Appendices and Glossary
... atoms in a molecule because ionic substances are not molecular. However, it still gives the mole ratio of the elements. One mole of NaCl contains one mole of Na1+ ions and one mole of Cl1- ions. Ionic compounds with polyatomic ions are somewhat more complicated. One mole of sodium sulfate, Na2SO4, c ...
... atoms in a molecule because ionic substances are not molecular. However, it still gives the mole ratio of the elements. One mole of NaCl contains one mole of Na1+ ions and one mole of Cl1- ions. Ionic compounds with polyatomic ions are somewhat more complicated. One mole of sodium sulfate, Na2SO4, c ...
Interacting many-body systems in quantum wells: Evidence for
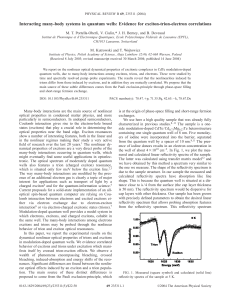
... We use here a high quality sample that was already fully characterized in previous studies.8–11 The sample is a oneside modulation-doped CdTe/ Cd0.73Mg0.27Te heterostructure containing one single quantum well of 8 nm. Five monolayers of iodine were incorporated into the barrier, separated from the q ...
... We use here a high quality sample that was already fully characterized in previous studies.8–11 The sample is a oneside modulation-doped CdTe/ Cd0.73Mg0.27Te heterostructure containing one single quantum well of 8 nm. Five monolayers of iodine were incorporated into the barrier, separated from the q ...
Chapter 4 Elements and Symbols
... Because both systems are currently in use, they are both shown on the periodic table in this text and are included in our discussions of elements and group numbers. ...
... Because both systems are currently in use, they are both shown on the periodic table in this text and are included in our discussions of elements and group numbers. ...
HOMOGENEOUS LINEWIDTH AND SPECTRAL
... 2-5. First three energy levels of En for hydrogenic atoms. ..............................................19 2-6. General energy diagram for insulators and semiconductors....................................21 2-7. a) The single particle picture of an electron and hole in a semiconductor. The electron ...
... 2-5. First three energy levels of En for hydrogenic atoms. ..............................................19 2-6. General energy diagram for insulators and semiconductors....................................21 2-7. a) The single particle picture of an electron and hole in a semiconductor. The electron ...
Answers - Pearson
... Particles are in constant random motion and collide with each other and with the walls of the container in perfectly elastic collisions. The kinetic energy of the particles increases with temperature. There are no inter-particle forces and the volume of the particles is negligible relative to the vo ...
... Particles are in constant random motion and collide with each other and with the walls of the container in perfectly elastic collisions. The kinetic energy of the particles increases with temperature. There are no inter-particle forces and the volume of the particles is negligible relative to the vo ...
Chemical reactivity of ultracold polar molecules: investigation of H+
... The quantum mechanical coupled-channel hyperspherical coordinate method of Skouteris et al. [58] is used to solve the Schrödinger equation for the motion of the three nu- ...
... The quantum mechanical coupled-channel hyperspherical coordinate method of Skouteris et al. [58] is used to solve the Schrödinger equation for the motion of the three nu- ...
Do Neutrino Oscillations Conserve Energy?
... among the full spectrum of energy of neutrinos incident upon it, which permits observation of oscillations of neutrinos from a three-body decay at greater distance from the source than would be possible if the detector merely identified the presence of a neutrino but with no information as to its en ...
... among the full spectrum of energy of neutrinos incident upon it, which permits observation of oscillations of neutrinos from a three-body decay at greater distance from the source than would be possible if the detector merely identified the presence of a neutrino but with no information as to its en ...
Chapter
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
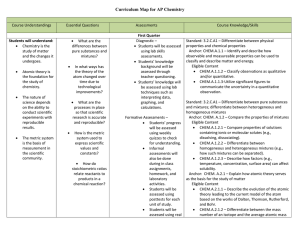
AP Chemistry Curriculum Map - Belle Vernon Area School District
... What are the processes in place so that scientific research is accurate and reproducible? ...
... What are the processes in place so that scientific research is accurate and reproducible? ...
RIKEN Accelerator Progress Report
... ions were investigated. In addition to these atomic collision experiments, study of beam-foil spectroscopy has started this year and its first report is given in this volume. Some theoretical activities also began to work from this year mainly in the field of heavy particle collision, rearrangement ...
... ions were investigated. In addition to these atomic collision experiments, study of beam-foil spectroscopy has started this year and its first report is given in this volume. Some theoretical activities also began to work from this year mainly in the field of heavy particle collision, rearrangement ...
Review Study Guide for the Final
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
... To what volume, in liters, must you dilute a solution containing 4 liters of 0.100 M of Ca(OH)2 to obtain a 0.00100 M solution as calcium hydroxide (Ca(OH)2)? ...
A Biodegradable Polydopamine‐Derived Electrode Material for High
... Inspired by the redox activity of catechol/o-benzoquinone in DA for neuronal communication, we chose PDA as both electrode and binder material for LIBs and SIBs, wherein electrons are transferred from catechol to o-benzoquinone to provide quinone-rich PDA in the optimized oxidation state through the ...
... Inspired by the redox activity of catechol/o-benzoquinone in DA for neuronal communication, we chose PDA as both electrode and binder material for LIBs and SIBs, wherein electrons are transferred from catechol to o-benzoquinone to provide quinone-rich PDA in the optimized oxidation state through the ...
X-ray photoelectron spectroscopy

X-ray photoelectron spectroscopy (XPS) is a surface-sensitive quantitative spectroscopic technique that measures the elemental composition at the parts per thousand range, empirical formula, chemical state and electronic state of the elements that exist within a material. XPS spectra are obtained by irradiating a material with a beam of X-rays while simultaneously measuring the kinetic energy and number of electrons that escape from the top 0 to 10 nm of the material being analyzed. XPS requires high vacuum (P ~ 10−8 millibar) or ultra-high vacuum (UHV; P < 10−9 millibar) conditions, although a current area of development is ambient-pressure XPS, in which samples are analyzed at pressures of a few tens of millibar.XPS is a surface chemical analysis technique that can be used to analyze the surface chemistry of a material in its as-received state, or after some treatment, for example: fracturing, cutting or scraping in air or UHV to expose the bulk chemistry, ion beam etching to clean off some or all of the surface contamination (with mild ion etching) or to intentionally expose deeper layers of the sample (with more extensive ion etching) in depth-profiling XPS, exposure to heat to study the changes due to heating, exposure to reactive gases or solutions, exposure to ion beam implant, exposure to ultraviolet light.XPS is also known as ESCA (Electron Spectroscopy for Chemical Analysis), an abbreviation introduced by Kai Siegbahn's research group to emphasize the chemical (rather than merely elemental) information that the technique provides.In principle XPS detects all elements. In practice, using typical laboratory-scale X-ray sources, XPS detects all elements with an atomic number (Z) of 3 (lithium) and above. It cannot easily detect hydrogen (Z = 1) or helium (Z = 2).Detection limits for most of the elements (on a modern instrument) are in the parts per thousand range. Detection limits of parts per million (ppm) are possible, but require special conditions: concentration at top surface or very long collection time (overnight).XPS is routinely used to analyze inorganic compounds, metal alloys, semiconductors, polymers, elements, catalysts, glasses, ceramics, paints, papers, inks, woods, plant parts, make-up, teeth, bones, medical implants, bio-materials, viscous oils, glues, ion-modified materials and many others.XPS is less routinely used to analyze the hydrated forms of some of the above materials by freezing the samples in their hydrated state in an ultra pure environment, and allowing or causing multilayers of ice to sublime away prior to analysis. Such hydrated XPS analysis allows hydrated sample structures, which may be different from vacuum-dehydrated sample structures, to be studied in their more relevant as-used hydrated structure. Many bio-materials such as hydrogels are examples of such samples.