Electron attachment to molecular clusters by collisional charge transfer
... and the electron affinity of the acceptor molecule. In previous work, this electron transfer process has been used with fast alkali atom beams produced by charge exchange or sputtering to determine electron affinities for many molecules." Here we adapt this method, with two essential amendments, to ...
... and the electron affinity of the acceptor molecule. In previous work, this electron transfer process has been used with fast alkali atom beams produced by charge exchange or sputtering to determine electron affinities for many molecules." Here we adapt this method, with two essential amendments, to ...
The SimSoup Guide - Chris Gordon
... Atoms are of different types. These types are called elements. An atom has a mass and a number of electrons that orbit a nucleus. Some of these are valence electrons that can participate in bonds with other atoms. ...
... Atoms are of different types. These types are called elements. An atom has a mass and a number of electrons that orbit a nucleus. Some of these are valence electrons that can participate in bonds with other atoms. ...
Chemistry - talcher autonomous college
... Kapustinskii expression for lattice energy. Madelung constant, Born-Haber cycle and its application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Resonanc ...
... Kapustinskii expression for lattice energy. Madelung constant, Born-Haber cycle and its application, Solvation energy. (ii) Covalent bond: Lewis structure, Valence Bond theory (Heitler-London approach). Energetics of hybridization, equivalent and non-equivalent hybrid orbitals. Bent’s rule, Resonanc ...
SQA CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
... All substances are made up of particles called atoms, ions or molecules, and these particles are constantly moving. The degree of movement depends upon the state of the substance. This is known as the "kinetic model" of matter. In any sample of solution, liquid or gas there is a range of kinetic ene ...
Chemistry 2 Higher revision mark scheme
... forms an oxide that reacts with alkalis with chlorine forms a molecular chloride 1 of these for 1 mark semi-conductor suggests in between this, or any other for 1further mark [NB Maximum of 2 for arguing metal/non-metal only] Under each head 1 wrong reason → maximum of 1 available 2 wrong reasons → ...
... forms an oxide that reacts with alkalis with chlorine forms a molecular chloride 1 of these for 1 mark semi-conductor suggests in between this, or any other for 1further mark [NB Maximum of 2 for arguing metal/non-metal only] Under each head 1 wrong reason → maximum of 1 available 2 wrong reasons → ...
Chapter 3 Molecules, Compounds, and Chemical Equations
... • Empirical Formulas describe the kinds of elements found in the compound and the ratio of their atoms. They do not describe how many atoms, the order of attachment, or the shape. The formulas for ionic compounds are empirical. ...
... • Empirical Formulas describe the kinds of elements found in the compound and the ratio of their atoms. They do not describe how many atoms, the order of attachment, or the shape. The formulas for ionic compounds are empirical. ...
Chem101 - Lecture 2 Elements Elements
... determined by comparing them to the mass of the carbon-12 isotope. • The unit of mass that is used is called the atomic mass unit and is represented by the symbol u. • The atomic mass unit is equal to exactly 1/12 the mass of the carbon-12 ...
... determined by comparing them to the mass of the carbon-12 isotope. • The unit of mass that is used is called the atomic mass unit and is represented by the symbol u. • The atomic mass unit is equal to exactly 1/12 the mass of the carbon-12 ...
`A` LEVEL H2 CHEMISTRY ORGANIC REACTIONS SUMMARY By
... (a) identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses (b) deduce the behaviour of beams of protons, neutrons and electrons in an electric field (c) describe the distribution of mass and charges within an atom (d) deduce the numbers of proton ...
... (a) identify and describe protons, neutrons and electrons in terms of their relative charges and relative masses (b) deduce the behaviour of beams of protons, neutrons and electrons in an electric field (c) describe the distribution of mass and charges within an atom (d) deduce the numbers of proton ...
CfE Higher Chemistry Unit 1: Chemical Changes and Structure
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
... the substance. This is known as the 'kinetic model' of matter. In any sample of solution, liquid or gas there is a range of kinetic energies known as an energy distribution. The collision theory of reactions suggests that, for a chemical reaction to occur, particles must collide. Simple collision is ...
Beginning Chemistry
... electrical energy, and nuclear energy. In general, it is possible to convert each of these forms of energy to others. Except for reactions in which the quantity of matter is changed, as in nuclear reactions, the law of conservation of energy is obeyed. In fact, many chemical reactions are carried ou ...
... electrical energy, and nuclear energy. In general, it is possible to convert each of these forms of energy to others. Except for reactions in which the quantity of matter is changed, as in nuclear reactions, the law of conservation of energy is obeyed. In fact, many chemical reactions are carried ou ...
Personal Tutor - Macmillan Learning
... components: precision and accuracy. Precision refers to how closely measurements of the same quantity agree. A high-precision measurement is one that produces very nearly the same result each time it is measured. Accuracy is how well measurements agree with the accepted or true value. It is possible ...
... components: precision and accuracy. Precision refers to how closely measurements of the same quantity agree. A high-precision measurement is one that produces very nearly the same result each time it is measured. Accuracy is how well measurements agree with the accepted or true value. It is possible ...
CHM 423 Coordination Chemistry
... Coordination Chemistry involves the study of coordination compounds, their structures, properties and applications. The term ‘Coordination’ used to describe these compounds stems from the nature of chemical bond that leads to the formation of these compounds. This chemical bond called coordinate cov ...
... Coordination Chemistry involves the study of coordination compounds, their structures, properties and applications. The term ‘Coordination’ used to describe these compounds stems from the nature of chemical bond that leads to the formation of these compounds. This chemical bond called coordinate cov ...
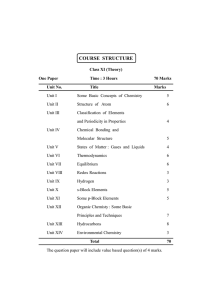
COURSE STRUCTURE
... For a given value of ‘l’, ml has a total of (2l + 1) values ranging from l to +l including ‘0’. It determines the orientation of orbital. (4) Magnetic spin quantum number (ms) It can take the values of +½ or ½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons ...
... For a given value of ‘l’, ml has a total of (2l + 1) values ranging from l to +l including ‘0’. It determines the orientation of orbital. (4) Magnetic spin quantum number (ms) It can take the values of +½ or ½ and determines the orientation of spin. Pauli’s Exclusion Principle : ‘‘No two electrons ...
Chemical-Principles-7th-Edition-Zumdahl-Test-Bank
... A) the ratio of the masses of the elements in a compound is always the same. B) it is not possible for the same two elements to form more than one compound. C) if the same two elements form two different compounds, they do so in the same ratio. D) the total mass after a chemical change is the same a ...
... A) the ratio of the masses of the elements in a compound is always the same. B) it is not possible for the same two elements to form more than one compound. C) if the same two elements form two different compounds, they do so in the same ratio. D) the total mass after a chemical change is the same a ...
chapter twenty-one transition metals and coordination chemistry
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
... The crystal field diagrams are different because the geometries of where the ligands point is different. The tetrahedrally oriented ligands point differently in relationship to the d-orbitals than do the octahedrally oriented ligands. Plus, we have more ligands in an octahedral complex. See Figure 2 ...
Organic Chemistry - University of California, Riverside
... The general formulas R-X, R-OH, and R-NH2 suggest two different ways to view these classes of compounds. One way is for us to imagine that an alkyl group R replaces H in HNH2 (ammonia), H-OH (water), and the hydrogen halides H-X (X = F, Cl, Br, or I). We can also view haloalkanes (R-X), alcohols (R- ...
... The general formulas R-X, R-OH, and R-NH2 suggest two different ways to view these classes of compounds. One way is for us to imagine that an alkyl group R replaces H in HNH2 (ammonia), H-OH (water), and the hydrogen halides H-X (X = F, Cl, Br, or I). We can also view haloalkanes (R-X), alcohols (R- ...
Fluorinated Butatrienes - diss.fu-berlin.de
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
... wurde ein Enin-Isomer entdeckt, das erstaunlicherweise stabiler als sein Butatrien Isomer ist, obwohl es an der Dreifachbindung fluoriert ist. Eben jene Fluorierung an der Dreifachbindung ist eigentlich notwendig um die Energie fluorierter But-1-en-3-ine relativ ...
Chapter 3 Molecules Molecules, Compounds, and Chemical
... P S, S and trace amounts of other elements. The main element that is the focus of organic chemistry is carbon. ...
... P S, S and trace amounts of other elements. The main element that is the focus of organic chemistry is carbon. ...
3: Haloalkanes, Alcohols, Ethers, and Amines
... is in the first row of the periodic table. As a result, it forms only one chemical bond and has no unshared electrons (Figure [graphic 3.3]). Chemical Reactivity of Unshared Electron Pairs. Unshared electron pairs are chemically reactive and can participate in chemical bond formation. For example, w ...
... is in the first row of the periodic table. As a result, it forms only one chemical bond and has no unshared electrons (Figure [graphic 3.3]). Chemical Reactivity of Unshared Electron Pairs. Unshared electron pairs are chemically reactive and can participate in chemical bond formation. For example, w ...
Regents Chemistry - New York Science Teacher
... substances have molecules that contain two carbon atoms, one oxygen atom, and six hydrogen atoms. These two substances must be ...
... substances have molecules that contain two carbon atoms, one oxygen atom, and six hydrogen atoms. These two substances must be ...
- Free Documents
... he pays some additional attention to the details and nomenclature surrounding a particular spectroscopic subject. after studying this book. No special background is necessary for the study of this book. is but should also provide that comfortable feeling of knowing quotwhat happeningquot when spectr ...
... he pays some additional attention to the details and nomenclature surrounding a particular spectroscopic subject. after studying this book. No special background is necessary for the study of this book. is but should also provide that comfortable feeling of knowing quotwhat happeningquot when spectr ...
Here
... (b) The testosterone molecular ion peak is at 288, whereas the clostebol molecular ion peak is at 322. There are peaks at both these positions, though the peak at 322 is smaller. It would appear that this mass spectrum does not rule out clostebol. However, chlorinated compounds generally show ...
... (b) The testosterone molecular ion peak is at 288, whereas the clostebol molecular ion peak is at 322. There are peaks at both these positions, though the peak at 322 is smaller. It would appear that this mass spectrum does not rule out clostebol. However, chlorinated compounds generally show ...
Chemical Reactivity as Described by Quantum Chemical Methods
... Schrödinger's equation occupied a central position in this new theory and, although later on complemented by its relativistic analogue by Dirac, stood the test of time and has been for now 75 years the central equation for the description both of the internal structure of atoms and molecules and the ...
... Schrödinger's equation occupied a central position in this new theory and, although later on complemented by its relativistic analogue by Dirac, stood the test of time and has been for now 75 years the central equation for the description both of the internal structure of atoms and molecules and the ...
Molecular orbital diagram
A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical bonding in molecules in terms of molecular orbital theory in general and the linear combination of atomic orbitals (LCAO) molecular orbital method in particular. A fundamental principle of these theories is that as atoms bond to form molecules, a certain number of atomic orbitals combine to form the same number of molecular orbitals, although the electrons involved may be redistributed among the orbitals. This tool is very well suited for simple diatomic molecules such as dihydrogen, dioxygen, and carbon monoxide but becomes more complex when discussing even comparatively simple polyatomic molecules, such as methane. MO diagrams can explain why some molecules exist and others do not. They can also predict bond strength, as well as the electronic transitions that can take place.