친환경 촉매 Iron (III) phosphate: 실온/무용매 반응조건에서 알코올과
... Also, isoamyl acetate is a kind of flavor reagent with fruit taste. It is traditionally prepared with H2SO4 as catalyst.7 The use of H2SO4 often causes the problems such as corrosion for equipments and pollution for environment. Until now, the tried replaces include FeCl3, CuSO4, ferric tri-dodecane ...
... Also, isoamyl acetate is a kind of flavor reagent with fruit taste. It is traditionally prepared with H2SO4 as catalyst.7 The use of H2SO4 often causes the problems such as corrosion for equipments and pollution for environment. Until now, the tried replaces include FeCl3, CuSO4, ferric tri-dodecane ...
H 2
... product that is not easily observed. An equation is a chemical recipe. If it isn’t balanced, it is like a recipe that doesn’t include any amounts. It would be useless to try to make something from such a recipe. ...
... product that is not easily observed. An equation is a chemical recipe. If it isn’t balanced, it is like a recipe that doesn’t include any amounts. It would be useless to try to make something from such a recipe. ...
AP Chemistry Syllabus – Joliet Township High School
... Emphasis is placed on depth of understanding of a topic, rather than breadth of topics. In addition, students will spend at least five hours a week studying outside of class. ...
... Emphasis is placed on depth of understanding of a topic, rather than breadth of topics. In addition, students will spend at least five hours a week studying outside of class. ...
Teacher Background - Online Learning Exchange
... amount of a product to expect. Chemists use balanced chemical equations as a basis for calculating how much reactant is needed or how much product will be formed in a reaction. When you know the quantity of one substance in a reaction, you can calculate the quantity of any other substance consumed o ...
... amount of a product to expect. Chemists use balanced chemical equations as a basis for calculating how much reactant is needed or how much product will be formed in a reaction. When you know the quantity of one substance in a reaction, you can calculate the quantity of any other substance consumed o ...
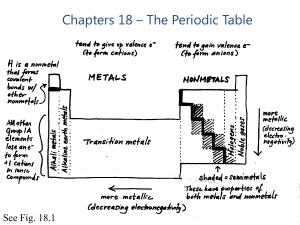
Chapters 18 – The Periodic Table
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
... agent such as potassium chlorate together with tetraphosphorus trisulfide (P4S3), glass and binder. The phosphorus sulfide is easily ignited, the potassium chlorate decomposes to give oxygen, which in turn causes the phosphorus sulfide to burn more vigorously. The head of safety matches are made of ...
1. The compound which could act both as oxidising as well as
... A mixture of Na2C2O4 (A) and KHC2O4 . H2C2O4 (B) required equal volumes of 0.1 M KMnO4 and 0.1 M NaOH, separately. Molar ratio of A and B in this mixture is (a) 1 : 1 (b) 1 : 5.5 (c) 5.5 : 1 (d) 3.1 : 1 3 mole of a mixture of FeSO4 and Fe2(SO4)3 required 100 ml. of 2 M KMnO4 solution in acidic mediu ...
... A mixture of Na2C2O4 (A) and KHC2O4 . H2C2O4 (B) required equal volumes of 0.1 M KMnO4 and 0.1 M NaOH, separately. Molar ratio of A and B in this mixture is (a) 1 : 1 (b) 1 : 5.5 (c) 5.5 : 1 (d) 3.1 : 1 3 mole of a mixture of FeSO4 and Fe2(SO4)3 required 100 ml. of 2 M KMnO4 solution in acidic mediu ...
- Catalyst
... Step 1) Assign oxidation numbers to all elements in the equation. Step 2) From the changes in oxidation numbers, identify the oxidized and reduced species. Step 3) Compute the number of electrons lost in the oxidation and gained in the reduction from the oxidation number changes. Draw tie-lines betw ...
... Step 1) Assign oxidation numbers to all elements in the equation. Step 2) From the changes in oxidation numbers, identify the oxidized and reduced species. Step 3) Compute the number of electrons lost in the oxidation and gained in the reduction from the oxidation number changes. Draw tie-lines betw ...
chemical reactions and energy changes
... different concentrations of sugar. Concentration can be specified in a number of ways, one of which would be the mass of dissolved sugar in a particular volume of solution, i.e. grams per litre. However, chemical reactions are more conveniently expressed as the number of moles (abbreviated as mol) o ...
... different concentrations of sugar. Concentration can be specified in a number of ways, one of which would be the mass of dissolved sugar in a particular volume of solution, i.e. grams per litre. However, chemical reactions are more conveniently expressed as the number of moles (abbreviated as mol) o ...
GCE Chemistry Question Paper Unit 05 - Energetics, Redox
... The diagram shows a non-rechargeable cell that can be used to power electronic devices. The relevant half-equations for this cell are equations 2 and 4 in the table above. ...
... The diagram shows a non-rechargeable cell that can be used to power electronic devices. The relevant half-equations for this cell are equations 2 and 4 in the table above. ...
Answers - Scioly.org
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
... 20. The student concludes that she has synthesized ethyl butanoate. Use evidence from the two experiments to support or to refute her claim. The peak of highest mass to charge ratio is approximately 116; therefore, the unknown molecule would have a molecular mass of 116. Ethyl butanoate has the chem ...
Part II - KFUPM Faculty List
... Thermodynamics of Living Systems Thermodynamics have a great effect in biological sciences, such as processes taking place inside our bodies. such as processes taking place inside our bodies. Many chemical reactions carried out inside the body (such as DNA and protein formation) are not sponta ...
... Thermodynamics of Living Systems Thermodynamics have a great effect in biological sciences, such as processes taking place inside our bodies. such as processes taking place inside our bodies. Many chemical reactions carried out inside the body (such as DNA and protein formation) are not sponta ...
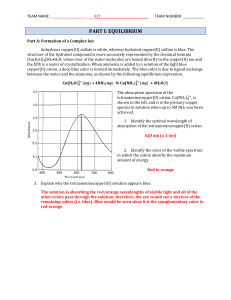
Chapter 17 lecture notes on Chemical Equilibria
... value of Q starts off very small (reactants in denominator) compared to the final K value. Q increases until the point that the forward and reverse reaction rates are equal, and the system is at equilibrium; then Kc can be calculated. We can establish the following rules for describing where the rea ...
... value of Q starts off very small (reactants in denominator) compared to the final K value. Q increases until the point that the forward and reverse reaction rates are equal, and the system is at equilibrium; then Kc can be calculated. We can establish the following rules for describing where the rea ...
www.xtremepapers.net
... There are forty questions on this paper. Answer all questions. For each question there are four possible answers A, B, C, and D. Choose the one you consider correct and record your choice in soft pencil on the separate answer sheet. Read the instructions on the answer sheet very carefully. Each corr ...
... There are forty questions on this paper. Answer all questions. For each question there are four possible answers A, B, C, and D. Choose the one you consider correct and record your choice in soft pencil on the separate answer sheet. Read the instructions on the answer sheet very carefully. Each corr ...
welcome to ap chemistry - Garnet Valley School District
... I am delighted that you have chosen to take AP chemistry this fall. It is a challenging course and a strong foundation is necessary for your success. The material included in the summer work packet will enable you to cover the basics of chemistry, which are covered in chapters 1, 2, 3 and 4 of the t ...
... I am delighted that you have chosen to take AP chemistry this fall. It is a challenging course and a strong foundation is necessary for your success. The material included in the summer work packet will enable you to cover the basics of chemistry, which are covered in chapters 1, 2, 3 and 4 of the t ...
Chemistry - NIC Karnataka
... Fundamental concepts in organic reactions:mechanism – definition , fission of covalent bond – homolytic and heterolytic, carbanion, carbocation, alkyl free radicals, examples.Compare the stabilities of 1°, 2°, 3° carbocations and alkyl free radicals. Nucleophiles and electrophiles, examples. Electro ...
... Fundamental concepts in organic reactions:mechanism – definition , fission of covalent bond – homolytic and heterolytic, carbanion, carbocation, alkyl free radicals, examples.Compare the stabilities of 1°, 2°, 3° carbocations and alkyl free radicals. Nucleophiles and electrophiles, examples. Electro ...
Oregon State University, Summer 2009 Chemistry 121 Midterm
... This exam consists of 20 multiple-choice questions. Each multiple-choice question has 5 points associated with it. Select the best answer by filling in the corresponding circle on the rear page of the answer sheet. If you have any questions before the exam, please ask. If you have any questions duri ...
... This exam consists of 20 multiple-choice questions. Each multiple-choice question has 5 points associated with it. Select the best answer by filling in the corresponding circle on the rear page of the answer sheet. If you have any questions before the exam, please ask. If you have any questions duri ...
Chemistry 211 - George Mason University
... – Mass: measured in grams; tells how much of an object there is; related to weight, which is the gravitational pull on the object. – Length: measured in meters – Temperature: measured in K or °C. Each is based upon the same reference temperatures: freezing and boiling point of water. • Conversion fr ...
... – Mass: measured in grams; tells how much of an object there is; related to weight, which is the gravitational pull on the object. – Length: measured in meters – Temperature: measured in K or °C. Each is based upon the same reference temperatures: freezing and boiling point of water. • Conversion fr ...
Document
... The First Law: Thermochemistry • For example, it is impractical to measure the heat evolved when carbon burns to carbon monoxide in a limited amount of oxygen. However, one can measure the heat of reaction for carbon to completely burn to carbon dioxide in an excess of oxygen. And heat evolved can ...
... The First Law: Thermochemistry • For example, it is impractical to measure the heat evolved when carbon burns to carbon monoxide in a limited amount of oxygen. However, one can measure the heat of reaction for carbon to completely burn to carbon dioxide in an excess of oxygen. And heat evolved can ...
Chemical Equations
... Thus, the same collection of atoms is present after a reaction as before the reaction. The changes that occur during a reaction just involve the rearrangement of atoms. A chemical equation shows how the atoms are rearranged. ...
... Thus, the same collection of atoms is present after a reaction as before the reaction. The changes that occur during a reaction just involve the rearrangement of atoms. A chemical equation shows how the atoms are rearranged. ...
Final Exam - Dawson College
... Limestone, CaCO3, reacts with hydrochloric acid to form calcium chloride, water and carbon dioxide according to the following reaction: CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g) How many liters of CO2 gas will be formed at 755 torr and 33.0°C by the reaction of 2.35 g of limestone with an ex ...
... Limestone, CaCO3, reacts with hydrochloric acid to form calcium chloride, water and carbon dioxide according to the following reaction: CaCO3(s) + 2HCl(aq) → CaCl2(aq) + H2O(l) + CO2(g) How many liters of CO2 gas will be formed at 755 torr and 33.0°C by the reaction of 2.35 g of limestone with an ex ...
unit (4) calculations and chemical reactions
... A reversible reaction proceeds in both the forward and a reverse direction. The forward reaction is called “the reaction to the right”, and the reverse reaction is called “the reaction to the left.” Let’s examine a typical reversible reaction using the equation above. When we add compound A to compo ...
... A reversible reaction proceeds in both the forward and a reverse direction. The forward reaction is called “the reaction to the right”, and the reverse reaction is called “the reaction to the left.” Let’s examine a typical reversible reaction using the equation above. When we add compound A to compo ...
Energetics - WordPress.com
... ΔH = +96.0 kJ mol-1 This is interesting not only because it is a reaction between two solids, but also because the temperature decrease is such that the flask will often stick to the bench after the reaction. When there is a change of state from solid to a liquid, or a liquid to a gas at a const ...
... ΔH = +96.0 kJ mol-1 This is interesting not only because it is a reaction between two solids, but also because the temperature decrease is such that the flask will often stick to the bench after the reaction. When there is a change of state from solid to a liquid, or a liquid to a gas at a const ...