KHOA: HÓA HỌC - CCS - Trường Đại học Sư phạm Hà Nội
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
... element is a substance comprised of a single type of atom. The elements are the building blocks of our nature. An element is either discovered in nature or synthesized in the laboratory in pure form that cannot be separated into simpler substances by chemical methods. Currently, there are about 118 ...
The applicability of activities in kinetic expressions Haubrock, J.
... by using a pressure controller (Brooks 5866). The pressure in the reactor was monitored by a pressure transducer (Dresser), with a maximum absolute deviation of 0.15 mbar under the experimental conditions applied. Furthermore, the temperatures in the reactor and the gas supply vessel were measured b ...
... by using a pressure controller (Brooks 5866). The pressure in the reactor was monitored by a pressure transducer (Dresser), with a maximum absolute deviation of 0.15 mbar under the experimental conditions applied. Furthermore, the temperatures in the reactor and the gas supply vessel were measured b ...
Interaction of the Adenine-Thymine Watson
... Structures and energetics of complexes between the adenine-thymine Watson-Crick (AT WC) and adenineadenine reverse-Hoogsteen (AA rH) DNA base pairs and hydrated (five water molecules) Mg2+, Ca2+, Sr2+, Ba2+, Zn2+, Cd2+, and Hg2+ metal cations were studied using high-level quantum chemical techniques ...
... Structures and energetics of complexes between the adenine-thymine Watson-Crick (AT WC) and adenineadenine reverse-Hoogsteen (AA rH) DNA base pairs and hydrated (five water molecules) Mg2+, Ca2+, Sr2+, Ba2+, Zn2+, Cd2+, and Hg2+ metal cations were studied using high-level quantum chemical techniques ...
chemistry - Textbooks Online
... The negative and positive ions so formed attains stable noble gas electronic configurations. The noble gases (with the exception of helium which has two electrons in the outermost shell) have filled outer shell electronic configuration of eight electrons (octet of electrons) with a general represent ...
... The negative and positive ions so formed attains stable noble gas electronic configurations. The noble gases (with the exception of helium which has two electrons in the outermost shell) have filled outer shell electronic configuration of eight electrons (octet of electrons) with a general represent ...
Osmium(VIII) Catalyzed Oxidation of 6-Aminopenicillanic Acid
... The reactions catalyzed by transition metal ions are of great interest [10] due to their involvement in many important industrial processes, such as hydrogenation, carbonylation reactions and also low pressure polymerization of ethylene and propene. The role of ...
... The reactions catalyzed by transition metal ions are of great interest [10] due to their involvement in many important industrial processes, such as hydrogenation, carbonylation reactions and also low pressure polymerization of ethylene and propene. The role of ...
National German competition
... In contrast to pure water that has a pH of 7, rainwater reacts to show a slightly acid reaction because of dissolved carbon dioxide. Some of the reasons for this phenomenon are natural and some are caused by man. In air, sulfur dioxide and nitrogen monooxide are oxidized to sulfur trioxide and nitro ...
... In contrast to pure water that has a pH of 7, rainwater reacts to show a slightly acid reaction because of dissolved carbon dioxide. Some of the reasons for this phenomenon are natural and some are caused by man. In air, sulfur dioxide and nitrogen monooxide are oxidized to sulfur trioxide and nitro ...
Document
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
TRO Chapter 4
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
Hydrated metal ions in aqueous solution: How regular are their
... Experimental hydration dynamics and theoretical simulations of hydrated metal ions will not be presented or discussed as it lies outside the scope of this paper even though they give a very important contribution to the understanding of the physical-chemical properties of hydrated metal ions in aque ...
... Experimental hydration dynamics and theoretical simulations of hydrated metal ions will not be presented or discussed as it lies outside the scope of this paper even though they give a very important contribution to the understanding of the physical-chemical properties of hydrated metal ions in aque ...
Analytical Chemistry - University of Delhi
... 1. Estimation of sodium carbonate and sodium hydrogen carbonate present in a mixture. 2. Estimation of oxalic acid by titrating it with KMnO4. 3. Estimation of water of crystallization in Mohr’s salt by titrating with KMnO4. 4. Estimation of Fe (II) ions by titrating it with K2Cr2O7 using internal i ...
... 1. Estimation of sodium carbonate and sodium hydrogen carbonate present in a mixture. 2. Estimation of oxalic acid by titrating it with KMnO4. 3. Estimation of water of crystallization in Mohr’s salt by titrating with KMnO4. 4. Estimation of Fe (II) ions by titrating it with K2Cr2O7 using internal i ...
Syllabus Advanced Level and Advanced Subsidiary Level
... It is intended that candidates should be directed towards the practice of experimental skills throughout the whole period of their course of study. Candidates’ experimental skills will be tested in papers 3 and 5. Paper 3 is a practical examination that will test the skills of manipulation of appara ...
... It is intended that candidates should be directed towards the practice of experimental skills throughout the whole period of their course of study. Candidates’ experimental skills will be tested in papers 3 and 5. Paper 3 is a practical examination that will test the skills of manipulation of appara ...
Thermodynamics of Ion Association in the Saturated Solution of
... diphenylamine sulfonate in the mixed solvent (ethanol+water) from solubility and the mean activity coefficient values obtained at 298.15K. The solubility of solutes in mixed solvents depends primarily on the solvation of the solutes or their constituent ions by the components of the solvent mixtures ...
... diphenylamine sulfonate in the mixed solvent (ethanol+water) from solubility and the mean activity coefficient values obtained at 298.15K. The solubility of solutes in mixed solvents depends primarily on the solvation of the solutes or their constituent ions by the components of the solvent mixtures ...
1.7AMINES
... 1. Amines have an unpleasant odours. (the odour of decaying fish is attributed to simple amines). 2. Because of the hydrogen bonding amines have higher BP than hydrocarbons of similar molar mass. Since a nitrogen—hydrogen bond is less polar than an oxygen—hydrogen bond, the hydrogen bonding in prima ...
... 1. Amines have an unpleasant odours. (the odour of decaying fish is attributed to simple amines). 2. Because of the hydrogen bonding amines have higher BP than hydrocarbons of similar molar mass. Since a nitrogen—hydrogen bond is less polar than an oxygen—hydrogen bond, the hydrogen bonding in prima ...
Decrease = stress More Fe(OH) 2 dissolves in response Solubility
... Common Ion Effect and Solubility • Consider the solubility equilibrium of AgCl. AgCl(s) <=> Ag+(aq) + Cl-(aq) • How does adding excess NaCl affect the solubility equilibrium? NaCl(s) Na+(aq) + Cl-(aq) 2 sources of ClCl- is common ion ...
... Common Ion Effect and Solubility • Consider the solubility equilibrium of AgCl. AgCl(s) <=> Ag+(aq) + Cl-(aq) • How does adding excess NaCl affect the solubility equilibrium? NaCl(s) Na+(aq) + Cl-(aq) 2 sources of ClCl- is common ion ...
Chapter 4 Chemical Quantities and Aqueous Reactions
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
... other ionic compounds, like AgCl, dissolve hardly at all in water at room temperature compounds that dissolve in a solvent are said to be soluble, while those that do not are said to be insoluble NaCl is soluble in water, AgCl is insoluble in water the degree of solubility depends on the tempera ...
ammines ncert solution
... N−atom is delocalized over the benzene ring. In C6H5CH2NH2, N is not directly attached to the benzene ring. Thus, its lone pair is not delocalized over the benzene ring. Therefore, the electrons on the N atom are more easily available for protonation in C6H5CH2NH2 than in C6H5NH2 i.e., C6H5CH2NH2 is ...
... N−atom is delocalized over the benzene ring. In C6H5CH2NH2, N is not directly attached to the benzene ring. Thus, its lone pair is not delocalized over the benzene ring. Therefore, the electrons on the N atom are more easily available for protonation in C6H5CH2NH2 than in C6H5NH2 i.e., C6H5CH2NH2 is ...
EQUILIBRIUM
... 1 point is earned for determining initial number of moles of HF and OH1 point is earned for setting up and doing correct subtraction mol F- formed = mol NaOH added = 0.0060 mol F0.0060 mol F- / (0.015 + 0.025) L of solution = 0.15 M F1 point is earned for determining the number of moles of F1 point ...
... 1 point is earned for determining initial number of moles of HF and OH1 point is earned for setting up and doing correct subtraction mol F- formed = mol NaOH added = 0.0060 mol F0.0060 mol F- / (0.015 + 0.025) L of solution = 0.15 M F1 point is earned for determining the number of moles of F1 point ...
Solutions
... One of the main difficulties in the study of solutions is the wide variety of variables and units used to specify concentration (in the case of ideal mixtures, only molar fractions, and sometimes mass fractions, were common). Generically speaking, concentrations in a mixture express the quantitative ...
... One of the main difficulties in the study of solutions is the wide variety of variables and units used to specify concentration (in the case of ideal mixtures, only molar fractions, and sometimes mass fractions, were common). Generically speaking, concentrations in a mixture express the quantitative ...
EQUILIBRIUM
... 1 point is earned for determining initial number of moles of HF and OH1 point is earned for setting up and doing correct subtraction mol F- formed = mol NaOH added = 0.0060 mol F0.0060 mol F- / (0.015 + 0.025) L of solution = 0.15 M F1 point is earned for determining the number of moles of F1 point ...
... 1 point is earned for determining initial number of moles of HF and OH1 point is earned for setting up and doing correct subtraction mol F- formed = mol NaOH added = 0.0060 mol F0.0060 mol F- / (0.015 + 0.025) L of solution = 0.15 M F1 point is earned for determining the number of moles of F1 point ...
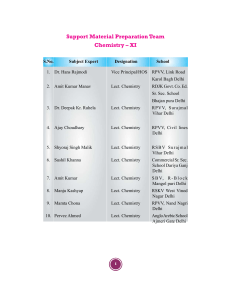
Support Material
... occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of ®rst element of the group, Boron-physical and chemical properties, some important compounds, Borax, Boric acid, Boron Hydrides, Aluminium: Reactions with acids and alkalies, uses. Group 14 E ...
... occurrence, variation of properties, oxidation states, trends in chemical reactivity, anomalous properties of ®rst element of the group, Boron-physical and chemical properties, some important compounds, Borax, Boric acid, Boron Hydrides, Aluminium: Reactions with acids and alkalies, uses. Group 14 E ...
Problem 1-2 - IPN-Kiel
... iv) Calcium Which acid/base properties do the aqueous solutions of the products show? ...
... iv) Calcium Which acid/base properties do the aqueous solutions of the products show? ...
Spring 2005
... 14. (3 pts) What is the oxidation number (charge) on C in H2C2O4? Explain your reasoning. 15. (8 pts) How many atoms of nitrogen are there in 15.3 mg of Ba(NO3)2? 16. (8 pts) What is the mass percent of oxygen in KMnO4? 17. (8 pts) The balanced reaction of muriatic acid with lime is 2 HCl + CaO ! Ca ...
... 14. (3 pts) What is the oxidation number (charge) on C in H2C2O4? Explain your reasoning. 15. (8 pts) How many atoms of nitrogen are there in 15.3 mg of Ba(NO3)2? 16. (8 pts) What is the mass percent of oxygen in KMnO4? 17. (8 pts) The balanced reaction of muriatic acid with lime is 2 HCl + CaO ! Ca ...
- Kendriya Vidyalaya No. 2 Raipur
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...