Ch 18 Power Point
... Na+ (aq ) + NO3- (aq ) + AgCl(s ) • If chemically equivalent amounts of the two solutes are mixed, almost all of the Ag+ ions and Cl− ions combine and separate from the solution as a precipitate of AgCl. • AgCl is only very sparingly soluble in water. • The reaction thus effectively goes to completi ...
... Na+ (aq ) + NO3- (aq ) + AgCl(s ) • If chemically equivalent amounts of the two solutes are mixed, almost all of the Ag+ ions and Cl− ions combine and separate from the solution as a precipitate of AgCl. • AgCl is only very sparingly soluble in water. • The reaction thus effectively goes to completi ...
Kinetics of crystal nucleation in ionic solutions
... IS = 0.09 M. At a given X and IS the respective size of barite crystals decreased with increasing b2 in chloride salts of different cations and remained constant in sodium salts of different anions. We suggest that ionic salts affect the kinetics of barite nucleation and growth due to their influence on ...
... IS = 0.09 M. At a given X and IS the respective size of barite crystals decreased with increasing b2 in chloride salts of different cations and remained constant in sodium salts of different anions. We suggest that ionic salts affect the kinetics of barite nucleation and growth due to their influence on ...
The Inhibition of Carbon Steel Corrosion in Hydrochloric Acid
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
... MXads refer to the halide ion and ion – pair respectively in the adsorbed state. This ion – pair interaction increases the surface coverage thereby reducing metal dissolution. The order of decreasing inhibition efficiency of the investigated compounds on addition of a specific concentration of the u ...
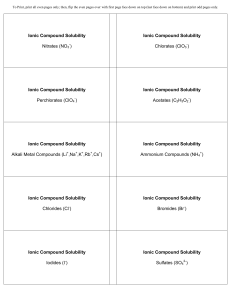
Ionic Compound Solubility Nitrates (NO3 ) Ionic Compound
... Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
... Definition A substance that is able to donate a H+ ion (a proton) and, hence, increases the concentration of H+(aq) when it dissolves in water. ...
Here
... (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero size of the gas molecules and the attractive forces between them. (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decreas ...
... (d) van der Waals equation is a relation between the pressure, temperature and volume of a gas that accounts for the non‐zero size of the gas molecules and the attractive forces between them. (e) Gibbs free energy, G = H − TS, combines enthalpy and entropy to give a quantity which must decreas ...
chem textbook 2015 - Manitowoc Public School District
... The intent of the following information is to give answers and suggestions to questions that students often ask, it is meant to work in conjunction with Suggestions for Boosting Grades. “It makes sense in class but not when I get home.” This generally means that your notes are incomplete, meaning th ...
... The intent of the following information is to give answers and suggestions to questions that students often ask, it is meant to work in conjunction with Suggestions for Boosting Grades. “It makes sense in class but not when I get home.” This generally means that your notes are incomplete, meaning th ...
Chemical Reactions
... presence of oxygen. If the reactant is a compound containing on carbon, hydrogen and oxygen, the products are water and carbon dioxide. CH4(g) +2 O2(g) 2 H2O (l) + CO2(g) ...
... presence of oxygen. If the reactant is a compound containing on carbon, hydrogen and oxygen, the products are water and carbon dioxide. CH4(g) +2 O2(g) 2 H2O (l) + CO2(g) ...
PART 3-ICHO 11-15
... first quantitatively separated from the precipitate, and then hydrogen sulphide was passed through the separated solution to saturation. The resulting precipitate containing metal B was separated, washed and dried. The mass of the precipitate was 0.6613 g. The precipitate containing the compounds of ...
... first quantitatively separated from the precipitate, and then hydrogen sulphide was passed through the separated solution to saturation. The resulting precipitate containing metal B was separated, washed and dried. The mass of the precipitate was 0.6613 g. The precipitate containing the compounds of ...
Question Bank for Pre Board Exam(XII Chemistry)
... 2. Why urea has a sharp melting point but glass does not have? 3. A NaCl crystal is found to have Cs Cl structure. Guess how it might have happened? 4. Why is Frenkel defect not found in pure alkali metal halides? 5. NaCl and Cs Cl have similar formula. Then why they have different structures? 6. No ...
... 2. Why urea has a sharp melting point but glass does not have? 3. A NaCl crystal is found to have Cs Cl structure. Guess how it might have happened? 4. Why is Frenkel defect not found in pure alkali metal halides? 5. NaCl and Cs Cl have similar formula. Then why they have different structures? 6. No ...
Organic Chemistry
... by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)benzene (PS-DIB) as the oxidant under mild conditions. PS-DIB, a yellow powder, was prepared from iodinatio ...
... by the oxidation of thiols. For this work, we have developed a new preparative method for the disulfide using inexpensive, recyclable, and relatively non-toxic polymer-supported (diacetoxyiodo)benzene (PS-DIB) as the oxidant under mild conditions. PS-DIB, a yellow powder, was prepared from iodinatio ...
kcse chemistry questions
... Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled water. Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium h ...
... Describe how the following reagents can be used to prepare lead sulphate solid potassium sulphate, solid lead carbonate, dilute nitric acid and distilled water. Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium h ...
Chaptr 4 HW PacketAK - Summit Academy High School
... HCl – strong acid e) NH3 – weak base You should have memorized the six “strong” acids—those that can completely dissociate in water—memorizing these six means that anything else that is not one of ...
... HCl – strong acid e) NH3 – weak base You should have memorized the six “strong” acids—those that can completely dissociate in water—memorizing these six means that anything else that is not one of ...
الشريحة 1
... In all our discussions of solutions, we must be careful to distinguish the physical process of solution formation from chemical reactions that lead to a solution. e.g., nickel metal is dissolved on contact with HCl solution because the following reaction occurs: ...
... In all our discussions of solutions, we must be careful to distinguish the physical process of solution formation from chemical reactions that lead to a solution. e.g., nickel metal is dissolved on contact with HCl solution because the following reaction occurs: ...
Reactions between dyes of the Alizarin Green series and vanadates
... In all ternary complexes VL2S4 (I) or VL 2 S 6 (II, III) the number of the bonded molecules of tenside is greater than the number of the sulfo groups in the complex. This fact leads to the assumption that the second (/) or third (II, III) molecule of tenside is bonded to convenient negative or stron ...
... In all ternary complexes VL2S4 (I) or VL 2 S 6 (II, III) the number of the bonded molecules of tenside is greater than the number of the sulfo groups in the complex. This fact leads to the assumption that the second (/) or third (II, III) molecule of tenside is bonded to convenient negative or stron ...
HEAd START TO A LEVEL CHEMISTRY WORKbOOK
... A molecule is the smallest, electrically neutral, particle of an element or compound that can exist on its own. An ion is an atom, or group of atoms, which carries an electric charge. You need to know these definitions by heart, but you also need to be able to recognise the formulae of atoms and mol ...
... A molecule is the smallest, electrically neutral, particle of an element or compound that can exist on its own. An ion is an atom, or group of atoms, which carries an electric charge. You need to know these definitions by heart, but you also need to be able to recognise the formulae of atoms and mol ...
pH and ORP Applications Cyanide Waste Treatment
... Sulfuric acid is typically used to lower the pH to about 8.5, allowing the second oxidation to occur more rapidly. Acid addition must have fail-safe design, since pH levels below 7 can generate highly toxic hydrogen cyanide if the first-stage oxidation was not complete. Additional chlorine is added, ...
... Sulfuric acid is typically used to lower the pH to about 8.5, allowing the second oxidation to occur more rapidly. Acid addition must have fail-safe design, since pH levels below 7 can generate highly toxic hydrogen cyanide if the first-stage oxidation was not complete. Additional chlorine is added, ...
184
... correlation consistent basis sets. The theoretical rate constant and activation energy are in excellent agreement with experiment. The rates for BCl + H, BCl + Cl, BH + H, BH + Cl, and B + Cl2 have also been examined but only using empirical BEBO surfaces.7,8 There are no systematic, high-level stud ...
... correlation consistent basis sets. The theoretical rate constant and activation energy are in excellent agreement with experiment. The rates for BCl + H, BCl + Cl, BH + H, BH + Cl, and B + Cl2 have also been examined but only using empirical BEBO surfaces.7,8 There are no systematic, high-level stud ...
alkalinity of groundwater samples
... Where the first term represents the reported alkalinity in mg/L CaCO3, the second term is the formula weight of CaCO3 and the third term is the mole ratio from the neutralization reaction, Eqn. {4]. The speciation diagram shown below can be easily derived from the acid dissociation constants (Ka’s) ...
... Where the first term represents the reported alkalinity in mg/L CaCO3, the second term is the formula weight of CaCO3 and the third term is the mole ratio from the neutralization reaction, Eqn. {4]. The speciation diagram shown below can be easily derived from the acid dissociation constants (Ka’s) ...
Study materials of Chemistry for class XII
... (where k = dissociation constant, C = concentration and α = degree of dissociation ) ...
... (where k = dissociation constant, C = concentration and α = degree of dissociation ) ...
2014 International Practice Exam: Chemistry
... Note: .this .year, .tables .of .equations .and .constants .are .provided .in .the .exam .booklets .for .both . sections .of .the .exam . Students .are .not .allowed .to .use .calculators .in .Section .I .of .the .AP .Chemistry .Exam . .However, . students .are .permitted .to .use .four-function, .sc ...
... Note: .this .year, .tables .of .equations .and .constants .are .provided .in .the .exam .booklets .for .both . sections .of .the .exam . Students .are .not .allowed .to .use .calculators .in .Section .I .of .the .AP .Chemistry .Exam . .However, . students .are .permitted .to .use .four-function, .sc ...
Mole Concept - Shailendra Kumar Chemistry
... agents, can be prepared by reaction of NaH with B 2H 6 according to the equation 2 NaBH4. How many grams of NaBH4 can be prepared by reaction 2 NaH + B2H6 between 8.55 g of NaH and 6.75 g of B2H6 ? Which reactant is limiting, and how many grams of the excess reactant will be left over ? Ans : 13.5 g ...
... agents, can be prepared by reaction of NaH with B 2H 6 according to the equation 2 NaBH4. How many grams of NaBH4 can be prepared by reaction 2 NaH + B2H6 between 8.55 g of NaH and 6.75 g of B2H6 ? Which reactant is limiting, and how many grams of the excess reactant will be left over ? Ans : 13.5 g ...