bond
... of new materials like these relies on the principles of molecular structure introduced in this chapter. ...
... of new materials like these relies on the principles of molecular structure introduced in this chapter. ...
Chapter 7 Ionic and Metallic Bonding
... – Ions are strongly bonded together. – Structure is rigid. 2. High melting points Coordination number- number of ions of opposite charge surrounding it ...
... – Ions are strongly bonded together. – Structure is rigid. 2. High melting points Coordination number- number of ions of opposite charge surrounding it ...
Balancing Chemical Equations
... conservation of mass states that matter cannot either be created or destroyed in a chemical reaction. This means that the mass on both sides of an equation comparing reactants to products must be the same. ...
... conservation of mass states that matter cannot either be created or destroyed in a chemical reaction. This means that the mass on both sides of an equation comparing reactants to products must be the same. ...
ap quick review
... Electrolysis decompose compound into elements (water in dilute acids or solutions of dilute acids) Hydrogen peroxide water + oxygen Metallic carbonates metal oxides + carbon dioxide Hydrolysis = compound reacting with water. Watch for soluble salts that contain anions of weak acid the an ...
... Electrolysis decompose compound into elements (water in dilute acids or solutions of dilute acids) Hydrogen peroxide water + oxygen Metallic carbonates metal oxides + carbon dioxide Hydrolysis = compound reacting with water. Watch for soluble salts that contain anions of weak acid the an ...
Begin Chemical Equations Practice
... • If 58.5 grams of NaCl is decomposed, and 23 grams of Na is formed, how many grams of Cl2 must also be formed? ...
... • If 58.5 grams of NaCl is decomposed, and 23 grams of Na is formed, how many grams of Cl2 must also be formed? ...
specimen
... The student had added the exact amount of calcium required to react with the hydrochloric acid used. After carrying out the experiment, the student accidentally added some more calcium. The student was surprised that the extra calcium still reacted. Explain this observation. Include an equation in y ...
... The student had added the exact amount of calcium required to react with the hydrochloric acid used. After carrying out the experiment, the student accidentally added some more calcium. The student was surprised that the extra calcium still reacted. Explain this observation. Include an equation in y ...
CHEMISTRY IM 06 SYLLABUS
... Chemistry of some common functional groups: alcohols; ethers; aldehydes and ketones; carboxylic acids; amines. The treatment should be an outline survey and should include the following topics (mechanisms will not be tested): (a) alcohols: oxidation, dehydration to alkenes and ethers, esterification ...
... Chemistry of some common functional groups: alcohols; ethers; aldehydes and ketones; carboxylic acids; amines. The treatment should be an outline survey and should include the following topics (mechanisms will not be tested): (a) alcohols: oxidation, dehydration to alkenes and ethers, esterification ...
CHEMISTRY IM 06 SYLLABUS
... Chemistry of some common functional groups: alcohols; ethers; aldehydes and ketones; carboxylic acids; amines. The treatment should be an outline survey and should include the following topics (mechanisms will not be tested): (a) alcohols: oxidation, dehydration to alkenes and ethers, esterification ...
... Chemistry of some common functional groups: alcohols; ethers; aldehydes and ketones; carboxylic acids; amines. The treatment should be an outline survey and should include the following topics (mechanisms will not be tested): (a) alcohols: oxidation, dehydration to alkenes and ethers, esterification ...
Pre-AP Chemistry
... 25. nuclear fission and fusion 26. nuclear waste and radiation 27. radiation properties 28. wave measurements 29. energy of radiation 30. spectral studies, types of spectra, generating spectra 31. Bohr’s Theory 32. wave mechanics of electrons 33. quantum theory, quantum numbers 34. types of electron ...
... 25. nuclear fission and fusion 26. nuclear waste and radiation 27. radiation properties 28. wave measurements 29. energy of radiation 30. spectral studies, types of spectra, generating spectra 31. Bohr’s Theory 32. wave mechanics of electrons 33. quantum theory, quantum numbers 34. types of electron ...
Notes - Organization of Matter
... mixed and cannot be visibly distinguished. The particles of the substances are so small that they cannot be easily seen. Another name for a homogeneous mixture is a ...
... mixed and cannot be visibly distinguished. The particles of the substances are so small that they cannot be easily seen. Another name for a homogeneous mixture is a ...
Semester I CP Chemistry Review
... covalent bonds? What are the electrons doing in a covalent bond? 2 or more nonmetals Electrons are being shared between the atoms ...
... covalent bonds? What are the electrons doing in a covalent bond? 2 or more nonmetals Electrons are being shared between the atoms ...
Document
... 39. When naming a transition metal that has more than one common ionic charge, the numerical value of the charge is indicated by a ____________________. 40. What are the components of binary molecular compounds? ...
... 39. When naming a transition metal that has more than one common ionic charge, the numerical value of the charge is indicated by a ____________________. 40. What are the components of binary molecular compounds? ...
The chemical elements are fundamental building materials of matter
... • 1.A: All matter is made of atoms. There are a limited number of types of atoms: these are the elements. • 1.B: The atoms of each element have unique structures arising from interactions between electrons and nuclei. • 1.C: Elements display periodicity in their properties when the elements are orga ...
... • 1.A: All matter is made of atoms. There are a limited number of types of atoms: these are the elements. • 1.B: The atoms of each element have unique structures arising from interactions between electrons and nuclei. • 1.C: Elements display periodicity in their properties when the elements are orga ...
Discovery of the Electron, Models & Theories
... • Said we can’t assign to an electron a position in space at a given time, nor follow it in its orbit ...
... • Said we can’t assign to an electron a position in space at a given time, nor follow it in its orbit ...
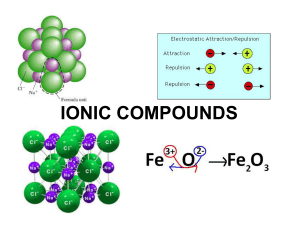
IONIC BONDS MAIN GROUP CHEMISTRY
... • Valence electron config: ns1 • This single s electron is easily lost to form +1 cations. Therefore, these elements have low Ei, are very strong reducing agents (recall Activity Series), metallic, very reactive so they are not found in nature in the elemental form. • Reduction of metal chlorides yi ...
... • Valence electron config: ns1 • This single s electron is easily lost to form +1 cations. Therefore, these elements have low Ei, are very strong reducing agents (recall Activity Series), metallic, very reactive so they are not found in nature in the elemental form. • Reduction of metal chlorides yi ...
Part II - Web site of Dr. Charles Berks
... Binary ionic bonding forms between a metal and a nonmetal whose electronegativities widely differ. This type of bonding produces clusters or aggregates of ions that are more stable than the isolated atoms from which they are formed. This stability is due to the electrostatic attractions that exist b ...
... Binary ionic bonding forms between a metal and a nonmetal whose electronegativities widely differ. This type of bonding produces clusters or aggregates of ions that are more stable than the isolated atoms from which they are formed. This stability is due to the electrostatic attractions that exist b ...
Atomic Structure
... destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions! Atoms of an element have a characteristic average mass which is unique to that element. Atoms of any one element differ in properties from atoms of another element ...
... destroyed in ordinary chemical reactions. However, these changes CAN occur in nuclear reactions! Atoms of an element have a characteristic average mass which is unique to that element. Atoms of any one element differ in properties from atoms of another element ...
Chapter 7 Ionic and Metallic Bonding
... This is a chemical formula, which shows the kinds and numbers of atoms in the smallest representative particle of the ...
... This is a chemical formula, which shows the kinds and numbers of atoms in the smallest representative particle of the ...
30 - Edgemead High School
... Describe a covalent chemical bond as a shared pair of electrons Describe and apply simple rules to deduce bond formation, viz. o different atoms, each with an unpaired valence electron can share these electrons to form a chemical bond o different atoms with paired valence electrons called lone pairs ...
... Describe a covalent chemical bond as a shared pair of electrons Describe and apply simple rules to deduce bond formation, viz. o different atoms, each with an unpaired valence electron can share these electrons to form a chemical bond o different atoms with paired valence electrons called lone pairs ...
Section 8.3 Names and Formulas of Ionic Compounds Formula Unit
... • Many ionic compounds contain polyatomic ions, which are ions made up of more than one atom. • The charge given to a polyatomic ion applies to the entire group of atoms. ...
... • Many ionic compounds contain polyatomic ions, which are ions made up of more than one atom. • The charge given to a polyatomic ion applies to the entire group of atoms. ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.