Section 4.8
... consumed and limits amount of product • Reactant in excess – reactant present in greater quantity than limiting reactant • Theoretical Yield – amount of product made based on consumption of all the limiting reactant • Actual Yield – amount of product actually produced • Percent Yield – (actual/theor ...
... consumed and limits amount of product • Reactant in excess – reactant present in greater quantity than limiting reactant • Theoretical Yield – amount of product made based on consumption of all the limiting reactant • Actual Yield – amount of product actually produced • Percent Yield – (actual/theor ...
Chapter 2 Geochemical Reactions
... ionic bond in minerals, it differs in that the anion and cation are separated by the surrounding hydration sheath of water molecules. Electrostatic bonding also occurs between ions and charged surfaces such as clays and solid organic matter. ...
... ionic bond in minerals, it differs in that the anion and cation are separated by the surrounding hydration sheath of water molecules. Electrostatic bonding also occurs between ions and charged surfaces such as clays and solid organic matter. ...
Full Text PDF
... the second group μeffisbgerthanμSo,.p-biculngrsoyfthe ions of the second group. This trend can be easily justified if one supposes that kSE increases as the density of unpaired metal electrons increases at the collision site between complex and Ps atom (i.e. at the _complex boundaries and that the e ...
... the second group μeffisbgerthanμSo,.p-biculngrsoyfthe ions of the second group. This trend can be easily justified if one supposes that kSE increases as the density of unpaired metal electrons increases at the collision site between complex and Ps atom (i.e. at the _complex boundaries and that the e ...
- Angelo State University
... • In the examples we’ve seen, we have assumed that all of the reactions “go to completion” — that is, that all reactant molecules are converted into products. In real life, some product is almost always lost due to small amounts of contamination present in the glassware, impurities in the reactants, ...
... • In the examples we’ve seen, we have assumed that all of the reactions “go to completion” — that is, that all reactant molecules are converted into products. In real life, some product is almost always lost due to small amounts of contamination present in the glassware, impurities in the reactants, ...
Higher Chemistry Resources Guide - Glow Blogs
... attraction that can operate between all atoms and molecules. These forces are much weaker than all other types of bonding. They are formed as a result of electrostatic attraction between temporary dipoles and induced dipoles caused by movement of electrons in atoms and molecules. The strength of Lon ...
... attraction that can operate between all atoms and molecules. These forces are much weaker than all other types of bonding. They are formed as a result of electrostatic attraction between temporary dipoles and induced dipoles caused by movement of electrons in atoms and molecules. The strength of Lon ...
Physical Chemistry 1.pdf
... of thermodynamics, chemical kinetics, and kinetic molecular theory. In our study of thermodynamics (thermodynamics comes from the Greek words for “heat” and “power”), we shall be looking at the first and second laws of thermodynamics as they apply to volume-pressure processes and energy changes asso ...
... of thermodynamics, chemical kinetics, and kinetic molecular theory. In our study of thermodynamics (thermodynamics comes from the Greek words for “heat” and “power”), we shall be looking at the first and second laws of thermodynamics as they apply to volume-pressure processes and energy changes asso ...
4.6 M - Thierry Karsenti
... Entropy (S). A direct measure of the degree of disorder or randomness of a system. Equilibrium. A condition in which an infinitesimal change in a variable in the opposite direction results in opposite change in the state. In chemical reactions, it represents the situation in which the reactants and ...
... Entropy (S). A direct measure of the degree of disorder or randomness of a system. Equilibrium. A condition in which an infinitesimal change in a variable in the opposite direction results in opposite change in the state. In chemical reactions, it represents the situation in which the reactants and ...
Power Point for Equilibrium
... The Concept of Equilibrium • Consider colorless frozen N2O4. At room temperature, it decomposes to brown NO2: N2O4(g) 2NO2(g). • At some time, the color stops changing and we have a mixture of N2O4 and NO2. • Chemical equilibrium is the point at which the rate of the forward reaction is equal to ...
... The Concept of Equilibrium • Consider colorless frozen N2O4. At room temperature, it decomposes to brown NO2: N2O4(g) 2NO2(g). • At some time, the color stops changing and we have a mixture of N2O4 and NO2. • Chemical equilibrium is the point at which the rate of the forward reaction is equal to ...
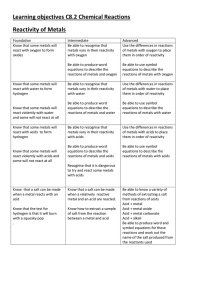
Learning objectives C8.2 Chemical Reactions Reactivity of Metals
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
... methods of extracting a salt from reactions of acids Acid + metal Acid + metal oxide Acid + metal carbonate Acid + alkali Be able to produce word and symbol equations for these reactions and work out the name of the salt produced from the reactants used ...
Reaction Kinetics - National Open University of Nigeria
... CHM 407: Reaction Kinetics concerns with the speed or rates of chemical reactions. The study of reaction rates allows for the prediction of how fast it will take a reaction mixture to reach equilibrium. It also account for how the reaction rate would be optimised by controlling certain factors such ...
... CHM 407: Reaction Kinetics concerns with the speed or rates of chemical reactions. The study of reaction rates allows for the prediction of how fast it will take a reaction mixture to reach equilibrium. It also account for how the reaction rate would be optimised by controlling certain factors such ...
ap 2005 chemistry_b scoring guidelines - AP Central
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
... Write the formulas to show the reactants and the products for any FIVE of the laboratory situations described below. Answers to more than five choices will not be graded. In all cases, a reaction occurs. Assume that solutions are aqueous unless otherwise indicated. Represent substances in solution a ...
AP® Chemistry
... Many students wonder whether or not to guess the answers to the multiple-choice questions about which they are not certain. It is improbable that mere guessing will improve a score. However, if a student has some knowledge of the question and is able to eliminate one or more answer choices as wrong, ...
... Many students wonder whether or not to guess the answers to the multiple-choice questions about which they are not certain. It is improbable that mere guessing will improve a score. However, if a student has some knowledge of the question and is able to eliminate one or more answer choices as wrong, ...
3 CO 2 (g) + 4 H 2 O(l)
... When we heat a chemical in isolation (i.e. when no reaction occurs) two things can happen. Either the chemical will simply get hotter without changing its phase or at a certain temperature the species will undergo a phase change. In chapter 1, we quantified the amount of products that could be expec ...
... When we heat a chemical in isolation (i.e. when no reaction occurs) two things can happen. Either the chemical will simply get hotter without changing its phase or at a certain temperature the species will undergo a phase change. In chapter 1, we quantified the amount of products that could be expec ...
Chapter 9 Lota_2 Dæmi A4 Varmafræði
... pitcher reach a final temperature? The tea is mostly water, so assume that it has the same density (1.0 g/mL), molar mass, heat capacity (75.3 J K–1 mol–1), and heat of fusion (6.0 kJ/mol) as pure water. The heat capacity of ice is 37.7 J K–1 ...
... pitcher reach a final temperature? The tea is mostly water, so assume that it has the same density (1.0 g/mL), molar mass, heat capacity (75.3 J K–1 mol–1), and heat of fusion (6.0 kJ/mol) as pure water. The heat capacity of ice is 37.7 J K–1 ...
Harrisburg Area Community College 2013/2014
... instead of holding the paperclip in my hand, because the balance is supposedly better. But be careful; the assumption that I should get a better value from the balance implies a whole bunch of other factors (such as calibrating the balance and using the balance correctly. Two general terms used to d ...
... instead of holding the paperclip in my hand, because the balance is supposedly better. But be careful; the assumption that I should get a better value from the balance implies a whole bunch of other factors (such as calibrating the balance and using the balance correctly. Two general terms used to d ...
Redox Reactions - Hillsborough County Public Schools
... Replacement Reactions are always redox reactions! Combustion reactions are always redox reactions! Any time an oxidation number changes (which means electrons are gained or lost) during the reaction, a redox reaction is occurring. ...
... Replacement Reactions are always redox reactions! Combustion reactions are always redox reactions! Any time an oxidation number changes (which means electrons are gained or lost) during the reaction, a redox reaction is occurring. ...
Energetics - chemistryatdulwich
... size of ion: the smaller the ion , the larger the electrostatic attraction and the more energy is needed to sublime the ions. charge of the ion: the larger the charge, the greater the electrostatic attraction. The smaller the ionic radius and the greater the charge of the ion the greater the charg ...
... size of ion: the smaller the ion , the larger the electrostatic attraction and the more energy is needed to sublime the ions. charge of the ion: the larger the charge, the greater the electrostatic attraction. The smaller the ionic radius and the greater the charge of the ion the greater the charg ...
Deans Community High School Intermediate 2 Revision Notes www
... When writing a word equation, we use the symbol (meaning 'reacts to give') and not the = sign. The reason is that if we used an equal sign, we are saying that the products are the same as the reactants. This is not the case, as all chemical reactions produce a new substance. For example, the reactio ...
... When writing a word equation, we use the symbol (meaning 'reacts to give') and not the = sign. The reason is that if we used an equal sign, we are saying that the products are the same as the reactants. This is not the case, as all chemical reactions produce a new substance. For example, the reactio ...
1984 Advanced Placement Exam
... effect on this reaction? (A) The rate of reaction increases. (B) The rate of reaction decreases. (C) The value of the equilibrium constant increases. (D) The value of the equilibrium constant decreases. (E) Neither the rate nor the value of the equilibrium constant is changed. 27. The critical tempe ...
... effect on this reaction? (A) The rate of reaction increases. (B) The rate of reaction decreases. (C) The value of the equilibrium constant increases. (D) The value of the equilibrium constant decreases. (E) Neither the rate nor the value of the equilibrium constant is changed. 27. The critical tempe ...
honors chemistry harvard-westlake second semester final exam
... Li is a smaller atom than Na. 10. The hybrid orbitals of the carbon atom in methane, CH4 , are called a. sp b. sp2 c. sp3 d. sp4 11. Of the following molecules, all of which have polar bonds, the non-polar molecule is a. HCl (linear) b. H2O (v-shaped) c. NH3 (trigonal pyramid) d. CCl4 (tetrahedron) ...
... Li is a smaller atom than Na. 10. The hybrid orbitals of the carbon atom in methane, CH4 , are called a. sp b. sp2 c. sp3 d. sp4 11. Of the following molecules, all of which have polar bonds, the non-polar molecule is a. HCl (linear) b. H2O (v-shaped) c. NH3 (trigonal pyramid) d. CCl4 (tetrahedron) ...
chemical change
... AN INTRODUCTION TO CHEMICAL REACTIONS EVIDENCE FOR A CHEMICAL REACTION The evidence for a chemical reaction occurring, is the formation of a substance which is different from the original reactant or reactant, this is often accompanied by changes in energy, which are measured as temperature changes. ...
... AN INTRODUCTION TO CHEMICAL REACTIONS EVIDENCE FOR A CHEMICAL REACTION The evidence for a chemical reaction occurring, is the formation of a substance which is different from the original reactant or reactant, this is often accompanied by changes in energy, which are measured as temperature changes. ...