200 Things to Know to Pass the Chemistry Regents
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
200 Ways to Pass the Chemistry
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
200things2know
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
... 15. Dalton’s model of the atom was a solid sphere of matter that was uniform throughout. 16. The Bohr Model of the atom placed electrons in “planet-like” orbits around the nucleus of an atom. 17. The current, wave-mechanical model of the atom has electrons in “clouds” (orbitals) around the nucleus. ...
Chemistry - Target Publications
... Answers to the two sections are to be written in the same answer book. iii. Figures to the right hand side indicate full marks. iv. Write balanced chemical equations and draw neat and labelled diagrams, wherever necessary. v. Use of logarithmic table is allowed. vi. Answer to every question must be ...
... Answers to the two sections are to be written in the same answer book. iii. Figures to the right hand side indicate full marks. iv. Write balanced chemical equations and draw neat and labelled diagrams, wherever necessary. v. Use of logarithmic table is allowed. vi. Answer to every question must be ...
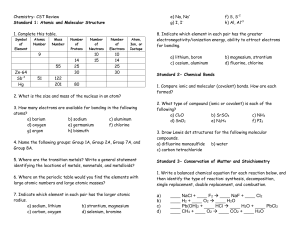
Chemistry- CST Review
... 4. Calculate the molarity of each of the following solutions: a) 0.60 mol of NaCl dissolved in 1.6 L of solution. b) 25.2 g of potassium nitrate, KNO3, in enough water to make 150.0 mL of solution. 5. Calculate the number of grams of solute needed to prepare each of the following solutions: a) 4500. ...
... 4. Calculate the molarity of each of the following solutions: a) 0.60 mol of NaCl dissolved in 1.6 L of solution. b) 25.2 g of potassium nitrate, KNO3, in enough water to make 150.0 mL of solution. 5. Calculate the number of grams of solute needed to prepare each of the following solutions: a) 4500. ...
Labs - newtunings.com
... nature of the reactants, surface area, and the presence of a catalyst. 3.4g A catalyst provides an alternate reaction pathway, which has a lower activation energy than an uncatalyzed reaction. 3.4h Some chemical and physical changes can reach equilibrium. 3.4i At equilibrium the rate of the forward ...
... nature of the reactants, surface area, and the presence of a catalyst. 3.4g A catalyst provides an alternate reaction pathway, which has a lower activation energy than an uncatalyzed reaction. 3.4h Some chemical and physical changes can reach equilibrium. 3.4i At equilibrium the rate of the forward ...
Ch. 1-- Matter and Change
... number of each kind of atom is the same. The ___________ Sometimes it appears that the reactant and product masses are not gas was probably a reactant or product in the equal, but a _______ reaction, and that is making the difference! ...
... number of each kind of atom is the same. The ___________ Sometimes it appears that the reactant and product masses are not gas was probably a reactant or product in the equal, but a _______ reaction, and that is making the difference! ...
Ch.5
... calculate the amount of product that will form during a reaction - called the THEORETICAL YIELD The amount of product that actually forms during a chemical reaction is called the ACTUAL YIELD The actual yield is often less than the ...
... calculate the amount of product that will form during a reaction - called the THEORETICAL YIELD The amount of product that actually forms during a chemical reaction is called the ACTUAL YIELD The actual yield is often less than the ...
Unit 6 – Chemical Reactions: Particles and Energy
... A Chemical Reaction is a process in which one or more substances change into new substance(s) having different physical and chemical properties. ...
... A Chemical Reaction is a process in which one or more substances change into new substance(s) having different physical and chemical properties. ...
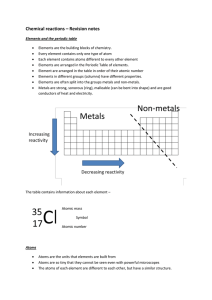
Chemical reactions and equations
... reactants and products 3) Add any symbols needed for state of matter, catalysts, heat, etc. 4) Count the number of atoms of each element. Include ...
... reactants and products 3) Add any symbols needed for state of matter, catalysts, heat, etc. 4) Count the number of atoms of each element. Include ...
Dr. Audrey Lugo`s AP Chemistry Course Syllabus
... 4. Non-ideal behavior (qualitative aspects) III. Reactions (35–40%) A. Reaction types 1. Acid-base reactions; concepts of Arrhenius, Brønsted-Lowry, and Lewis; coordination complexes; amphoterism 2. Precipitation reactions 3. Oxidation-reduction reactions a. Oxidation number b. The role of the elect ...
... 4. Non-ideal behavior (qualitative aspects) III. Reactions (35–40%) A. Reaction types 1. Acid-base reactions; concepts of Arrhenius, Brønsted-Lowry, and Lewis; coordination complexes; amphoterism 2. Precipitation reactions 3. Oxidation-reduction reactions a. Oxidation number b. The role of the elect ...
worksheet Ka Kb buffers Ksp
... If NaCl is added to a 0.010 M solution of AgNO3 in water at 25ºC, at what [Cl ] does precipitation of AgCl begin? Ksp for AgCl = 1.8x10-10 ...
... If NaCl is added to a 0.010 M solution of AgNO3 in water at 25ºC, at what [Cl ] does precipitation of AgCl begin? Ksp for AgCl = 1.8x10-10 ...
Chemical reaction

A chemical reaction is a process that leads to the transformation of one set of chemical substances to another. Classically, chemical reactions encompass changes that only involve the positions of electrons in the forming and breaking of chemical bonds between atoms, with no change to the nuclei (no change to the elements present), and can often be described by a chemical equation. Nuclear chemistry is a sub-discipline of chemistry that involves the chemical reactions of unstable and radioactive elements where both electronic and nuclear changes may occur.The substance (or substances) initially involved in a chemical reaction are called reactants or reagents. Chemical reactions are usually characterized by a chemical change, and they yield one or more products, which usually have properties different from the reactants. Reactions often consist of a sequence of individual sub-steps, the so-called elementary reactions, and the information on the precise course of action is part of the reaction mechanism. Chemical reactions are described with chemical equations, which symbolically present the starting materials, end products, and sometimes intermediate products and reaction conditions.Chemical reactions happen at a characteristic reaction rate at a given temperature and chemical concentration. Typically, reaction rates increase with increasing temperature because there is more thermal energy available to reach the activation energy necessary for breaking bonds between atoms.Reactions may proceed in the forward or reverse direction until they go to completion or reach equilibrium. Reactions that proceed in the forward direction to approach equilibrium are often described as spontaneous, requiring no input of free energy to go forward. Non-spontaneous reactions require input of free energy to go forward (examples include charging a battery by applying an external electrical power source, or photosynthesis driven by absorption of electromagnetic radiation in the form of sunlight).Different chemical reactions are used in combinations during chemical synthesis in order to obtain a desired product. In biochemistry, a consecutive series of chemical reactions (where the product of one reaction is the reactant of the next reaction) form metabolic pathways. These reactions are often catalyzed by protein enzymes. Enzymes increase the rates of biochemical reactions, so that metabolic syntheses and decompositions impossible under ordinary conditions can occur at the temperatures and concentrations present within a cell.The general concept of a chemical reaction has been extended to reactions between entities smaller than atoms, including nuclear reactions, radioactive decays, and reactions between elementary particles as described by quantum field theory.