Electronic structure of quantum dots
... The properties of quasi-two-dimensional semiconductor quantum dots are reviewed. Experimental techniques for measuring the electronic shell structure and the effect of magnetic fields are briefly described. The electronic structure is analyzed in terms of simple single-particle models, density-funct ...
... The properties of quasi-two-dimensional semiconductor quantum dots are reviewed. Experimental techniques for measuring the electronic shell structure and the effect of magnetic fields are briefly described. The electronic structure is analyzed in terms of simple single-particle models, density-funct ...
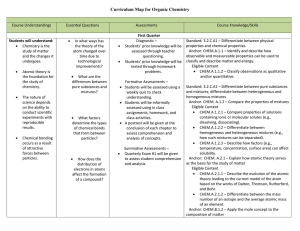
Organic Chemistry Curriculum Map - Belle Vernon Area School District
... CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an element on the periodic table to its electron configuration and compare its reactivity to the reactivity of other elements in the table. ...
... CHEM.A.2.2.1 – Predict the ground state electronic configurations and/or orbital diagram for a given atom or ion. Standard: 3.2.C.A2 – Relate the position of an element on the periodic table to its electron configuration and compare its reactivity to the reactivity of other elements in the table. ...
Quantum Dynamics of Condensates, Atomtronic Systems, and
... physics. It is common to view these interactions through one of two lenses. Either light alters the properties of the atoms or atoms alter the properties of the light. There are several ways of examining the atoms and photons: classically, semiclassically, or quantum mechanically. The system under c ...
... physics. It is common to view these interactions through one of two lenses. Either light alters the properties of the atoms or atoms alter the properties of the light. There are several ways of examining the atoms and photons: classically, semiclassically, or quantum mechanically. The system under c ...
Theoretical studies of chemical dynamics on Charge recombination reactions Sifiso Musa Nkambule
... Sifiso Musa Nkambule ...
... Sifiso Musa Nkambule ...
electron transport in quantum dots. - Leo Kouwenhoven
... single charges. Since these early experiments there have been many successes in the field of metallic junctions which are reviewed in other chapters of this volume. The advent of the scanning tunneling microscope (STM) [27] has renewed interest in Coulomb blockade in small grains. STMs can both imag ...
... single charges. Since these early experiments there have been many successes in the field of metallic junctions which are reviewed in other chapters of this volume. The advent of the scanning tunneling microscope (STM) [27] has renewed interest in Coulomb blockade in small grains. STMs can both imag ...
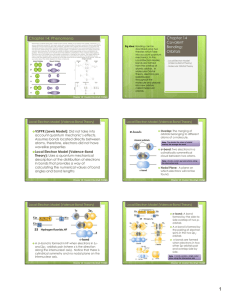
Chapter 14: Phenomena Chapter 14 Covalent Bonding: Orbitals
... Phenomena: Scientists knew that in order to form a bond, orbitals on two atoms must overlap. However, px, py, and pz orbitals are located 90˚ from each other and compounds like CH4 (which would form bonds using their p orbitals) do not have bond angles of 90˚. Therefore, scientists had to explain th ...
... Phenomena: Scientists knew that in order to form a bond, orbitals on two atoms must overlap. However, px, py, and pz orbitals are located 90˚ from each other and compounds like CH4 (which would form bonds using their p orbitals) do not have bond angles of 90˚. Therefore, scientists had to explain th ...
chemistry - Ethiopian Ministry of Education
... The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated by distance. Since chemistry is ...
... The universe is just like a very big chemical laboratory, rearranging atoms and subatomic particles to produce elements and compounds. While planets are made up of rocks which are nothing but arrangement of compounds, an atmosphere is a mixture of compounds separated by distance. Since chemistry is ...
Publication: Electronic properties of liquid ammonia: A sequential
... natural sciences. They are essential in many chemical reactions affecting molecular properties compared to the isolated in-vacuum situation. Among these systems, polar liquids have a special place. In addition, protic liquid systems are very important in physical, chemical, and biological processes. ...
... natural sciences. They are essential in many chemical reactions affecting molecular properties compared to the isolated in-vacuum situation. Among these systems, polar liquids have a special place. In addition, protic liquid systems are very important in physical, chemical, and biological processes. ...
The physics behind chemistry, and the Periodic Table
... The next physical level brings in the quantum electrodynamical (QED) effects. For lightelement problems, such as the hydrogen-atom Lamb shift, precise properties of the hydrogen molecules, or the spectra of the lithium atom, all these effects are already clearly seen, because the accuracy of both th ...
... The next physical level brings in the quantum electrodynamical (QED) effects. For lightelement problems, such as the hydrogen-atom Lamb shift, precise properties of the hydrogen molecules, or the spectra of the lithium atom, all these effects are already clearly seen, because the accuracy of both th ...
Chemistry: Percent Yield
... 17: 3.4e Equal volumes of gases at the same temperature and pressure contain an equal number of particles. 33: 3.2b Types of chemical reactions include synthesis, decomposition, single replacement, and double replacement 36: M1.1C – Use algebraic and geometric representations to describe and compare ...
... 17: 3.4e Equal volumes of gases at the same temperature and pressure contain an equal number of particles. 33: 3.2b Types of chemical reactions include synthesis, decomposition, single replacement, and double replacement 36: M1.1C – Use algebraic and geometric representations to describe and compare ...
Using JCP format
... It should be noted that the derivatives of r MEP and R MEP with respect to ␥ need only be known at discrete values of ␥, so that explicit expressions for these functions are not required. The derivatives are instead calculated numerically, together with the values of r MEP and R MEP , as the MEP is ...
... It should be noted that the derivatives of r MEP and R MEP with respect to ␥ need only be known at discrete values of ␥, so that explicit expressions for these functions are not required. The derivatives are instead calculated numerically, together with the values of r MEP and R MEP , as the MEP is ...
Chapter 1 The Kondo screening cloud: what it is and
... at this length scale represent both an experimental challenge to find sufficiently ideal systems and also an opportunity to study the limits of validity of the basic model. We will be concerned with two types of length dependence. The first type, analysed in Sec. 1.2, involves a single impurity in a ...
... at this length scale represent both an experimental challenge to find sufficiently ideal systems and also an opportunity to study the limits of validity of the basic model. We will be concerned with two types of length dependence. The first type, analysed in Sec. 1.2, involves a single impurity in a ...
High Rydberg states of DABCO: Spectroscopy, ionization potential
... in the extraction region, and a high voltage on an ion reflector in front of the detector. The bias field ~between 0.5 and 4 V/cm! is present either before or shortly ~,100 ns! after the excitation laser pulse arrives. This bias field causes all ions to slowly drift upwards, apart from the neutral m ...
... in the extraction region, and a high voltage on an ion reflector in front of the detector. The bias field ~between 0.5 and 4 V/cm! is present either before or shortly ~,100 ns! after the excitation laser pulse arrives. This bias field causes all ions to slowly drift upwards, apart from the neutral m ...
X-ray photoelectron spectroscopy

X-ray photoelectron spectroscopy (XPS) is a surface-sensitive quantitative spectroscopic technique that measures the elemental composition at the parts per thousand range, empirical formula, chemical state and electronic state of the elements that exist within a material. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 0 to 10 nm of the material being analyzed. XPS requires high vacuum (P ~ 10−8 millibar) or ultra-high vacuum (UHV; P < 10−9 millibar) conditions, although a current area of development is ambient-pressure XPS, in which samples are analyzed at pressures of a few tens of millibar.XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its as-received state, or after some treatment, for example: fracturing, cutting or scraping in air or UHV to expose the bulk chemistry, ion beam etching to clean off some or all of the surface contamination (with mild ion etching) or to intentionally expose deeper layers of the sample (with more extensive ion etching) in depth-profiling XPS, exposure to heat to study the changes due to heating, exposure to reactive gases or solutions, exposure to ion beam implant, exposure to ultraviolet light.XPS is also known as ESCA (Electron Spectroscopy for Chemical Analysis), an abbreviation introduced by Kai Siegbahn's research group to emphasize the chemical (rather than merely elemental) information that the technique provides.In principle XPS detects all elements. In practice, using typical laboratory-scale X-ray sources, XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot easily detect hydrogen (Z = 1) or helium (Z = 2).Detection limits for most of the elements (on a modern instrument) are in the parts per thousand range. Detection limits of parts per million (ppm) are possible, but require special conditions: concentration at top surface or very long collection time (overnight).XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, viscous oils, glues, ion-modified materials and many others.XPS is less routinely used to analyze the hydrated forms of some of the above materials by freezing the samples in their hydrated state in an ultra pure environment, and allowing or causing multilayers of ice to sublime away prior to analysis. Such hydrated XPS analysis allows hydrated sample structures, which may be different from vacuum-dehydrated sample structures, to be studied in their more relevant as-used hydrated structure. Many bio-materials such as hydrogels are examples of such samples.