Study of excited states of fluorinated copper phthalocyanine by inner
... 691.2 eV, which is higher by 1.4 eV than the photon energy of the lowest NEXAFS peak position. At hν = 691.2 eV, the fluorine K-edge NEXAFS spectra do not show a peak at any incidence angle (see Fig. 2a), indicating that (i) a transition to an unoccupied state which is distributed at the C–F bond in ...
... 691.2 eV, which is higher by 1.4 eV than the photon energy of the lowest NEXAFS peak position. At hν = 691.2 eV, the fluorine K-edge NEXAFS spectra do not show a peak at any incidence angle (see Fig. 2a), indicating that (i) a transition to an unoccupied state which is distributed at the C–F bond in ...
CHEM 250Q
... 77. A researcher uses several procedures to separate a rock sample into different chemicals. A mass of 50 grams of one chemical is produced. If this chemical cannot be separated into other chemicals, then it is best described as A. ...
... 77. A researcher uses several procedures to separate a rock sample into different chemicals. A mass of 50 grams of one chemical is produced. If this chemical cannot be separated into other chemicals, then it is best described as A. ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) CO2(g) + H2O(g) The C atoms are ...
... Write the balanced equation for the reaction that occurs when methanol,CH3OH(l), is burned in air. When any compound containing C, H, and O is combusted, it reacts with the O2(g) in air to produce CO2(g) and H2O(g). Thus, the unbalanced equation is CH3OH(l) + O2(g) CO2(g) + H2O(g) The C atoms are ...
Electron momentum spectroscopy study on valence electronic
... of the outermost orbital is one, while ADC(3)/6-31G method predicted almost the same pole strengths for these four outermost orbitals [10]. It is unlikely caused by an experimental artificial because both the factors at 1500 eV and at 600 eV were less than one. The possible reason is that the ADC(3) ...
... of the outermost orbital is one, while ADC(3)/6-31G method predicted almost the same pole strengths for these four outermost orbitals [10]. It is unlikely caused by an experimental artificial because both the factors at 1500 eV and at 600 eV were less than one. The possible reason is that the ADC(3) ...
Regents Chemistry - New York Science Teacher
... (4) The concentration of the products and the concentration of the reactants are correct constant. ...
... (4) The concentration of the products and the concentration of the reactants are correct constant. ...
Chemistry - An Introduction for Medical and Hea..
... in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1T 4LP, UK, withou ...
... in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1T 4LP, UK, withou ...
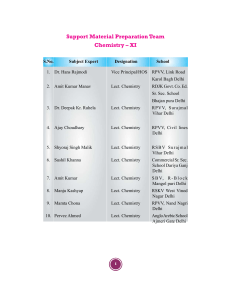
Support Material
... electrons in orbitals : Aufbau principle, Pauli’s exclusion principle and Hund’s rule, electronic con®guration of atoms, stability of half ®lled and completely ®lled orbitals. Unit III : Classi®cation of Elements and Periodicity in Properties 08 Periods Signi®cance of classi® cation, brief history o ...
... electrons in orbitals : Aufbau principle, Pauli’s exclusion principle and Hund’s rule, electronic con®guration of atoms, stability of half ®lled and completely ®lled orbitals. Unit III : Classi®cation of Elements and Periodicity in Properties 08 Periods Signi®cance of classi® cation, brief history o ...
Chemistry: An Introduction for Medical and Health Sciences - E
... in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1T 4LP, UK, withou ...
... in any form or by any means, electronic, mechanical, photocopying, recording, scanning or otherwise, except under the terms of the Copyright, Designs and Patents Act 1988 or under the terms of a licence issued by the Copyright Licensing Agency Ltd, 90 Tottenham Court Road, London W1T 4LP, UK, withou ...
Excitation of Rydberg states in rubidium with near infrared diode lasers
... have been exploited in a number of recent experiments with ultracold atoms. Their large polarizabilities allow pairs of Rydberg atoms to interact strongly over distances of several microns. With the application of a small electric field, a dipole-dipole energy exchange between pairs of atoms can be ...
... have been exploited in a number of recent experiments with ultracold atoms. Their large polarizabilities allow pairs of Rydberg atoms to interact strongly over distances of several microns. With the application of a small electric field, a dipole-dipole energy exchange between pairs of atoms can be ...
Here
... (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decrease for any processes that actually happens. (f) Lewisite is a chlorinate alkyl arsenic compound which was produced as a chemical weapon causing blisters and lung irritation. (g) A Lewis base ...
... (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decrease for any processes that actually happens. (f) Lewisite is a chlorinate alkyl arsenic compound which was produced as a chemical weapon causing blisters and lung irritation. (g) A Lewis base ...
Educação - Química Nova
... The quasi-separability of the charges allows the use of this simple functional model. In general, why can we consider the possibility of describing many-electron atoms based on a hydrogen-like atom model, e.g., electronic distribution? This is possible since the interaction among electrons, in gener ...
... The quasi-separability of the charges allows the use of this simple functional model. In general, why can we consider the possibility of describing many-electron atoms based on a hydrogen-like atom model, e.g., electronic distribution? This is possible since the interaction among electrons, in gener ...
The Physics, Chemistry and Perception of Colored Flames
... seemed to be repeating properties of earlier elements, he started a new row. Thus the elements lithium, sodium and potassium all came to fall in the same column, because they all behaved similarly. Today we understand the underlying reason why the Periodic Table appears as it does and why the proper ...
... seemed to be repeating properties of earlier elements, he started a new row. Thus the elements lithium, sodium and potassium all came to fall in the same column, because they all behaved similarly. Today we understand the underlying reason why the Periodic Table appears as it does and why the proper ...
...detail
... Close-packing: hcp (ABAB…. type), ccp (ABCABC…..type); holes: cubic, tetrahedral and octahedral; radius ratio principle and different polyhedra; structures of MX (NaCl, CsCl and ZnS) , MX2 (CaF2, SiO2, TiO2)/M2X (Na2O), MX3 (AlF3) type solids; Born-Haber cycle, lattice energy, Madelung constant and ...
... Close-packing: hcp (ABAB…. type), ccp (ABCABC…..type); holes: cubic, tetrahedral and octahedral; radius ratio principle and different polyhedra; structures of MX (NaCl, CsCl and ZnS) , MX2 (CaF2, SiO2, TiO2)/M2X (Na2O), MX3 (AlF3) type solids; Born-Haber cycle, lattice energy, Madelung constant and ...
Core_Class_Science_Chemistry_for_the_web 838.3 KB
... Atoms of the same element that have a different number of neutrons are called isotopes. Isotopes that give off radiation are called radioactive isotopes. A compound is a pure substance formed when two or more different elements combine. Compounds are always formed from a specific combination of elem ...
... Atoms of the same element that have a different number of neutrons are called isotopes. Isotopes that give off radiation are called radioactive isotopes. A compound is a pure substance formed when two or more different elements combine. Compounds are always formed from a specific combination of elem ...
Chapter 4
... 3) The oxidation state of oxygen in compounds is -2, except in peroxides, such as H2O2 where it is -1. 4) The oxidation state of hydrogen in compounds is +1, except in metal hydrides, like NaH, where it is -1. ...
... 3) The oxidation state of oxygen in compounds is -2, except in peroxides, such as H2O2 where it is -1. 4) The oxidation state of hydrogen in compounds is +1, except in metal hydrides, like NaH, where it is -1. ...
Ionic Liquids Beyond Simple Solvents: Glimpses at the State of the
... The example shown in Scheme 3 is one in which the organocatalyst is covalently attached to an IL cation. In a recent review, Headley and Ni have called this concept ionic-liquidsupported (ILS) catalysis.[41] Apart from this organocatalytic niche, hardly any convincing example of succesful and sensib ...
... The example shown in Scheme 3 is one in which the organocatalyst is covalently attached to an IL cation. In a recent review, Headley and Ni have called this concept ionic-liquidsupported (ILS) catalysis.[41] Apart from this organocatalytic niche, hardly any convincing example of succesful and sensib ...
Chem-CH8-Review Guide
... where x, y, and z represent the number of atoms in one molecule of the compound (or, in the case of the empirical formula, the lowest whole-number ratio). Our problem, then, is to determine these numbers. Thus, we have been given mass-percent data for the atoms and need to determine the number of at ...
... where x, y, and z represent the number of atoms in one molecule of the compound (or, in the case of the empirical formula, the lowest whole-number ratio). Our problem, then, is to determine these numbers. Thus, we have been given mass-percent data for the atoms and need to determine the number of at ...
Measuring Matter
... you can combine these two definitions into one problem. EXAMPLE: How many molecules are there in 90.1 grams of water? 2 H = 2 x(1.01) = 2.02 O = 1 x (16.00) = 16.00 ...
... you can combine these two definitions into one problem. EXAMPLE: How many molecules are there in 90.1 grams of water? 2 H = 2 x(1.01) = 2.02 O = 1 x (16.00) = 16.00 ...
chemistry 2.1
... • how much is present in a chemical substance, or • how much is involved in a chemical reaction, for one or more chemicals. Significant figures indicate the level of accuracy of the data and/or apparatus. In calculations, final answers typically include three significant figures, and no rounding sho ...
... • how much is present in a chemical substance, or • how much is involved in a chemical reaction, for one or more chemicals. Significant figures indicate the level of accuracy of the data and/or apparatus. In calculations, final answers typically include three significant figures, and no rounding sho ...
Physics Today
... Although impressed with Bohr’s success, many contemporary scientists were offended by his blithe introduction of amenable postulates.1,3 Yet three of his key postulates were destined to become pervasive in quantum mechanics. Foremost was his concept of stationary states, although bereft of electron ...
... Although impressed with Bohr’s success, many contemporary scientists were offended by his blithe introduction of amenable postulates.1,3 Yet three of his key postulates were destined to become pervasive in quantum mechanics. Foremost was his concept of stationary states, although bereft of electron ...
acids, bases and solution equilibria
... A more complete understanding of how species act as acids or bases requires greater knowledge of molecular orbital theory, but we will touch upon some examples here. -acceptor ligands (-acids) CO, CN, O2. Ligands such as these use their anti-bonding -orbitals as acceptor orbitals. ...
... A more complete understanding of how species act as acids or bases requires greater knowledge of molecular orbital theory, but we will touch upon some examples here. -acceptor ligands (-acids) CO, CN, O2. Ligands such as these use their anti-bonding -orbitals as acceptor orbitals. ...
Demonstration of a neutral atom controlled
... conditional state transfer, instead of the more usual conditional phase, can be further generalized to efficiently generate many-atom entanglement [24,25]. Our experimental approach shown in Fig. 2 follows that described more fully in [21] and the associated supplementary information. Single 87 Rb a ...
... conditional state transfer, instead of the more usual conditional phase, can be further generalized to efficiently generate many-atom entanglement [24,25]. Our experimental approach shown in Fig. 2 follows that described more fully in [21] and the associated supplementary information. Single 87 Rb a ...
© NCERT not to be republished
... 33. Why E values for Mn, Ni and Zn are more negative than expected? 34. Why first ionisation enthalpy of Cr is lower than that of Zn ? 35. Transition elements show high melting points. Why? 36. When Cu2+ ion is treated with KI, a white precipitate is formed. Explain the reaction with the help of che ...
... 33. Why E values for Mn, Ni and Zn are more negative than expected? 34. Why first ionisation enthalpy of Cr is lower than that of Zn ? 35. Transition elements show high melting points. Why? 36. When Cu2+ ion is treated with KI, a white precipitate is formed. Explain the reaction with the help of che ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.