some basic concepts of chemistry
... substances. Glucose contains carbon, hydrogen and oxygen in a fixed ratio and thus, like all other pure substances has a fixed composition. Also, the constituents of pure substances cannot be separated by simple physical methods. Pure substances can be further classified into elements and compounds. ...
... substances. Glucose contains carbon, hydrogen and oxygen in a fixed ratio and thus, like all other pure substances has a fixed composition. Also, the constituents of pure substances cannot be separated by simple physical methods. Pure substances can be further classified into elements and compounds. ...
Solvent effects on excited state relaxation phenomena
... solvatochromism, studies on solvent effects on the dynamics or kinetics of excited state decay are much less numerous. ...
... solvatochromism, studies on solvent effects on the dynamics or kinetics of excited state decay are much less numerous. ...
ض ( ا ء ا ط ك ا رر 108 1) -
... What is the ion symbol for an atom with twenty (20) protons and eighteen (18) electrons? ...
... What is the ion symbol for an atom with twenty (20) protons and eighteen (18) electrons? ...
Molar Mass
... • Is the simplest whole number ratio of the atoms • Is calculated by dividing the subscripts in the molecular formula by a whole number to give the lowest ratio C5H10O5 5 = C1H2O1 = ...
... • Is the simplest whole number ratio of the atoms • Is calculated by dividing the subscripts in the molecular formula by a whole number to give the lowest ratio C5H10O5 5 = C1H2O1 = ...
1 - A-Level Chemistry
... no lone / spare / non-bonded pair of electrons only score M2 if M1 correct or give ‘H’ in M1 ...
... no lone / spare / non-bonded pair of electrons only score M2 if M1 correct or give ‘H’ in M1 ...
Solutions
... ‣ The quick story is molecules have a negative end and a positive end. ‣ The negative end of one molecule sticks to the positive end of another. ‣ We’ll discuss the rest in Chapter 11. ‣ Ionic Solids are held together by one type of intermolecular force. ‣ It’s a simpler story. ‣ The cations stick t ...
... ‣ The quick story is molecules have a negative end and a positive end. ‣ The negative end of one molecule sticks to the positive end of another. ‣ We’ll discuss the rest in Chapter 11. ‣ Ionic Solids are held together by one type of intermolecular force. ‣ It’s a simpler story. ‣ The cations stick t ...
Journal of Molecular Catalysis A: Chemical Enhancing
... observed. So we concluded that anhydrous [bmim]HSO4 accelerates the nucleophilic substitution reaction by its ionic character, whereas presence of water decelerate the reaction probably due to its solvation effect. Reaction does not proceed at all in other solvents like water, methanol and dimethyl ...
... observed. So we concluded that anhydrous [bmim]HSO4 accelerates the nucleophilic substitution reaction by its ionic character, whereas presence of water decelerate the reaction probably due to its solvation effect. Reaction does not proceed at all in other solvents like water, methanol and dimethyl ...
Qualitative Analysis Test for Ions
... *(b) Magnesium has an electronic configuration of 2.8.2. Oxygen has an electronic configuration of 2.6. Explain, in terms of their electronic configurations, how magnesium and oxygen atoms react to form the ionic compound magnesium oxide, MgO, and include a description of the structure of solid magn ...
... *(b) Magnesium has an electronic configuration of 2.8.2. Oxygen has an electronic configuration of 2.6. Explain, in terms of their electronic configurations, how magnesium and oxygen atoms react to form the ionic compound magnesium oxide, MgO, and include a description of the structure of solid magn ...
The mole and calculations
... Molar Mass: the mass of a substance per 1 mole of its entities (atoms, molecules, ions, or formula units). Units for molar mass are grams/mole. Once again, the periodic table is used to calculate the molar mass (previously we called this molecular mass - it's the same thing!!) 1.) to find the mo ...
... Molar Mass: the mass of a substance per 1 mole of its entities (atoms, molecules, ions, or formula units). Units for molar mass are grams/mole. Once again, the periodic table is used to calculate the molar mass (previously we called this molecular mass - it's the same thing!!) 1.) to find the mo ...
Chapter 3 Stoichiometry: Calculations with Chemical
... • Most often involve hydrocarbons reacting with oxygen in the air ...
... • Most often involve hydrocarbons reacting with oxygen in the air ...
A tunable two-impurity Kondo system in an atomic point contact
... resonance. Approaching the two cobalt atoms towards each other, the interaction rises and the resonance splits due to exchange interaction. Further reduction of the tip-sample distance increases the interaction and hence the separation of the two peaks. For much stronger exchange interactions, the s ...
... resonance. Approaching the two cobalt atoms towards each other, the interaction rises and the resonance splits due to exchange interaction. Further reduction of the tip-sample distance increases the interaction and hence the separation of the two peaks. For much stronger exchange interactions, the s ...
chemistry - Textbooks Online
... matter which makes up our world and of the interactions between particles on which it depends. The ancient Greek philosophers had their own ideas of the nature of matter, proposing atoms as the smallest indivisible particles. However, although these ideas seems to fit with modern models of matter, s ...
... matter which makes up our world and of the interactions between particles on which it depends. The ancient Greek philosophers had their own ideas of the nature of matter, proposing atoms as the smallest indivisible particles. However, although these ideas seems to fit with modern models of matter, s ...
Naming Compounds - Kowenscience.com
... and solid phases because van der Waals forces keep the molecules attracted to each other ...
... and solid phases because van der Waals forces keep the molecules attracted to each other ...
Chapter 1 Review Questions
... 26. Water molecules have a partial negative charge on the end of the molecule occupied by the oxygen atom and a partial positive charge on the end occupied by the hydrogen atoms. The result is that water molecules can readily dissolve ionic compounds because the positive and negative ions can be pul ...
... 26. Water molecules have a partial negative charge on the end of the molecule occupied by the oxygen atom and a partial positive charge on the end occupied by the hydrogen atoms. The result is that water molecules can readily dissolve ionic compounds because the positive and negative ions can be pul ...
Inorganic and organic chemistry 2
... The coordination number is the number of coordinate bonds between the ligand(s) and the central metal atom or ion. EDTA4− is a hexadentate ligand, so one ion forms six coordinate bonds. The other ligands are monodentate so each forms a single coordinate bond with the central metal atom or ion. ...
... The coordination number is the number of coordinate bonds between the ligand(s) and the central metal atom or ion. EDTA4− is a hexadentate ligand, so one ion forms six coordinate bonds. The other ligands are monodentate so each forms a single coordinate bond with the central metal atom or ion. ...
Document
... • we can simplify the equation for the formation of AgCl by omitting all ions that do not participate in the reaction Net ionic equation: ...
... • we can simplify the equation for the formation of AgCl by omitting all ions that do not participate in the reaction Net ionic equation: ...
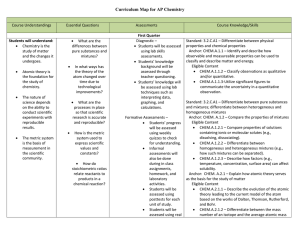
AP Chemistry Curriculum Map - Belle Vernon Area School District
... properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical properties led to the arrangement of elements on the periodic table. Compare and/or predict the properties (e.g., electron affinity, ionizatio ...
... properties of atoms allow for the prediction of physical and chemical properties. Eligible Content CHEM.A.2.3.1 – Explain how the periodicity of chemical properties led to the arrangement of elements on the periodic table. Compare and/or predict the properties (e.g., electron affinity, ionizatio ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.