- Kendriya Vidyalaya No. 2 Raipur
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...
TRIPURA UNIVERSITY SURYAMANINAGAR SYLLABUS FOR B.Sc THREE-YEAR DEGREE (GENERAL AND HONOURS) COURSE
... Determination of concentration of each of HCl and CH3COOH in a mixture conductometrically using standard NaOH solution. ...
... Determination of concentration of each of HCl and CH3COOH in a mixture conductometrically using standard NaOH solution. ...
Topic 9 Oxidation and Reduction Answers - slider-dpchemistry-11
... Rule/s: Three rules are used here. Firstly, hydrogen always has an oxidation of +1 (except in combination with reactive metals such as Na when it is -1). Secondly, oxygen always has an oxidation state of –2 (except in H2O2 where it is -1). These known values are used first. Finally, as all these mol ...
... Rule/s: Three rules are used here. Firstly, hydrogen always has an oxidation of +1 (except in combination with reactive metals such as Na when it is -1). Secondly, oxygen always has an oxidation state of –2 (except in H2O2 where it is -1). These known values are used first. Finally, as all these mol ...
www.xtremepapers.net
... Paper 1 This paper will consist of 40 questions, thirty of the direct choice type and ten of the multiple completion type, all with four options. All questions will be based on the AS syllabus. Candidates will answer all questions. Paper 2 This paper will consist of a variable number of structured q ...
... Paper 1 This paper will consist of 40 questions, thirty of the direct choice type and ten of the multiple completion type, all with four options. All questions will be based on the AS syllabus. Candidates will answer all questions. Paper 2 This paper will consist of a variable number of structured q ...
Stabilization of Quinapril by Incorporating Hydrogen Bonding
... pattern in the former case. In view of the above, attempts have been made to design a system wherein addition of a molecule could improve hydrogen bonding which in turn could impart stability to quinapril hydrochloride, preferably the free base. One such approach is to form a ‘co-crystal’ with a sui ...
... pattern in the former case. In view of the above, attempts have been made to design a system wherein addition of a molecule could improve hydrogen bonding which in turn could impart stability to quinapril hydrochloride, preferably the free base. One such approach is to form a ‘co-crystal’ with a sui ...
Instructor`s Resource Manual
... General Chemistry, Eighth Edition, is designed to give the instructor the greatest flexibility in creating a course for his or her students and to make the process of teaching with the text as smooth as possible. The careful, logical, and clear development of material in each chapter, with its appro ...
... General Chemistry, Eighth Edition, is designed to give the instructor the greatest flexibility in creating a course for his or her students and to make the process of teaching with the text as smooth as possible. The careful, logical, and clear development of material in each chapter, with its appro ...
Moles Class Packet Unit 2
... chemical compound can be represented by a specific chemical formula and assigned a name based on the IUPAC system. (3.1cc) Types of chemical formulas include empirical, molecular, and structural. (3.1ee) The empirical formula of a compound is the simplest whole-number ratio of atoms of the elements ...
... chemical compound can be represented by a specific chemical formula and assigned a name based on the IUPAC system. (3.1cc) Types of chemical formulas include empirical, molecular, and structural. (3.1ee) The empirical formula of a compound is the simplest whole-number ratio of atoms of the elements ...
Honors Chemistry Lab Fall
... across the entire surface, and it will continue to do so until a complete monolayer of stearic acid has been formed. If your rst few drops do not spread and evaporate quickly, either your water or watch glass is still dirty. ...
... across the entire surface, and it will continue to do so until a complete monolayer of stearic acid has been formed. If your rst few drops do not spread and evaporate quickly, either your water or watch glass is still dirty. ...
Study Material - Class- XI- Chemistry
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...
... have been prepared artificially. Elements are further classified into metals, non-metals and metalloids. Compounds A compound is a pure substance made up of two or more elements combined in a definite proportion by mass, which could be split by suitable chemical methods. Characteristics of compound ...
Chemical Reactions
... same number of particles. • Moles are numbers of particles • You can treat reactions as if they happen liters at a time, as long as you keep the temperature and pressure the same. ...
... same number of particles. • Moles are numbers of particles • You can treat reactions as if they happen liters at a time, as long as you keep the temperature and pressure the same. ...
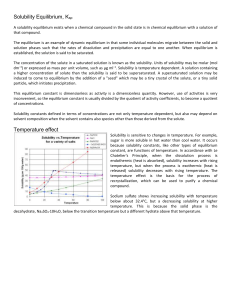
At equilibrium
... At any given temperature the equilibrium pressure of CO2 (/atm) is equal to the equilibrium constant K and is independent of the relative amounts of CaO and CaCO3 present. K increases with T (=> endothermic reaction) and exceeds 1 at above 1000oC (--> equilm pCO2 > 1 atm). It is then simply necessar ...
... At any given temperature the equilibrium pressure of CO2 (/atm) is equal to the equilibrium constant K and is independent of the relative amounts of CaO and CaCO3 present. K increases with T (=> endothermic reaction) and exceeds 1 at above 1000oC (--> equilm pCO2 > 1 atm). It is then simply necessar ...
Chapter 19
... The chemistry of oxidation-reduction reactions is not limited to atoms of an element changing to ions or the reverse. Some redox reactions involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents th ...
... The chemistry of oxidation-reduction reactions is not limited to atoms of an element changing to ions or the reverse. Some redox reactions involve changes in molecular substances or polyatomic ions in which atoms are covalently bonded to other atoms. For example, the following equation represents th ...
Low-Temperature Alkaline pH Hydrolysis of Oxygen-Free
... nitrogen-rich atmosphere is the site of important organic chemistry. This paper focuses on the organic aerosols produced in Titan’s atmosphere that play an important role in atmospheric and surface processes and in organic chemistry as it applies to exobiological interests. To produce reliable labor ...
... nitrogen-rich atmosphere is the site of important organic chemistry. This paper focuses on the organic aerosols produced in Titan’s atmosphere that play an important role in atmospheric and surface processes and in organic chemistry as it applies to exobiological interests. To produce reliable labor ...
Reactions of first-row transition metal ions with propargyl alcohol in
... Transition metal ions are reactive toward many organic substrates because their valence electron configurations make it possible for the metal ions to insert into organic bonds.8 Following insertion, migration of hydrogen and other functional groups occurs to yield new organic species. It has been p ...
... Transition metal ions are reactive toward many organic substrates because their valence electron configurations make it possible for the metal ions to insert into organic bonds.8 Following insertion, migration of hydrogen and other functional groups occurs to yield new organic species. It has been p ...
Synthesis and Structural Studies of Calcium and Magnesium
... This enormous research project would not have been possible without the grace of God and able leadership and support of two distinguished professors: Dr. Karin Ruhlandt and Dr. Jon Zubieta of the Chemistry Department of Syracuse University, who nurtured the seed for the bone project. I am especially ...
... This enormous research project would not have been possible without the grace of God and able leadership and support of two distinguished professors: Dr. Karin Ruhlandt and Dr. Jon Zubieta of the Chemistry Department of Syracuse University, who nurtured the seed for the bone project. I am especially ...