals with one being spin “up” and the other ...
Slide 1
... PT contains exactly 6.02x1023 atoms of that element. • And is equal to how much 1 mole of that sample would weigh in grams ...
... PT contains exactly 6.02x1023 atoms of that element. • And is equal to how much 1 mole of that sample would weigh in grams ...
of the atom.
... 3. Multiply step one by step two. Grams to Moles 1. Find the number of grams given in the ...
... 3. Multiply step one by step two. Grams to Moles 1. Find the number of grams given in the ...
The Atomic Theory Chem 111
... B - Definitions: 1) Atom is the smallest basic unit of an element that can enter into a chemical reaction. It is also the smallest unit that cannot be broken down into another chemical substance. 2) Electron is the negatively charged, subatomic particle with a very low mass. 3) Radioactivity is the ...
... B - Definitions: 1) Atom is the smallest basic unit of an element that can enter into a chemical reaction. It is also the smallest unit that cannot be broken down into another chemical substance. 2) Electron is the negatively charged, subatomic particle with a very low mass. 3) Radioactivity is the ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.