Publication : Relativistic Coupled Cluster Calculations with
... implementation of the MLSO formalism into the Tel Aviv atomic computational package allows us to obtain the VP and SE contributions beyond the usual mean-field level, namely at the DCB-FSCCSD level. The individual QED contributions are presented in Table III. To test the validity of these results, w ...
... implementation of the MLSO formalism into the Tel Aviv atomic computational package allows us to obtain the VP and SE contributions beyond the usual mean-field level, namely at the DCB-FSCCSD level. The individual QED contributions are presented in Table III. To test the validity of these results, w ...
Chapter 3 Chemical Reactions and Reaction Stoichiometry
... contains 6.02 × 1023 C2H2 molecules. Because there are two C atoms in each molecule, this sample contains 12.04 × 1023 C atoms. Because each CO2 molecule contains one C atom, the CO2 sample contains 9 × 1023 C atoms. Hence, the order is 12 g 12C (6 × 1023 C atoms) < 9 × 1023 CO2 molecules (9 × 1023 ...
... contains 6.02 × 1023 C2H2 molecules. Because there are two C atoms in each molecule, this sample contains 12.04 × 1023 C atoms. Because each CO2 molecule contains one C atom, the CO2 sample contains 9 × 1023 C atoms. Hence, the order is 12 g 12C (6 × 1023 C atoms) < 9 × 1023 CO2 molecules (9 × 1023 ...
Partial fulfillment of DiVincenzo criteria using manipulation of Rb
... detailed experimental setup is reported by Pearman et al [6] (for simplicity reasons both lasers could be tuned using DF-SAS, or PS, however, both DF-SAS and PS have been used due to other ongoing experiments, where these two stabilization method were part of the setup). This second laser is produce ...
... detailed experimental setup is reported by Pearman et al [6] (for simplicity reasons both lasers could be tuned using DF-SAS, or PS, however, both DF-SAS and PS have been used due to other ongoing experiments, where these two stabilization method were part of the setup). This second laser is produce ...
Structural and Spectroscopic Studies of Solvated Metal Ions Alireza Abbasi Doctoral Thesis
... 8.0(1), respectively, with decreasing size of the ion. The crystal structures at ambient temperature indicate randomly distributed vacancies of the capping oxygen atoms, and 2H solid-state NMR of the diamagnetic [M(H2O)n](CF3SO3)3, M = Sc, Lu, Y and La compounds revealed increasing mobility of the w ...
... 8.0(1), respectively, with decreasing size of the ion. The crystal structures at ambient temperature indicate randomly distributed vacancies of the capping oxygen atoms, and 2H solid-state NMR of the diamagnetic [M(H2O)n](CF3SO3)3, M = Sc, Lu, Y and La compounds revealed increasing mobility of the w ...
unit (4) calculations and chemical reactions
... 1. Write the names, and then formulas of reactants (unit 3 naming). 2. Exchange cations and write the names, then formulas of products (unit 3 naming). 3. Write a chemical equation showing the formulas of reactants and products. 4. From the solubility rules include the (aq) for soluble and (s) for i ...
... 1. Write the names, and then formulas of reactants (unit 3 naming). 2. Exchange cations and write the names, then formulas of products (unit 3 naming). 3. Write a chemical equation showing the formulas of reactants and products. 4. From the solubility rules include the (aq) for soluble and (s) for i ...
Jahn−Teller Distortion in the Phosphorescent Excited State of Three
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
Chapter 2: Mass Relations in Formulas, Chemical Reactions, and
... Finally, one of the most important pieces of information conveyed by a chemical equation is the number of atoms, ions, formula units or molecules associated with each substance. The number in front of each substance is called the stoichiometric coefficients or more simply the coefficient. The bulk o ...
... Finally, one of the most important pieces of information conveyed by a chemical equation is the number of atoms, ions, formula units or molecules associated with each substance. The number in front of each substance is called the stoichiometric coefficients or more simply the coefficient. The bulk o ...
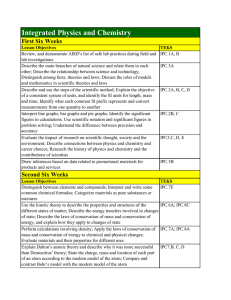
Integrated Physics and Chemistry
... Explain why atoms sometimes join to form bonds; Explain why some IPC.7D, E; IPC.9B atoms transfer their valence electrons to form ionic bonds, while other atoms share valence electrons to form covalent bonds; Differentiate between ionic, covalent, and metallic bonds; Compare the properties of substa ...
... Explain why atoms sometimes join to form bonds; Explain why some IPC.7D, E; IPC.9B atoms transfer their valence electrons to form ionic bonds, while other atoms share valence electrons to form covalent bonds; Differentiate between ionic, covalent, and metallic bonds; Compare the properties of substa ...
CHAPTER 3 STOICHIOMETRY:
... A chemical equation must have an equal number of atoms of each element on each side of the arrow. ...
... A chemical equation must have an equal number of atoms of each element on each side of the arrow. ...
MOLES! - ChemGod.com
... I have 36.45 g of water (H2O), how many moles of water is that? 1. What do you know? g of water 2. What do you want to know? moles of water 3. What do you need to know? molar mass of water ...
... I have 36.45 g of water (H2O), how many moles of water is that? 1. What do you know? g of water 2. What do you want to know? moles of water 3. What do you need to know? molar mass of water ...
Nonlinear Susceptibilities of Donor
... Conjugated polymers possess large nonlinear optical susceptibilities, and are good candidates for optical devices. Their nonlinear optical properties have therefore attracted considerable research activity.'-9 In addition, being one-dimensional systems, electron correlations have strong effects on t ...
... Conjugated polymers possess large nonlinear optical susceptibilities, and are good candidates for optical devices. Their nonlinear optical properties have therefore attracted considerable research activity.'-9 In addition, being one-dimensional systems, electron correlations have strong effects on t ...
Partitlon of the Dipole Moment and Atomic Polar Tensor: The Water
... elements, indicating that the total APT has basically atomic characteristics. APT transference should be done among molecules having this kind of behavior. A d d k g the atomic dipole flux in Table 11 to the homopolar dipole flux in Table 111 yields the “overlap” term from the CCF09-10model. For the ...
... elements, indicating that the total APT has basically atomic characteristics. APT transference should be done among molecules having this kind of behavior. A d d k g the atomic dipole flux in Table 11 to the homopolar dipole flux in Table 111 yields the “overlap” term from the CCF09-10model. For the ...
Critical analysis and extension of the Hirshfeld atoms in molecules
... shortcomings pertain to the deeper context of previous theoretical work on the Hirshfeld idea, but all are relevant to the way these charges are currently obtained computationally and used, for example, in reactivity studies. As will be shown, meaningful charges can be obtained from the present, new ...
... shortcomings pertain to the deeper context of previous theoretical work on the Hirshfeld idea, but all are relevant to the way these charges are currently obtained computationally and used, for example, in reactivity studies. As will be shown, meaningful charges can be obtained from the present, new ...
Entanglement with Negative Wigner Function of Almost 3000 Atoms
... generalized to the case of non-uniform coupling to the measurement light field (see Methods), we deduce an average entanglement depth of N a ~2,910+190 out of Na 5 3,100 atoms (Fig. 3e) using the angleaveraged density matrix. Our results represent the first (to our knowledge) experimental verificati ...
... generalized to the case of non-uniform coupling to the measurement light field (see Methods), we deduce an average entanglement depth of N a ~2,910+190 out of Na 5 3,100 atoms (Fig. 3e) using the angleaveraged density matrix. Our results represent the first (to our knowledge) experimental verificati ...
Ab Initio correlated all electron Dirac
... of single atom events.7 The question about the inertness of element 112,8,9 named copernicium,10 is a good example of the difficulty to characterize chemically the SHE. The theoretical chemical research on SHE is not easier to perform since accurate quantum molecular calculations should be based on ...
... of single atom events.7 The question about the inertness of element 112,8,9 named copernicium,10 is a good example of the difficulty to characterize chemically the SHE. The theoretical chemical research on SHE is not easier to perform since accurate quantum molecular calculations should be based on ...
Redox Reactions - Hillsborough County Public Schools
... A compound has an overall charge of zero, which means all the negative charges have to equal the positive charges. Examples: When calculating the oxidation number of N in NO2 , use the rules above to help you. You see that oxygen normally has an oxidation number of -2 and there are two oxygen atoms. ...
... A compound has an overall charge of zero, which means all the negative charges have to equal the positive charges. Examples: When calculating the oxidation number of N in NO2 , use the rules above to help you. You see that oxygen normally has an oxidation number of -2 and there are two oxygen atoms. ...
Modelling the Role of Charge in Atmospheric Particle
... change the radiative balance is to change the atmosphere and the surface of the Earth. In fact, mankind has been changing them since the dawn of civilization, through e.g. land usage, mining and biomass burning (Ruddiman and Thomson, 2011). The actual magnitude of this influence has been the topic ...
... change the radiative balance is to change the atmosphere and the surface of the Earth. In fact, mankind has been changing them since the dawn of civilization, through e.g. land usage, mining and biomass burning (Ruddiman and Thomson, 2011). The actual magnitude of this influence has been the topic ...
Formation of binary alloy cluster ions from group
... of group-14 elements and cobalt, binary alloy cluster anions of larger size are observed, but no binary alloy cluster cations. This fact shows that the numbers of valence electrons in the binary alloy clusters, i.e. their electronic structures, are the key to the stability of the binary alloy cluste ...
... of group-14 elements and cobalt, binary alloy cluster anions of larger size are observed, but no binary alloy cluster cations. This fact shows that the numbers of valence electrons in the binary alloy clusters, i.e. their electronic structures, are the key to the stability of the binary alloy cluste ...
chemistry writing team
... The lowest energy state of an electron in atom is called ground state (n = 1), when an electron absorb energy, it jumps to higher energy level called excited state, (first excited state n = 2 for H). The energy absorbed or emitted during electronic transition is given by the difference of the energi ...
... The lowest energy state of an electron in atom is called ground state (n = 1), when an electron absorb energy, it jumps to higher energy level called excited state, (first excited state n = 2 for H). The energy absorbed or emitted during electronic transition is given by the difference of the energi ...
Atomic Physics - Oxford Physics
... the 1/r potential. This allowed us to separate the radial and angular solutions. In the many electron case, for most of the time, a major part of the repulsion between one electron and the others acts towards the centre. So we replace the 1/r, hydrogen-like, potential with an effective potential due ...
... the 1/r potential. This allowed us to separate the radial and angular solutions. In the many electron case, for most of the time, a major part of the repulsion between one electron and the others acts towards the centre. So we replace the 1/r, hydrogen-like, potential with an effective potential due ...
Electronic and Electrochemical Properties of Platinum(H) and
... complexes, which are very common for Ir and Rh and, somewhat less so for Pt metal complexes, normally proceed via an oxidative addition process, involving the initially filled high energy d,z orbital, and in which the coordination sphere of the metal centre is extended. It cannot be deduced whether ...
... complexes, which are very common for Ir and Rh and, somewhat less so for Pt metal complexes, normally proceed via an oxidative addition process, involving the initially filled high energy d,z orbital, and in which the coordination sphere of the metal centre is extended. It cannot be deduced whether ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.